Advanced One-Step Grignard Route for Commercial Citalopram Production
Advanced One-Step Grignard Route for Commercial Citalopram Production
The global demand for high-quality antidepressants continues to drive innovation in pharmaceutical manufacturing, particularly for established drugs like Citalopram. Patent CN1133633C introduces a groundbreaking methodology that fundamentally alters the synthetic landscape for this critical selective serotonin reuptake inhibitor (SSRI). Unlike legacy processes that rely on multi-step sequences involving hazardous reagents, this invention leverages a sophisticated one-step Grignard reaction coupled with spontaneous cyclization. This technical advancement not only streamlines the production workflow but also addresses long-standing concerns regarding impurity profiles and environmental impact. For R&D directors and procurement specialists alike, understanding the nuances of this pathway is essential for securing a reliable citalopram intermediate supplier capable of meeting modern regulatory standards. The core of this innovation lies in the utilization of a specifically protected ketone intermediate, which serves as the pivotal substrate for the final assembly of the isobenzofuran core.
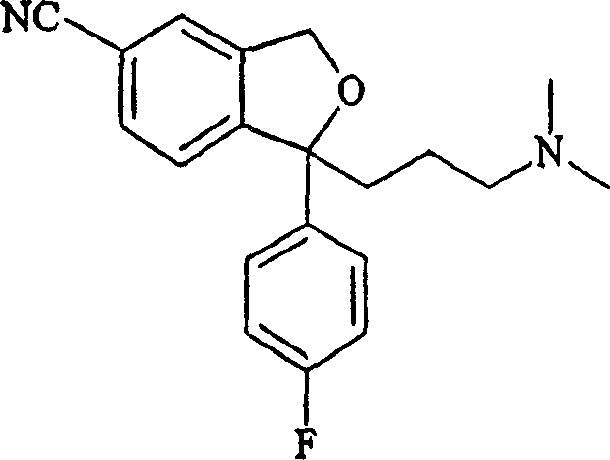
Citalopram, chemically known as 1-[3-(dimethylamino)propyl]-1-(4-fluorophenyl)-1,3-dihydro-5-isobenzofurancarbonitrile, has been a cornerstone in psychiatric pharmacotherapy for decades. Its therapeutic efficacy is well-documented, yet the complexity of its synthesis has often posed challenges for generic manufacturers seeking cost-effective routes. The patent data reveals that by manipulating the oxidation state and protecting groups of the precursor molecules, the final ring-closing step can be achieved with remarkable efficiency. This shift represents a significant leap forward in cost reduction in antidepressant manufacturing, moving away from stoichiometric heavy metal usage towards more catalytic and atom-economical transformations. As we delve deeper into the technical specifics, it becomes clear that this process offers a robust platform for the commercial scale-up of complex psychiatric drug intermediates, ensuring consistent supply continuity for the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Citalopram has been plagued by several inherent inefficiencies that impact both cost and safety. Early methods, such as those described in DE 2,657,271, necessitated the use of cuprous cyanide to introduce the critical nitrile functionality via a Rosenmund-von Braun type reaction. This approach presents severe drawbacks, including the generation of toxic copper-containing waste streams that require expensive remediation and disposal protocols. Furthermore, alternative pathways outlined in US Patent 4,650,884 relied on harsh dehydration conditions using concentrated sulfuric acid to effect ring closure. These aggressive acidic environments often lead to the formation of difficult-to-remove byproducts and pose significant corrosion risks to reactor vessels, thereby increasing maintenance costs and downtime. Additionally, the multi-step nature of these conventional routes, often involving separate Grignard additions followed by distinct cyclization events, results in cumulative yield losses and extended production cycles. For supply chain heads, these factors translate into higher volatility in lead times and increased exposure to regulatory scrutiny regarding waste management.
The Novel Approach
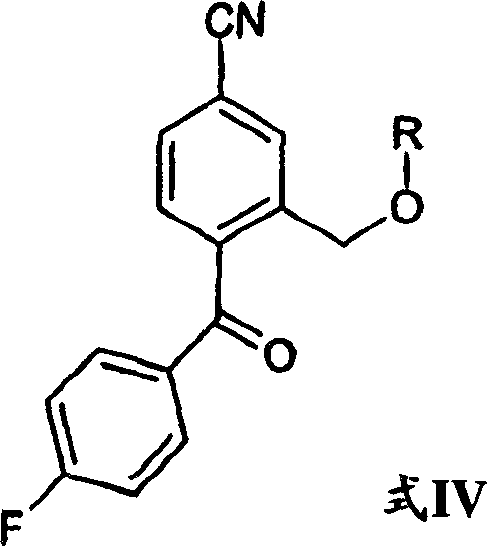
In stark contrast to these legacy techniques, the process disclosed in CN1133633C offers a streamlined solution that elegantly bypasses these bottlenecks. The heart of this novelty is the reaction of a specific protected ketone, designated as Formula IV, with 3-(N,N-dimethylamino)propylmagnesium halide. Remarkably, the patent demonstrates that the Grignard adduct formed in this reaction undergoes spontaneous ring closure to yield the target isobenzofuran structure without the need for external dehydrating agents or transition metal catalysts. This telescoping of the addition and cyclization steps into a single operational unit drastically simplifies the process flow. By eliminating the cuprous cyanide step entirely, the process removes a major source of heavy metal contamination, simplifying the purification train and enhancing the overall purity of the high-purity citalopram produced. Furthermore, the avoidance of concentrated sulfuric acid mitigates safety hazards and equipment corrosion, allowing for the use of standard stainless-steel reactors. This methodological shift provides a compelling value proposition for manufacturers aiming to optimize their production economics while adhering to stricter environmental, health, and safety (EHS) guidelines.
Mechanistic Insights into Spontaneous Cyclization and Grignard Addition
To fully appreciate the technical superiority of this route, one must examine the mechanistic underpinnings of the transformation. The reaction begins with the nucleophilic attack of the 3-(N,N-dimethylamino)propylmagnesium halide on the carbonyl carbon of the Formula IV intermediate. This ketone intermediate is uniquely designed with a protected hydroxymethyl group ortho to the carbonyl functionality. Upon addition of the Grignard reagent, a tertiary alcohol intermediate is generated in situ. Under the reaction conditions, typically maintained at low temperatures initially and then allowed to warm or worked up carefully, the proximity of the newly formed hydroxyl group (after deprotection or concurrent with the reaction dynamics) facilitates an intramolecular nucleophilic attack. This internal cyclization displaces the leaving group or interacts with the nitrile precursor functionality to close the five-membered lactol ring, which subsequently stabilizes into the isobenzofuran system. The spontaneity of this closure is driven by the thermodynamic stability of the resulting heterocyclic ring and the specific electronic activation provided by the adjacent substituents. This mechanism ensures that the stereochemical integrity and structural fidelity of the molecule are maintained throughout the critical bond-forming event.
![Preparation of the key Formula IV intermediate from 5-cyano-2-benzo[c]furanone via Grignard addition and protection](/insights/img/citalopram-synthesis-grignard-pharma-supplier-20260315093009-06.webp)
Controlling impurities in this Grignard-based sequence is achieved through precise management of the protecting group strategy. The patent highlights that the 'R' group in Formula IV, which acts as the protecting group for the hydroxymethyl moiety, plays a crucial role. Preferred embodiments utilize acyl groups such as pivaloyl or acetyl, which are stable enough to withstand the Grignard conditions yet can be managed during the workup. By selecting appropriate acyl chlorides for the protection step, manufacturers can minimize the formation of over-alkylation byproducts or elimination side reactions that often plague amine-containing Grignard syntheses. The oxidation step to generate the ketone precursor, preferably using sodium tungstate and hydrogen peroxide, is another critical control point. This catalytic oxidation system is highly selective for secondary alcohols, ensuring that the primary hydroxymethyl group remains available for protection without over-oxidation to carboxylic acids. This level of mechanistic control allows R&D teams to define tight specifications for raw materials and intermediates, ensuring that the final API meets stringent pharmacopeial standards for related substances.
How to Synthesize Citalopram Efficiently
The practical implementation of this synthesis route requires a disciplined approach to reaction engineering and unit operations. The process generally initiates with the preparation of the key ketone intermediate, which can be derived either from the oxidation of a diol precursor or directly from 5-cyano-2-benzo[c]furanone via Grignard addition and subsequent protection. Once the Formula IV intermediate is secured with high purity, it is dissolved in an anhydrous solvent such as tetrahydrofuran (THF) and cooled to control the exotherm of the subsequent Grignard addition. The 3-(N,N-dimethylamino)propylmagnesium chloride is then added dropwise, maintaining the temperature within a narrow window to prevent degradation of the sensitive amine functionality. Following the addition, the reaction mixture is stirred to allow for complete conversion and spontaneous cyclization. The workup typically involves quenching with a saturated ammonium chloride solution, followed by extraction and crystallization. Detailed standard operating procedures for each of these stages are critical for reproducibility.
- Preparation of the key protected ketone intermediate (Formula IV) via oxidation of the corresponding diol or Grignard addition to 5-cyano-2-benzo[c]furanone followed by protection.
- Reaction of the protected ketone with 3-(N,N-dimethylamino)propylmagnesium halide under controlled low-temperature conditions.
- Spontaneous ring closure and workup to isolate Citalopram base or its pharmaceutically acceptable salts without harsh dehydration agents.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this patented process translates into tangible operational benefits that extend beyond simple yield metrics. The primary advantage lies in the substantial cost savings achieved through process intensification. By consolidating multiple reaction steps into fewer operational units, the overall consumption of solvents, energy, and labor is significantly reduced. The elimination of cuprous cyanide not only removes a costly reagent but also eradicates the expense associated with heavy metal scavenging and hazardous waste disposal, which can constitute a significant portion of the variable manufacturing costs in traditional routes. Furthermore, the use of standard Grignard reagents and common acyl chlorides ensures that the raw material supply base is robust and diversified, reducing the risk of single-source bottlenecks. This resilience is vital for maintaining reducing lead time for high-purity pharmaceutical intermediates in a volatile global market.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the removal of expensive transition metal catalysts and corrosive mineral acids. Traditional methods requiring copper cyanide involve complex filtration and purification steps to meet residual metal limits, adding both time and cost. In this novel route, the absence of such metals simplifies the downstream processing, allowing for faster batch turnover. Additionally, the spontaneous cyclization eliminates the need for dedicated dehydration reactors and the associated heating/cooling cycles, leading to lower utility consumption. The qualitative improvement in process efficiency means that capital expenditure can be optimized, as existing multipurpose reactors can be utilized without the need for specialized glass-lined equipment resistant to hot concentrated sulfuric acid.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the availability of specialized reagents. This synthesis relies on commodity chemicals such as 4-fluorobromobenzene, magnesium, and dimethylaminopropyl chloride, which are produced at scale by numerous global chemical suppliers. This commoditization of the feedstock basket insulates the manufacturing process from supply shocks that might affect niche reagents. Moreover, the robustness of the Grignard reaction, when properly controlled, offers high predictability in terms of batch success rates. For supply chain heads, this predictability allows for more accurate inventory planning and reduces the need for excessive safety stock, thereby freeing up working capital. The ability to source key intermediates like the protected ketone from multiple qualified vendors further strengthens the supply network against disruptions.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden thermal hazards, but this route is inherently safer due to the moderate reaction conditions. The exotherms associated with Grignard additions are well-understood and manageable with standard jacketed reactors and dosing controls. From an environmental perspective, the process aligns with green chemistry principles by reducing the E-factor (mass of waste per mass of product). The avoidance of heavy metals and strong acids minimizes the load on wastewater treatment facilities and reduces the regulatory burden associated with environmental permits. This compliance advantage is increasingly valuable as global regulations tighten, ensuring that the manufacturing site remains operational without the risk of shutdowns due to environmental non-compliance. The cleaner profile also facilitates easier regulatory filings in major markets like the US and EU.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Citalopram synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of adopting this route for their specific production needs. The answers reflect the balance between theoretical chemistry and practical engineering constraints found in the patent examples.
Q: How does this new process improve upon traditional Citalopram synthesis methods?
A: Traditional methods often rely on toxic cuprous cyanide for nitrile introduction or harsh sulfuric acid dehydration for ring closure. This patented process utilizes a single-step Grignard reaction that induces spontaneous cyclization, eliminating heavy metal waste and corrosive acid handling, thereby significantly improving safety and environmental compliance.
Q: What are the critical quality attributes for the Formula IV intermediate?
A: The purity of the protected ketone intermediate (Formula IV) is paramount, as impurities here can carry through to the final API. The process emphasizes high-purity oxidation steps, preferably using tungstate catalysts, to ensure the carbonyl functionality is intact for the subsequent Grignard addition, minimizing side reactions.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is designed for scalability. It avoids difficult-to-control exotherms associated with strong acid dehydration and utilizes standard Grignard chemistry which is well-understood in kilo-lab and plant-scale operations. The spontaneous cyclization simplifies the downstream processing, reducing batch times.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Citalopram Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to a more efficient synthesis route requires a partner with deep technical expertise and proven execution capabilities. Our team has extensively analyzed the pathways described in CN1133633C and possesses the requisite knowledge to troubleshoot and optimize every stage of this Grignard-based process. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the laboratory success of this novel method translates seamlessly to industrial reality. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation to monitor critical parameters such as residual magnesium levels and enantiomeric purity, guaranteeing that every batch meets stringent purity specifications required by top-tier pharmaceutical companies.
We invite you to collaborate with us to leverage this technological advantage for your supply chain. Whether you are looking to qualify a new second source for Citalopram intermediates or seeking to completely revamp your existing manufacturing process, our technical procurement team is ready to assist. We encourage you to request a Customized Cost-Saving Analysis tailored to your current production volumes. By engaging with us, you gain access to specific COA data and route feasibility assessments that will empower you to make informed strategic decisions. Let us help you engineer a more resilient and cost-effective supply chain for this vital antidepressant medication.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
