Advanced Manufacturing of 2,4,5-Trifluorophenylacetic Acid for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks robust synthetic pathways for critical intermediates, particularly for high-volume antidiabetic medications like Sitagliptin. Patent CN113004142A introduces a groundbreaking preparation method for 2,4,5-trifluorophenylacetic acid, a pivotal building block in modern medicinal chemistry. This technical disclosure outlines a novel multi-step synthesis originating from m-dichlorobenzene, strategically bypassing the limitations of traditional fluorination and Grignard-based methodologies. By integrating nucleophilic fluorination with a refined diazotization-cracking sequence, this approach delivers exceptional control over impurity profiles while maintaining operational simplicity. For R&D directors and procurement specialists, understanding this technology is essential for securing a reliable pharmaceutical intermediate supplier capable of meeting stringent quality standards. The process not only enhances chemical efficiency but also aligns with modern green chemistry principles, offering a sustainable alternative for cost reduction in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,4,5-trifluorophenylacetic acid has relied on routes fraught with significant industrial hazards and economic inefficiencies. Traditional methods often utilize 1,2,4-trifluorobenzene as a starting material, requiring Friedel-Crafts acetylation or chloromethylation steps that generate substantial amounts of corrosive acidic wastewater and odorous sulfur-containing by-products. Furthermore, alternative pathways involving Grignard reagents necessitate strict anhydrous and anaerobic conditions, drastically increasing operational complexity and energy consumption. The use of extremely toxic cyanides in chloromethylation-cyanidation sequences poses severe safety risks, complicating regulatory compliance and waste management. Additionally, many existing processes suffer from low overall yields, often struggling to exceed 50% due to side reactions during fluorination or rearrangement steps. These technical bottlenecks result in higher production costs and inconsistent supply continuity, creating vulnerabilities for downstream pharmaceutical manufacturers seeking high-purity OLED material or API precursors.
The Novel Approach
In stark contrast, the methodology disclosed in CN113004142A leverages m-dichlorobenzene as a cost-effective and readily available feedstock to construct the trifluoro-aromatic core. This innovative route replaces hazardous cyanidation with a safer bromination and diethyl malonate substitution strategy, effectively eliminating the need for toxic reagents. The process incorporates solvent-free conditions for critical steps such as nitration, hydrogenation, and bromination, which simplifies post-reaction separation through static layering or distillation. By optimizing reaction temperatures and utilizing specific phase transfer catalysts, the new method achieves a total yield of approximately 58.2%, with individual steps frequently surpassing 90% efficiency. This streamlined workflow not only reduces the environmental footprint but also enhances the commercial scale-up of complex polymer additives and pharmaceutical intermediates. The ability to operate without stringent water-free constraints in key stages significantly lowers infrastructure requirements, making it an ideal candidate for large-scale industrial adoption.
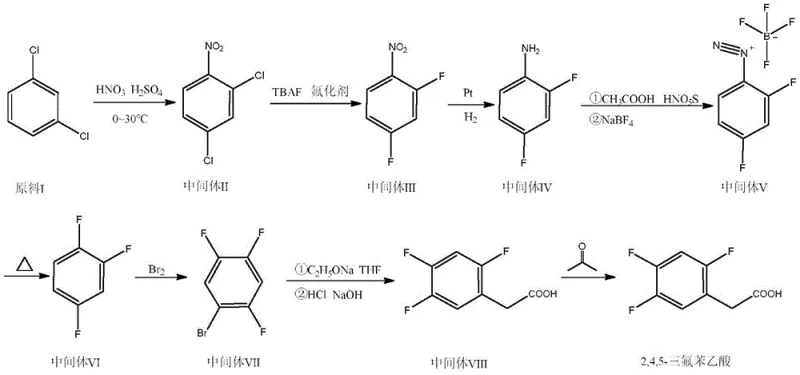
Mechanistic Insights into Nucleophilic Fluorination and Diazotization
The core chemical innovation lies in the precise execution of nucleophilic aromatic substitution using potassium fluoride facilitated by advanced phase transfer catalysts. In the second step of the synthesis, the nitro-dichlorobenzene intermediate undergoes fluorination in an aprotic solvent at elevated temperatures, typically around 160°C. The selection of catalysts such as tetramethylammonium bromide or trihexyltetradecyltetrafluorophosphonium borate is critical for activating the fluoride ion, enabling it to displace the chlorine atom efficiently. This mechanistic pathway avoids the low yields associated with direct fluorination of aniline derivatives seen in older patents. Following fluorination, the nitro group is reduced to an amine via catalytic hydrogenation, setting the stage for the diazotization-cracking sequence. This sequence converts the amine into a diazonium salt using nitrosyl sulfuric acid and sodium fluoborate, which upon thermal cracking at 250°C, releases nitrogen gas and installs the third fluorine atom. This specific arrangement ensures the correct 2,4,5-substitution pattern required for biological activity in DPP-IV inhibitors.
Impurity control is meticulously managed through the final substitution and hydrolysis stages, which define the quality of the reducing lead time for high-purity pharmaceutical intermediates. The brominated intermediate reacts with diethyl malonate in the presence of sodium ethoxide, where strict stoichiometric control prevents over-alkylation or polymerization. The reaction mechanism involves the formation of a monobasic substituted sodium salt, which selectively attacks the benzyl bromide position. Subsequent hydrolysis and decarboxylation are performed under controlled acidic conditions to yield the final acetic acid derivative. The patent specifies that maintaining a molar ratio of intermediate VII to sodium ethoxide to diethyl malonate at 1:3:3 is vital for maximizing yield and minimizing side products. This level of mechanistic precision ensures that the final product achieves a purity of 99.5%, satisfying the rigorous specifications demanded by global regulatory bodies for human therapeutic use.
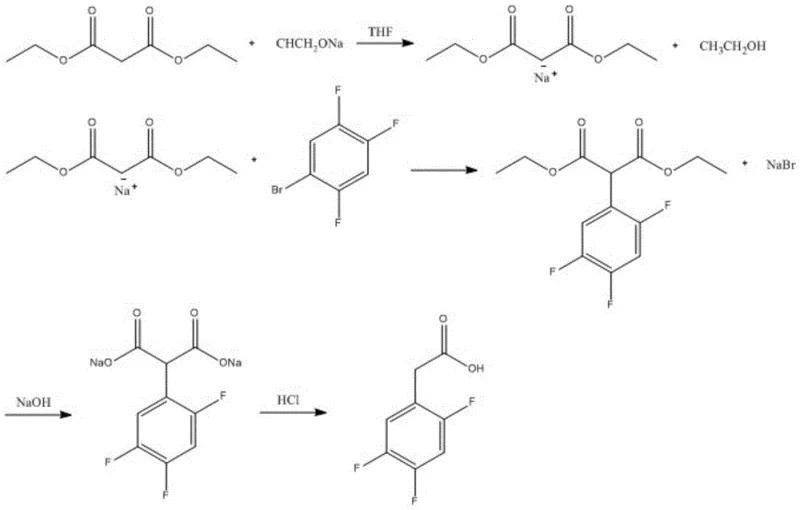
How to Synthesize 2,4,5-Trifluorophenylacetic Acid Efficiently
Implementing this synthesis requires careful attention to reaction parameters and safety protocols to ensure reproducibility and safety at scale. The process begins with the nitration of m-dichlorobenzene at controlled low temperatures to prevent polynitration, followed by a high-temperature fluorination step that demands robust pressure-rated equipment. Operators must monitor the diazotization phase closely, as the thermal cracking of the diazonium salt generates gaseous by-products that require efficient scrubbing systems. The final substitution step involves handling reactive alkoxides and brominated intermediates, necessitating inert atmosphere conditions to prevent moisture ingress which could degrade the reagents. While the general workflow is outlined here, the detailed standardized synthetic steps see the guide below for specific temperature ramps, addition rates, and workup procedures that guarantee optimal performance. Adhering to these optimized conditions is essential for achieving the reported yields and purity levels that make this route commercially viable.
- Nitration of m-dichlorobenzene followed by nucleophilic fluorination using potassium fluoride and phase transfer catalysts.
- Hydrogenation and diazotization-cracking sequence to establish the trifluoro-benzene core structure.
- Bromination and subsequent substitution with diethyl malonate, concluding with hydrolysis and purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this novel manufacturing process offers profound benefits that extend beyond simple chemical conversion. By shifting the raw material base to m-dichlorobenzene, manufacturers can decouple their supply chain from the volatile pricing and availability issues associated with specialized fluorinated anilines or benzyl chlorides. The elimination of toxic cyanides and the reduction of solvent-intensive steps translate directly into lower waste disposal costs and reduced regulatory burden, facilitating faster site approvals and continuous operation. For procurement managers, this means a more stable pricing structure and reduced risk of production stoppages due to environmental compliance audits. The simplified workup procedures, such as static layering and distillation instead of complex chromatographic separations, further drive down operational expenditures. These factors collectively contribute to substantial cost savings and enhanced supply chain reliability, ensuring that downstream partners receive consistent quality without interruption.
- Cost Reduction in Manufacturing: The economic advantages of this route are driven by the use of commodity chemicals and the minimization of expensive catalysts and solvents. By avoiding the need for cryogenic conditions required by Grignard reactions and eliminating the costly removal of heavy metal residues, the overall production cost is significantly reduced. The high yield of the final hydrolysis step, reported at 98%, ensures maximum material throughput, minimizing waste of valuable intermediates. Furthermore, the ability to recover and recycle solvents like tetrahydrofuran and diphenyl ether adds another layer of financial efficiency. These cumulative effects result in a highly competitive cost structure that allows for better margin management in the final API production.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the accessibility of starting materials and the robustness of the reaction conditions. Unlike routes dependent on single-source specialty reagents, m-dichlorobenzene is widely produced globally, mitigating the risk of raw material shortages. The process tolerance to minor variations in temperature and pressure makes it less susceptible to batch failures, ensuring a steady flow of goods to customers. This reliability is crucial for pharmaceutical companies managing just-in-time inventory systems for blockbuster drugs. By partnering with a manufacturer utilizing this technology, buyers can secure a long-term supply agreement that withstands market fluctuations and geopolitical disruptions.
- Scalability and Environmental Compliance: The design of this synthesis inherently supports scalability, with solvent-free steps reducing the volume of reactor capacity needed per kilogram of product. The reduction in hazardous waste generation aligns with increasingly strict global environmental regulations, future-proofing the manufacturing site against tighter emission standards. The absence of sulfur-containing odorous by-products improves workplace safety and community relations, which is often a hidden cost in chemical manufacturing. This green chemistry profile not only satisfies corporate sustainability goals but also streamlines the permitting process for capacity expansion, allowing for rapid response to increased market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this intermediate. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on performance metrics and operational feasibility. Understanding these details helps stakeholders evaluate the suitability of this material for their specific formulation needs. The responses cover aspects of purity, safety, and scalability, which are critical for decision-making in pharmaceutical development and procurement.
Q: What is the purity level achievable with this new synthesis method?
A: The novel preparation method described in patent CN113004142A achieves a final product purity of up to 99.5%, with intermediate purities consistently exceeding 97.5% after purification treatments.
Q: How does this process improve environmental compliance compared to traditional routes?
A: This route eliminates the need for highly toxic cyanides and reduces solvent usage in key steps like nitration and hydrogenation, significantly lowering three-waste generation and enhancing safety profiles.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process utilizes cheap and easily obtainable raw materials like m-dichlorobenzene and avoids harsh anhydrous conditions required by Grignard reactions, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4,5-Trifluorophenylacetic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to verify that every batch meets the highest international standards. Our facility is equipped to handle the specific requirements of fluorinated chemistry, including corrosion-resistant equipment and advanced waste treatment systems, guaranteeing both product quality and environmental stewardship. By leveraging our expertise in process optimization, we can help you secure a stable supply of this essential Sitagliptin intermediate.
We invite you to collaborate with us to optimize your supply chain and reduce overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential partners to contact us to request specific COA data and route feasibility assessments, allowing you to validate our capabilities against your internal standards. Together, we can drive innovation and efficiency in the pharmaceutical sector, ensuring that patients have access to affordable and effective treatments. Reach out today to discuss how our advanced synthesis technologies can support your long-term strategic goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
