Scalable Preparation Method of Avanafil Intermediates for Global Pharmaceutical Supply Chains
Scalable Preparation Method of Avanafil Intermediates for Global Pharmaceutical Supply Chains
The global demand for high-performance phosphodiesterase-5 (PDE-5) inhibitors continues to drive innovation in the pharmaceutical intermediate sector, specifically for compounds like Avanafil. A pivotal advancement in this field is detailed in patent CN103254180A, which discloses a novel preparation method for Avanafil (I). This technology represents a significant departure from traditional synthetic pathways by utilizing a streamlined approach that begins with 6-amino-1,2-dihydropyrimidine-2-one-5-carboxylic acid ethyl ester (XII). Unlike earlier methodologies that relied on complex multi-step sequences involving difficult-to-handle reagents, this invention focuses on atom economy and green chemistry principles. The core breakthrough lies in the strategic substitution and condensation reactions that construct the final molecule under mild thermal conditions, thereby enhancing both safety and efficiency. For R&D directors and supply chain managers, this patent offers a robust framework for producing high-purity API intermediates with reduced operational complexity. By shifting the synthetic paradigm away from cryogenic dependencies, this method addresses critical bottlenecks in the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
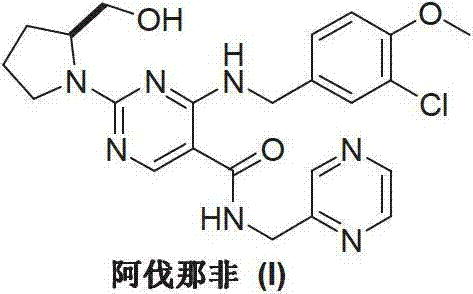
Prior art methods, such as those disclosed in international patents WO0183460 and WO0119802, have historically presented substantial challenges for industrial manufacturing. These conventional routes typically rely on the construction of the pyrimidine ring parent nucleus through intricate cyclization reactions involving methylthio urea and ethoxy methyne diethyl malonate. A critical drawback of these legacy processes is the requirement for extreme reaction conditions, particularly the use of n-Butyl Lithium at ultra-low temperatures around -78°C to generate carbanions for nucleophilic addition. Such cryogenic conditions necessitate specialized refrigeration equipment and strict anhydrous and oxygen-free environments, which drastically increase capital expenditure and operational costs. Furthermore, the involvement of multiple metal reagent steps often leads to complex separation difficulties and the potential for heavy metal contamination, posing significant hurdles for achieving the stringent purity specifications required for pharmaceutical applications. The cumulative effect of these factors renders the traditional synthetic routes economically inefficient and technically risky for large-scale production.
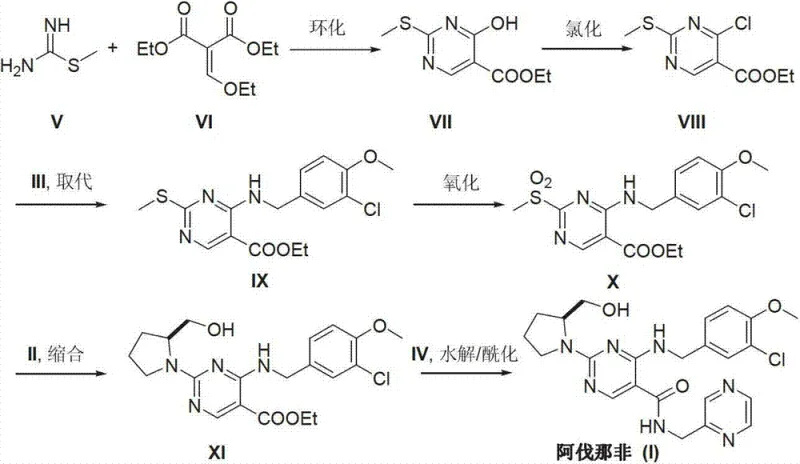
The Novel Approach
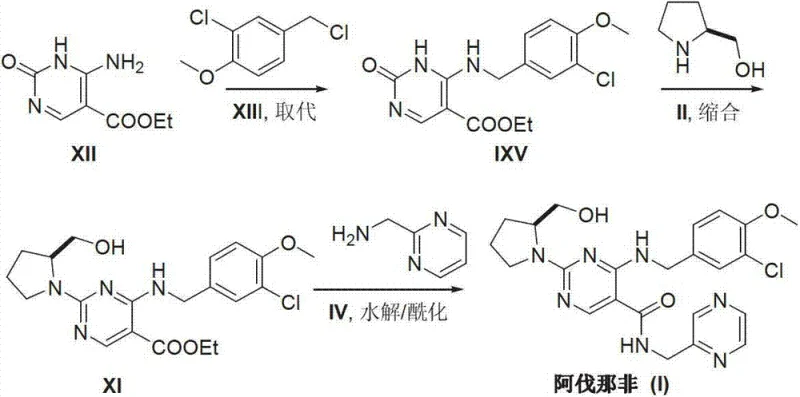
In stark contrast to the cumbersome legacy pathways, the novel approach described in CN103254180A introduces a simplified and economically viable strategy. This method initiates the synthesis with the commercially accessible 6-amino-1,2-dihydropyrimidine-2-one-5-carboxylic acid ethyl ester (XII), bypassing the need for de novo ring construction under harsh conditions. The process involves a direct substitution reaction with 3-chloro-4-methoxybenzyl chloride (XIII) followed by a condensation with S-hydroxymethyl pyrrolidine (II). This strategic redesign eliminates the need for ultra-low temperature reactors and hazardous metal reagents, replacing them with standard thermal conditions ranging from 50°C to 80°C. The simplicity of the reaction sequence not only accelerates the production timeline but also significantly reduces the generation of hazardous waste, aligning with modern environmental compliance standards. By focusing on readily available starting materials and straightforward workup procedures, this novel approach provides a reliable foundation for the cost reduction in pharmaceutical manufacturing of PDE-5 inhibitors.
Mechanistic Insights into Nucleophilic Substitution and Condensation
The chemical efficacy of this new route is grounded in the precise control of nucleophilic substitution and peptide-like condensation mechanisms. The initial step involves the nucleophilic attack of the amino group on the pyrimidine ring of compound (XII) onto the benzylic carbon of 3-chloro-4-methoxybenzyl chloride (XIII). This reaction is facilitated by the presence of an acid-binding agent such as triethylamine, which neutralizes the generated hydrogen chloride and drives the equilibrium toward the formation of the intermediate (IXV). The choice of solvent, typically ethanol, plays a crucial role in solubilizing the reactants while maintaining a polarity that supports the SN2-type displacement mechanism without promoting unwanted side reactions. Following this, the condensation step utilizes advanced coupling reagents like BOP (benzotriazole-1-yloxytris(dimethylamino)phosphonium hexafluorophosphate) to activate the carboxylic acid moiety. This activation allows for the efficient formation of the amide bond with the chiral pyrrolidine side chain (II), ensuring high stereochemical integrity which is vital for the biological activity of the final drug substance.
Impurity control is inherently managed through the selection of mild reaction conditions and specific reagents that minimize degradation pathways. In traditional methods, the use of strong bases and extreme temperatures often leads to the hydrolysis of ester groups or the racemization of chiral centers. However, in this optimized protocol, the reaction temperature is carefully maintained between 50°C and 60°C during the condensation phase, preventing thermal decomposition. Furthermore, the use of specific promoters like DBU (1,8-diazabicyclo[5.4.0]undec-7-ene) enhances the reaction rate without compromising the stability of the sensitive pyrimidine core. The final hydrolysis and acylation steps are conducted under controlled pH conditions, allowing for the selective transformation of the ester group while preserving the newly formed amide linkages. This meticulous control over the reaction environment ensures that the impurity profile remains within acceptable limits, reducing the burden on downstream purification processes and ultimately yielding a high-purity product suitable for clinical use.
How to Synthesize Avanafil Efficiently
The synthesis of Avanafil via this patented route offers a practical blueprint for laboratories and manufacturing facilities aiming to optimize their production capabilities. The process is designed to be operationally simple, utilizing standard glassware and heating equipment rather than specialized cryogenic setups. The initial substitution reaction can be performed in common solvents like ethanol with simple acid-base workups to isolate the intermediate. Subsequent condensation and acylation steps leverage widely available coupling agents and bases, making the technology transfer to pilot and commercial scales straightforward. For technical teams looking to implement this methodology, the key lies in maintaining strict stoichiometric control and monitoring reaction progress via TLC or HPLC to ensure complete conversion at each stage. The detailed standardized synthesis steps for implementing this efficient route are outlined below.
- Perform a substitution reaction between 6-amino-1,2-dihydropyrimidine-2-one-5-carboxylic acid ethyl ester (XII) and 3-chloro-4-methoxybenzyl chloride (XIII) using triethylamine as a base in ethanol to obtain intermediate IXV.
- Conduct a condensation reaction between intermediate IXV and S-hydroxymethyl pyrrolidine (II) using BOP as a condensing agent and DBU as a promoter in acetonitrile to generate intermediate XI.
- Hydrolyze intermediate XI followed by an acylation reaction with 2-methylamino pyrimidine (IV) using HOBt and DIEA to yield the final Avanafil product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates into tangible strategic benefits that extend beyond mere technical feasibility. The primary value proposition lies in the drastic simplification of the supply chain for raw materials. By utilizing 6-amino-1,2-dihydropyrimidine-2-one-5-carboxylic acid ethyl ester as a starting block, manufacturers can source materials from a broader base of suppliers, reducing dependency on niche vendors who provide specialized organometallic reagents. This diversification of the supply base enhances supply chain reliability and mitigates the risk of production stoppages due to raw material shortages. Moreover, the elimination of ultra-low temperature requirements removes the need for expensive energy-intensive cooling infrastructure, leading to substantial cost savings in utility consumption. The overall process efficiency is further bolstered by the reduced number of purification steps, which decreases solvent usage and waste disposal costs, contributing to a more sustainable and economically attractive manufacturing model.
- Cost Reduction in Manufacturing: The economic impact of this method is profound, primarily driven by the removal of costly cryogenic operations and expensive metal catalysts. Traditional routes requiring n-Butyl Lithium at -78°C incur high energy costs for refrigeration and require specialized containment systems to handle pyrophoric reagents safely. By shifting to a thermal process operating at moderate temperatures (50-80°C), the facility can utilize standard heating systems, resulting in significantly reduced energy expenditures. Additionally, the use of common organic solvents like ethanol and acetonitrile, which are cheaper and easier to recycle than the specialized solvents often needed for organometallic chemistry, further lowers the variable cost per kilogram. The streamlined workflow also reduces labor hours associated with complex setup and teardown procedures, delivering comprehensive cost optimization across the entire production lifecycle.
- Enhanced Supply Chain Reliability: From a logistics perspective, this route offers superior stability and predictability. The starting materials, such as 3-chloro-4-methoxybenzyl chloride and the pyrimidine ester, are commodity chemicals with established global supply networks, ensuring consistent availability and competitive pricing. This contrasts sharply with older methods that might rely on custom-synthesized intermediates with long lead times. The robustness of the reaction conditions means that the process is less susceptible to minor fluctuations in environmental parameters, leading to higher batch-to-batch consistency. For supply chain planners, this reliability translates into shorter lead times for high-purity pharmaceutical intermediates and the ability to maintain leaner inventory levels without risking stockouts. The simplified purification process also accelerates the release of finished goods, improving cash flow and responsiveness to market demand.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor often reveals hidden complexities, but this method is explicitly designed for industrial amplification. The absence of hazardous reagents like phosphorus oxychloride in the main sequence (used in some prior art for chlorination) and the avoidance of heavy metal catalysts simplify the waste treatment protocols. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the environmental footprint of the manufacturing site. The reaction exotherms are manageable under standard cooling conditions, eliminating the need for complex heat exchange systems required for highly reactive organometallic additions. Consequently, the process can be scaled up to multi-ton capacities with minimal engineering modifications, ensuring that the commercial scale-up of complex API intermediates proceeds smoothly and safely while meeting rigorous environmental standards.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is crucial for stakeholders evaluating its implementation. The following questions address common inquiries regarding the operational parameters and strategic benefits of this patented method. These answers are derived directly from the technical specifications and comparative data provided in the patent documentation, offering clarity on how this route outperforms historical precedents. Whether you are concerned about reaction safety, raw material sourcing, or final product quality, the insights below provide a comprehensive overview of the technology's capabilities.
Q: What are the primary advantages of this new Avanafil synthesis route over prior art?
A: The primary advantage is the elimination of harsh cryogenic conditions (-78°C) and expensive metal reagents like n-Butyl Lithium required in previous methods. This new route utilizes readily available starting materials and mild thermal conditions (50-80°C), significantly simplifying the process and reducing energy consumption.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method is suitable for industrialization amplification. The use of common solvents like ethanol and acetonitrile, along with standard acid-base workups, avoids the complex separation difficulties and anhydrous/oxygen-free constraints of older methods, making it highly scalable.
Q: What represents the key cost-saving factor in this manufacturing method?
A: Cost savings are driven by the use of economically accessible raw materials such as 6-amino-1,2-dihydropyrimidine-2-one-5-carboxylic acid ethyl ester and the removal of specialized low-temperature reactor requirements. Additionally, the streamlined three-step sequence reduces overall processing time and waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Avanafil Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the methodology described in CN103254180A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering high-purity Avanafil intermediates that meet stringent purity specifications through our rigorous QC labs and state-of-the-art manufacturing facilities. By leveraging this efficient and environmentally friendly synthesis strategy, we ensure that our clients receive a product that is not only cost-effective but also compliant with the highest international quality standards. Our dedication to process optimization allows us to offer a reliable supply of complex pharmaceutical intermediates that support your drug development and commercialization goals.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out to us to request specific COA data and route feasibility assessments for your upcoming projects. By partnering with NINGBO INNO PHARMCHEM, you gain access to a wealth of technical expertise and a robust production capacity designed to support your long-term growth in the pharmaceutical sector.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
