Advanced Catalytic Cyanation for High-Purity Citalopram Intermediate Manufacturing
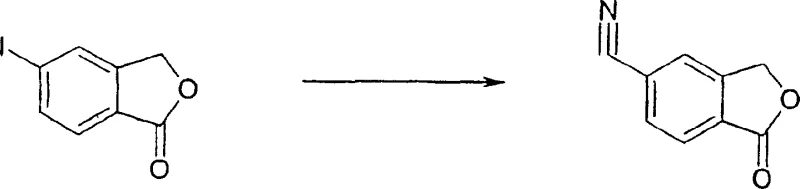
The pharmaceutical industry continuously seeks robust synthetic routes for high-volume antidepressant medications, and the production of citalopram remains a critical focus area for generic and innovator companies alike. Patent CN1211377C discloses a significant technological advancement in the preparation of 5-cyano-benzofuranone, which serves as a pivotal intermediate in the commercial synthesis of this widely prescribed serotonin reuptake inhibitor. This specific patent documentation outlines a novel catalytic methodology that fundamentally alters the traditional approach to introducing the cyano functionality at the 5-position of the isobenzofuranone core. By shifting the cyanation step to an earlier stage in the synthetic sequence, manufacturers can bypass extensive downstream purification protocols that have historically plagued the commercialization of this molecule. The technical implications of this disclosure are profound for R&D teams aiming to optimize impurity profiles and for supply chain managers looking to secure more reliable sources of high-purity pharmaceutical intermediates. The method described leverages transition metal catalysis to facilitate the exchange of halogen or sulfonyl groups with cyanide, offering a versatile platform that accommodates various starting materials including chloro, bromo, and iodo derivatives. This flexibility is crucial for maintaining supply continuity in the face of raw material fluctuations, ensuring that the production of this essential mental health medication remains stable and cost-effective on a global scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the commercial manufacture of citalopram has relied on methods that involve the exchange of a 5-bromo group with a cyano group at a later stage of the synthesis, a process that has proven to be technically cumbersome and economically inefficient on a large scale. The primary drawback of these conventional pathways lies in the relatively low yields obtained during the cyanation step, which directly impacts the overall material throughput and increases the cost of goods sold for the final active pharmaceutical ingredient. Furthermore, the product purity achieved through these older methods is often insufficient, necessitating complex and time-consuming purification procedures to remove unreacted starting materials and side products that are structurally similar to the desired intermediate. The difficulty in isolating the resulting citalopram from the corresponding 5-bromo compound creates a significant bottleneck in the production line, leading to extended lead times and increased operational costs for manufacturing facilities. These purification challenges often require the use of large volumes of solvents and additional reagents, which not only drives up expenses but also generates substantial chemical waste that must be treated and disposed of in compliance with increasingly stringent environmental regulations. Consequently, the reliance on these legacy processes poses a risk to supply chain reliability, as any disruption in the purification capacity can halt the entire production schedule for this critical antidepressant medication.
The Novel Approach
In stark contrast to the limitations of legacy processes, the novel approach detailed in the patent data introduces a catalytic cyanation method that enables the production of the key intermediate in high yield and with exceptional purity profiles right from the early stages of synthesis. This innovative route utilizes a transition metal catalyst to facilitate the direct exchange of a halogen or a specific sulfonyl group at the 5-position of the 3-H-isobenzofuran-1-one core with a cyanide group, effectively streamlining the synthetic pathway. By achieving the correct cyanide substitution early, the method avoids the extensive work-up procedures associated with the old cyanide exchange methods, thereby significantly reducing the processing time and resource consumption required for each batch. The intermediates generated through this new process are inherently easier to purify, allowing manufacturers to obtain the desired 5-cyano-3H-isobenzofuran-1-one in very high yields without the need for aggressive purification techniques that might degrade the product. This methodological shift not only enhances the efficiency of the manufacturing process but also improves the overall quality of the intermediate, which is critical for ensuring the safety and efficacy of the final drug product. The ability to produce such a high-quality intermediate consistently provides a strong foundation for the subsequent Grignard reactions that complete the synthesis of citalopram, ensuring a robust and reliable supply chain for the pharmaceutical industry.

Mechanistic Insights into Pd-Catalyzed Cyanation
The core of this technological breakthrough lies in the sophisticated catalytic cycle that drives the substitution of the halogen atom with the cyano group, a transformation that is mediated by palladium or nickel complexes in the presence of specific ligands. The reaction mechanism typically involves the oxidative addition of the aryl halide substrate to the low-valent metal center, forming an organometallic intermediate that is primed for the subsequent transmetallation step with the cyanide source. The use of ligands such as triphenylphosphine is critical in stabilizing the active catalytic species and facilitating the reductive elimination step that releases the desired nitrile product while regenerating the catalyst for another turnover. The patent data highlights the versatility of this system, noting that catalysts such as Pd(PPh3)4 or in situ generated Ni(0) complexes can be employed effectively, with the nickel system often being prepared by reducing Ni(II) precursors with zinc or magnesium powder in the presence of excess phosphine. This mechanistic understanding allows process chemists to fine-tune the reaction conditions, such as temperature and solvent choice, to maximize the turnover number of the catalyst and minimize the formation of homocoupling byproducts that can compromise the purity of the intermediate. The ability to operate at relatively mild temperatures, particularly when using palladium catalysts, further underscores the efficiency of this method, as it reduces the energy input required for the reaction and minimizes the thermal degradation of sensitive functional groups present in the molecule.
Impurity control is a paramount concern in the synthesis of pharmaceutical intermediates, and this catalytic method offers distinct advantages in managing the impurity profile of the final product. The high selectivity of the palladium or nickel catalyst for the cyanation reaction ensures that side reactions, such as hydrodehalogenation or homocoupling of the aryl halide, are kept to a minimum, resulting in a crude product that is already of high purity. The patent specifies that the intermediates produced are easy to purify, often requiring only a simple recrystallization from acetic acid to achieve analytical grade purity, which is a significant improvement over the complex chromatographic separations often needed for intermediates produced via older methods. Furthermore, the use of substoichiometric amounts of copper or zinc cyanide as a recyclable cyanide source helps to limit the introduction of excess metal impurities into the reaction mixture, simplifying the downstream metal scavenging processes. This level of control over the impurity spectrum is essential for meeting the stringent regulatory requirements for drug substances, as it ensures that the final citalopram product is free from genotoxic impurities and other harmful contaminants that could arise from inefficient synthetic steps. The robust nature of this catalytic system thus provides a reliable means of producing high-quality intermediates that are consistent from batch to batch, a key requirement for commercial pharmaceutical manufacturing.
How to Synthesize 5-Cyano-3H-isobenzofuran-1-one Efficiently
The practical implementation of this synthesis route involves a series of well-defined operational steps that leverage the catalytic efficiency described in the patent to achieve optimal results in a manufacturing setting. The process begins with the preparation of the reaction mixture, where the halogenated isobenzofuranone substrate is combined with a cyanide source such as zinc cyanide or sodium cyanide in a suitable polar aprotic solvent like DMF or NMP. To ensure the activity of the catalyst, the reaction is typically conducted under an inert atmosphere, such as argon or nitrogen, to prevent the oxidation of the low-valent metal species which would otherwise deactivate the catalytic cycle. The detailed standardized synthesis steps see the guide below for specific parameters regarding temperature, catalyst loading, and workup procedures that have been validated to produce high yields.
- Prepare the reaction mixture by combining the halogenated isobenzofuranone substrate with a cyanide source such as zinc cyanide or sodium cyanide in a polar aprotic solvent.
- Add a palladium or nickel catalyst system, optionally generated in situ using metal powders and phosphine ligands under an inert atmosphere.
- Heat the reaction mixture to the specified temperature range, monitor conversion, and isolate the high-purity cyano intermediate through aqueous workup and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel catalytic process translates into tangible strategic advantages that directly impact the bottom line and operational resilience of the pharmaceutical supply network. The elimination of complex and low-yielding purification steps significantly reduces the overall processing time and resource consumption, leading to substantial cost savings in the manufacturing of this critical pharmaceutical intermediate. By avoiding the need for extensive chromatographic separations and reducing the volume of solvents required for workup, facilities can lower their operational expenditures and minimize their environmental footprint, aligning with modern sustainability goals. The high yield and purity of the intermediate also mean that less raw material is wasted, improving the overall material efficiency of the production line and reducing the cost per kilogram of the final active ingredient. These efficiency gains are crucial in a competitive market where cost reduction in API manufacturing is a key driver for maintaining profitability while ensuring patient access to essential medications. The streamlined process also enhances the agility of the supply chain, allowing manufacturers to respond more quickly to changes in demand without the bottleneck of lengthy purification cycles.
- Cost Reduction in Manufacturing: The catalytic nature of this reaction allows for the use of minimal amounts of expensive transition metals, which are often a significant cost driver in fine chemical synthesis. By employing substoichiometric quantities of catalyst and utilizing readily available cyanide sources, the process avoids the high costs associated with stoichiometric reagents and the subsequent removal of heavy metal residues. This reduction in reagent costs, combined with the savings from simplified purification, results in a significantly lower cost of goods sold for the intermediate. The ability to operate at moderate temperatures further reduces energy consumption, contributing to overall cost efficiency. These factors collectively enable a more competitive pricing structure for the final drug product, benefiting both the manufacturer and the healthcare system.
- Enhanced Supply Chain Reliability: The versatility of the method, which accommodates chloro, bromo, and iodo starting materials, provides procurement teams with greater flexibility in sourcing raw materials. This flexibility mitigates the risk of supply disruptions caused by shortages of specific halogenated precursors, ensuring a continuous flow of materials into the production line. The robustness of the catalytic system also means that the process is less sensitive to minor variations in raw material quality, reducing the likelihood of batch failures and production delays. By securing a more reliable source of high-purity intermediates, pharmaceutical companies can better plan their production schedules and maintain adequate inventory levels to meet market demand. This reliability is essential for maintaining the trust of patients and healthcare providers who depend on the consistent availability of antidepressant medications.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard reaction conditions and equipment that are easily transferable from laboratory to commercial scale. The reduced use of solvents and the option for solvent-free or microwave-assisted conditions further simplify the scale-up process, minimizing the engineering challenges often associated with expanding new synthetic routes. From an environmental perspective, the lower waste generation and reduced energy consumption align with green chemistry principles, helping manufacturers meet increasingly strict regulatory requirements for emissions and waste disposal. The use of recyclable cyanide sources and the minimization of heavy metal waste also contribute to a more sustainable manufacturing process. These environmental benefits not only reduce compliance costs but also enhance the corporate social responsibility profile of the manufacturing organization.
Frequently Asked Questions (FAQ)
The following questions and answers are derived directly from the technical specifications and experimental data provided in the patent documentation to address common inquiries regarding the feasibility and benefits of this synthesis method. These insights are intended to clarify the operational parameters and advantages of the catalytic cyanation process for technical and commercial stakeholders evaluating this technology. Understanding these details is crucial for making informed decisions about process adoption and supply chain integration. The answers reflect the specific conditions and outcomes reported in the patent, ensuring accuracy and relevance for professional assessment.
Q: What are the advantages of the new cyanation method over traditional bromo-exchange?
A: The novel catalytic method avoids the low yields and difficult purification associated with traditional 5-bromo group exchange, allowing for high-purity product isolation at an earlier synthesis stage.
Q: Which catalysts are preferred for this specific intermediate synthesis?
A: Preferred catalysts include Palladium(0) complexes like Pd(PPh3)4 or Nickel(0) complexes generated in situ from NiCl2 and zinc powder with triphenylphosphine ligands.
Q: How does this process impact environmental compliance in manufacturing?
A: The process utilizes substoichiometric amounts of heavy metals and allows for solvent-free or low-solvent conditions, significantly reducing waste generation and environmental burden.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Citalopram Intermediate Supplier
The technical potential of this catalytic cyanation route represents a significant opportunity for optimizing the production of citalopram intermediates, and NINGBO INNO PHARMCHEM stands ready to support partners in realizing these benefits through our expert CDMO services. As a specialized contract development and manufacturing organization, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial operations. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest industry standards, guaranteeing that the intermediates we supply meet the exacting requirements of global pharmaceutical regulators. We understand the critical nature of antidepressant supply chains and are dedicated to providing a reliable and consistent source of high-quality materials that support the health and well-being of patients worldwide. Our team of experts is equipped to handle the complexities of transition metal catalysis and purification, delivering solutions that enhance both the efficiency and reliability of your manufacturing processes.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can be integrated into your supply chain to drive efficiency and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your operation and identify opportunities for optimization. We encourage you to contact us to obtain specific COA data and route feasibility assessments that will provide the confidence needed to move forward with this technology. Our goal is to establish a long-term partnership that supports your strategic objectives and ensures the continuous availability of essential medications. Let us help you navigate the complexities of fine chemical manufacturing and secure a competitive advantage in the global pharmaceutical market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
