Advanced Synthesis of 4-Acetoxy-2-Azetidinone: A Scalable Route for Carbapenem Intermediates
Advanced Synthesis of 4-Acetoxy-2-Azetidinone: A Scalable Route for Carbapenem Intermediates
The pharmaceutical industry continuously seeks robust and environmentally sustainable pathways for producing critical antibiotic intermediates, particularly those required for the synthesis of carbapenems. Patent CN101407486A discloses a highly efficient synthesis process for 4-acetoxy-2-azetidinone derivatives, specifically targeting the chiral intermediate (3R,4R)-4-acetoxy-3-[(1'-R-tert-butyldimethylsiloxy)ethyl]-2-azetidinone. This compound serves as a pivotal building block in the manufacture of broad-spectrum beta-lactam antibiotics. The disclosed methodology represents a significant technological leap by utilizing epoxybutyric acid as a primary starting material and employing a unique bis-functional linker strategy. By integrating mild reaction conditions with high-yield transformations, this process addresses the longstanding challenges of scalability and environmental compliance in fine chemical manufacturing. For R&D directors and procurement specialists, understanding the nuances of this route is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials consistently.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-acetoxy-2-azetidinone has been plagued by significant technical and environmental hurdles that hinder cost reduction in API manufacturing. Early methodologies, such as those modifying 6-aminopenicillanic acid (6-APA), were fraught with complexity, requiring tedious protection and deprotection sequences that resulted in low overall yields. Furthermore, these traditional routes often necessitated the use of hazardous heavy metal reagents, such as mercury bisulfate or lead tetraoxide, to facilitate key oxidative transformations. The reliance on such toxic substances not only escalates waste disposal costs but also introduces severe supply chain risks due to increasingly stringent global environmental regulations. Additionally, alternative routes utilizing phosgene for activation pose acute safety hazards, requiring specialized infrastructure that limits the number of qualified manufacturers. These factors collectively contribute to higher production costs and potential supply disruptions for downstream drug manufacturers.
The Novel Approach
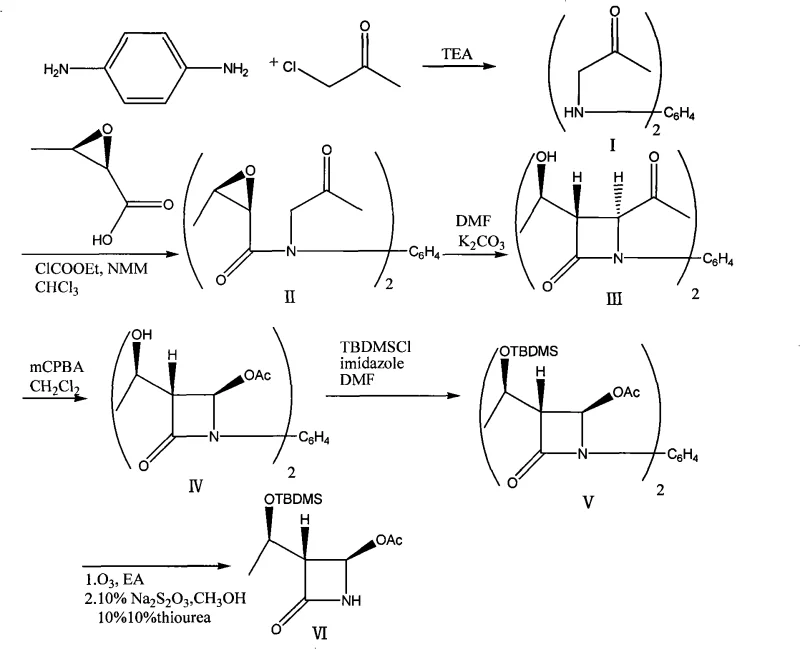
The innovative process outlined in the patent data offers a transformative solution by completely eliminating the need for toxic heavy metals and phosgene gas. Instead, it employs a strategic sequence beginning with the condensation of p-phenylenediamine and monochloroacetone to form a stable bis-ketone intermediate. This scaffold allows for the simultaneous processing of two chiral units, effectively doubling the throughput per batch until the final cleavage step. The subsequent cyclization and oxidation steps utilize standard organic reagents like potassium carbonate and m-chloroperbenzoic acid (mCPBA), which are readily available and easier to handle on a commercial scale. The final step involves ozonolysis to cleave the central aromatic ring, releasing the desired monomeric beta-lactam. This approach not only simplifies the purification process but also aligns with green chemistry principles, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing while ensuring a stable supply of high-purity products.
Mechanistic Insights into the Bis-Functional Cyclization and Oxidative Cleavage
The core of this synthetic strategy lies in the clever use of a 1,4-phenylene bridge to manage stereochemistry and reactivity during the formation of the beta-lactam ring. The mechanism begins with the activation of epoxybutyric acid, which is coupled to the bis-amino scaffold under controlled低温 conditions to preserve the epoxide integrity. Upon heating in the presence of a base like potassium carbonate, an intramolecular nucleophilic attack occurs, closing the four-membered azetidinone ring with high stereoselectivity to yield the (3R,4R) configuration. This cyclization is critical, as it establishes the three chiral centers required for biological activity in the final antibiotic. The subsequent Baeyer-Villiger oxidation converts the C-4 acetyl group into an acetoxy ester, a transformation that traditionally requires careful control to prevent over-oxidation or ring opening. By performing these reactions on the dimeric species, the process minimizes the loss of valuable chiral material and allows for efficient recrystallization of the intermediate before the final cleavage.
Following the installation of the acetoxy group and protection of the side-chain hydroxyl with a tert-butyldimethylsilyl (TBDMS) group, the molecule undergoes ozonolysis. This step is mechanistically distinct as it targets the electron-rich aromatic ring connecting the two beta-lactam units. Ozone reacts with the double bonds of the benzene ring to form ozonides, which are subsequently reduced using dimethyl sulfide or thiourea to yield the aldehyde or ketone fragments, effectively severing the dimer into two monomeric units. This oxidative cleavage is highly advantageous because it avoids the harsh acidic or basic conditions often required for hydrolytic cleavage of similar linkers, thereby preserving the sensitive beta-lactam moiety. The result is a high-purity product with minimal degradation byproducts, a crucial factor for meeting the stringent quality specifications demanded by regulatory agencies for antibiotic intermediates.
How to Synthesize (3R,4R)-4-Acetoxy-2-Azetidinone Efficiently
The execution of this synthesis requires precise control over reaction parameters to maximize yield and stereochemical purity. The process is divided into six distinct operational stages, beginning with the preparation of the bis-ketone linker and concluding with the oxidative release of the final monomer. Each step has been optimized to balance reaction kinetics with safety, ensuring that exothermic events are managed effectively. For example, the initial condensation is performed under reflux in triethylamine, while the epoxide coupling requires strict temperature maintenance between -10°C and -20°C to prevent polymerization. The cyclization step utilizes DMF as a solvent to solubilize the polar intermediates, facilitating the ring closure at elevated temperatures. Operators must adhere to strict protocols during the ozonolysis phase, monitoring ozone feed rates to ensure complete conversion without excessive oxidation of the beta-lactam ring. Detailed standard operating procedures for each stage are essential for reproducibility.
- Condensation of p-phenylenediamine with monochloroacetone in triethylamine to form 1,4-bis[(oxopropyl)amino]benzene.
- Coupling of the bis-amino intermediate with activated epoxybutyric acid using NMM and methyl chloroformate to generate the bis-epoxy precursor.
- Intramolecular cyclization using potassium carbonate in DMF at elevated temperatures to form the bis-beta-lactam core structure.
- Baeyer-Villiger oxidation using m-chloroperbenzoic acid (mCPBA) to convert the acetyl group to an acetoxy group.
- Protection of the hydroxyl group with TBDMSCl and imidazole, followed by oxidative cleavage of the central benzene ring via ozonolysis.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers substantial strategic benefits beyond mere technical feasibility. The elimination of regulated heavy metals like mercury and lead removes a significant bottleneck in the supply chain, as sourcing and disposing of these materials often involve complex permitting and logistics. By shifting to a chemistry based on organic oxidants and ozone, manufacturers can drastically simplify their waste treatment protocols, leading to significant cost savings in environmental compliance. Furthermore, the raw materials utilized in this process, such as epoxybutyric acid and chloroacetone, are commodity chemicals with robust global supply networks. This reduces the risk of raw material shortages that can plague more exotic synthetic routes, thereby enhancing supply chain reliability and ensuring consistent delivery schedules for downstream clients.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the high atom economy of the dimeric intermediate strategy. By processing two chiral units simultaneously until the final step, the facility effectively doubles its output capacity without proportionally increasing reactor volume or labor hours. Additionally, the avoidance of expensive transition metal catalysts and the associated scavenging resins required to remove them from the final product significantly lowers the bill of materials. The high yields reported in the patent examples, often exceeding 90% for key steps, further contribute to a lower cost of goods sold (COGS), allowing for more competitive pricing in the global market for antibiotic intermediates.
- Enhanced Supply Chain Reliability: Supply continuity is paramount for the production of life-saving antibiotics, and this route offers superior stability compared to fermentation-dependent or biocatalytic methods that may suffer from batch-to-batch variability. The purely synthetic nature of the process allows for predictable scaling and scheduling, independent of biological constraints. Moreover, the use of common solvents like ethyl acetate, dichloromethane, and DMF ensures that solvent recovery and recycling systems can be easily integrated, reducing dependency on external solvent suppliers. This self-sufficiency in material flow strengthens the overall resilience of the manufacturing operation against market fluctuations.
- Scalability and Environmental Compliance: From a scale-up perspective, the reaction conditions are remarkably mild, with most steps occurring between -20°C and 100°C, temperatures that are easily achievable in standard glass-lined steel reactors. The absence of high-pressure hydrogenation or cryogenic requirements simplifies the engineering controls needed for commercial production. Environmentally, the process generates less hazardous waste, as the primary byproducts are organic salts and water-soluble fragments that are easier to treat than heavy metal sludge. This alignment with green chemistry principles not only reduces disposal costs but also future-proofs the manufacturing site against tightening environmental legislation, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 4-acetoxy-2-azetidinone intermediates. These answers are derived directly from the technical specifications and comparative data provided in the patent literature. Understanding these details helps stakeholders evaluate the feasibility of integrating this supply source into their existing procurement strategies. The focus is on clarity regarding purity, safety, and the specific advantages over legacy manufacturing technologies.
Q: How does this synthesis route improve upon traditional methods using mercury or lead salts?
A: Traditional routes often rely on toxic heavy metal catalysts like mercury bisulfate or lead oxide, which create significant environmental hazards and require complex waste treatment. This novel process utilizes organic bases and ozone, eliminating heavy metal contamination entirely and simplifying downstream purification.
Q: What are the safety advantages regarding phosgene usage in this protocol?
A: Previous methodologies frequently employed phosgene or its equivalents for activating carboxylic acids, posing severe toxicity risks and requiring specialized containment equipment. The described method activates epoxybutyric acid using safer reagents like methyl chloroformate and N-methylmorpholine, significantly enhancing operational safety for industrial scale-up.
Q: What purity levels can be achieved with this synthetic pathway?
A: The patent data indicates that the final product, (3R,4R)-4-acetoxy-3-[(1'-R-tert-butyldimethylsiloxy)ethyl]-2-azetidinone, can be obtained with HPLC purity exceeding 98%. The use of recrystallization steps and selective ozonolysis contributes to a clean impurity profile suitable for API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Acetoxy-2-Azetidinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global fight against bacterial infections. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of major pharmaceutical companies. We are committed to maintaining stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the stereochemical integrity of every batch. Our facility is equipped to handle the specific reagents and conditions required for this advanced synthesis, including safe ozone generation and containment systems, guaranteeing a secure and consistent supply of this vital carbapenem precursor.
We invite you to collaborate with us to optimize your supply chain for antibiotic production. By leveraging our expertise in this specific synthetic route, we can provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us demonstrate how our commitment to innovation and quality can support your manufacturing goals and ensure the uninterrupted availability of essential medicines.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
