Advanced Fluralaner Synthesis: Scalable Routes for High-Purity Veterinary Intermediates
The pharmaceutical and agrochemical industries are constantly seeking robust synthetic pathways that balance high purity with economic feasibility, particularly for high-value veterinary active ingredients like Fluralaner. Patent CN114394946A introduces a transformative synthesis method that addresses the critical bottlenecks of traditional manufacturing by utilizing 4-bromo-2-methylbenzoic acid as a low-cost initial material. This innovative approach replaces expensive oxime-based precursors with a streamlined sequence involving condensation, substitution, oximation, chlorination, and cyclization reactions. The technical breakthrough lies not only in the selection of raw materials but also in the ingenious use of DMF as a catalytic reagent for acyl chloride formation, which significantly enhances the yield of the initial condensation step. By shifting the synthetic entry point to a widely available commodity chemical, this method offers a compelling solution for cost reduction in veterinary drug manufacturing while maintaining stringent quality standards required for global regulatory compliance. The process is designed with industrial scalability in mind, ensuring that reaction conditions remain mild and easily controllable throughout the multi-step sequence.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Fluralaner has been constrained by the reliance on costly and chemically unstable starting materials, such as 2-methyl-4-formaloximinobenzoic acid or its methyl ester derivatives, as documented in prior art like WO2005085216. These conventional routes often necessitate the oxidation of aldehyde oximes using reagents like NCS to generate chlorooxime intermediates, a step that introduces significant variability and potential safety hazards due to the exothermic nature of oxidation reactions. Furthermore, the condensation of these oxime precursors with amine components frequently suffers from self-condensation side reactions, leading to reduced overall yields and complex impurity profiles that are difficult to purge. The high market price of these specialized oxime starting materials directly inflates the production cost of the final API, creating a barrier to entry for generic manufacturers and limiting the economic viability of large-scale production. Additionally, the multi-step nature of these older pathways often requires harsh reaction conditions that can degrade sensitive functional groups, necessitating extensive purification protocols that further erode profit margins and extend lead times.
The Novel Approach
In stark contrast, the methodology disclosed in CN114394946A leverages 4-bromo-2-methylbenzoic acid, a commodity chemical with a stable supply chain and significantly lower unit cost, to construct the core scaffold of Fluralaner. This novel route strategically delays the formation of the sensitive oxime functionality until a later stage, thereby avoiding the instability issues associated with early-stage oxime handling. The process employs a DMF-catalyzed acyl chloride activation strategy that facilitates a high-yielding condensation with 2-amino-N-(2,2,2-trifluoroethyl)acetamide hydrochloride under mild alkaline conditions. By optimizing the reaction sequence to prioritize robust carbon-carbon and carbon-nitrogen bond formations early on, the method minimizes the generation of difficult-to-remove byproducts. This strategic redesign of the synthetic tree not only simplifies the operational workflow but also enhances the overall atom economy of the process. The result is a manufacturing protocol that is inherently safer, more predictable, and economically superior, making it an ideal candidate for commercial scale-up of complex isoxazoline compounds.
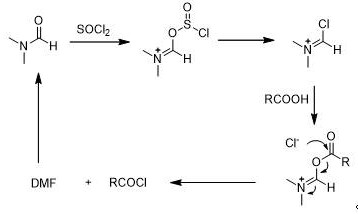
Mechanistic Insights into DMF-Catalyzed Acyl Chloride Formation
The cornerstone of this synthesis efficiency is the utilization of N,N-Dimethylformamide (DMF) as a nucleophilic catalyst during the conversion of the carboxylic acid to the corresponding acid chloride. Mechanistically, DMF reacts with the chlorinating agent, such as thionyl chloride or oxalyl chloride, to form a highly reactive Vilsmeier-Haack type chloroiminium salt intermediate. This activated species acts as a potent chlorinating agent that reacts rapidly with the carboxylic acid substrate at temperatures as low as 10-40°C, significantly lowering the activation energy required for the transformation. The formation of this intermediate prevents the accumulation of unreacted acid and minimizes thermal degradation of the sensitive aromatic ring system. Following the generation of the acid chloride in situ, the immediate addition of the amine nucleophile ensures rapid amidation, effectively trapping the reactive intermediate before it can undergo decomposition or side reactions. This catalytic cycle is crucial for achieving the high molar yields reported in the patent examples, demonstrating how subtle changes in reagent stoichiometry and catalytic loading can dramatically alter process performance.
Beyond the initial condensation, the control of impurities throughout the subsequent substitution and cyclization steps is paramount for meeting pharmaceutical grade specifications. The substitution of the bromo group with a cyano moiety using metal cyanides is conducted in polar aprotic solvents at elevated temperatures, a condition chosen to maximize nucleophilic attack while minimizing hydrolysis of the nitrile group. The subsequent oximation and chlorination steps are carefully buffered to prevent over-chlorination or the formation of dimeric impurities that often plague isoxazoline synthesis. The final 1,3-dipolar cycloaddition, which constructs the critical isoxazoline ring, is performed under basic conditions that promote the generation of the nitrile oxide dipole while suppressing competing polymerization pathways. By rigorously controlling the pH and temperature profiles at each stage, the process ensures that the final crude product possesses a purity profile that allows for straightforward recrystallization. This level of impurity control is essential for reducing lead time for high-purity veterinary intermediates, as it reduces the need for complex chromatographic separations that are impractical at multi-ton scales.
How to Synthesize Fluralaner Efficiently
The synthesis of Fluralaner via this patented route involves a logical progression of five distinct chemical transformations, each optimized for maximum yield and minimal waste generation. The process begins with the activation of the benzoic acid derivative, followed by the sequential installation of the trifluoroethyl amide side chain, the nitrile group, and finally the isoxazoline core. Operators must pay close attention to the stoichiometric ratios of the chlorinating agents and the precise temperature controls during the exothermic acyl chloride formation to ensure reproducibility. The detailed standardized operating procedures for each step, including solvent choices, reaction times, and workup protocols, are critical for translating this laboratory-scale success into a robust manufacturing process. For a comprehensive breakdown of the specific reaction parameters and isolation techniques, please refer to the technical guide below.
- Condensation of 4-bromo-2-methylbenzoic acid with 2-amino-N-(2,2,2-trifluoroethyl)acetamide using DMF-catalyzed acyl chloride formation.
- Substitution of the bromo group with a cyano group using metal cyanides in polar solvents.
- Oximation of the nitrile group followed by chlorination and 1,3-dipolar cycloaddition to form the isoxazoline ring.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the shift to 4-bromo-2-methylbenzoic acid as the starting material represents a significant strategic advantage in terms of raw material security and cost stability. Unlike specialized oxime derivatives that may be sourced from limited suppliers with volatile pricing, benzoic acid derivatives are produced globally in massive quantities for various industrial applications, ensuring a resilient supply chain. This abundance translates directly into cost reduction in agrochemical manufacturing, as the baseline cost of goods sold is fundamentally lowered without compromising the quality of the final active ingredient. Furthermore, the elimination of expensive oxidation reagents like NCS in the early stages reduces the consumption of hazardous chemicals, simplifying waste management and lowering environmental compliance costs. The robustness of the reaction conditions also means that the process is less susceptible to batch-to-batch variability, which is a key factor in maintaining consistent supply to downstream formulation partners.
- Cost Reduction in Manufacturing: The replacement of high-cost oxime precursors with commodity benzoic acids drastically lowers the direct material costs associated with Fluralaner production. Additionally, the high-yielding DMF-catalyzed condensation step minimizes the loss of valuable intermediates, improving the overall mass balance of the process. The use of common solvents like toluene, ethyl acetate, and DMF, which are easily recoverable and recyclable, further contributes to operational expenditure savings. By avoiding complex purification steps early in the synthesis, the process reduces the consumption of silica gel and other chromatography media, which are significant cost drivers in fine chemical manufacturing.
- Enhanced Supply Chain Reliability: Sourcing 4-bromo-2-methylbenzoic acid is far less risky than relying on niche intermediates, as it is a standard building block available from multiple global chemical suppliers. This diversification of the supply base mitigates the risk of production stoppages due to raw material shortages or logistics disruptions. The simplified synthetic route also shortens the overall production cycle time, allowing for faster turnaround on purchase orders and improved responsiveness to market demand fluctuations. Moreover, the stability of the intermediates generated in this pathway allows for potential storage and stockpiling, providing an additional buffer against supply chain volatility.
- Scalability and Environmental Compliance: The mild reaction temperatures and atmospheric pressure conditions described in the patent make this process highly amenable to scaling in standard stainless steel reactors without the need for specialized high-pressure equipment. The reduction in hazardous reagents and the ability to recycle solvents align with green chemistry principles, facilitating easier permitting and regulatory approval in environmentally strict jurisdictions. The high purity of the crude product reduces the volume of mother liquor and waste streams generated during purification, lowering the burden on wastewater treatment facilities. This environmental efficiency is increasingly important for maintaining a social license to operate and meeting the sustainability goals of major pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method, based on the specific data and claims found within the patent documentation. Understanding these nuances is critical for R&D teams evaluating the feasibility of technology transfer and for procurement officers assessing the long-term viability of the supply source. The answers provided reflect the mechanistic realities and process advantages inherent to this specific chemical pathway.
Q: What is the primary cost advantage of this Fluralaner synthesis method?
A: The process utilizes 4-bromo-2-methylbenzoic acid as a starting material, which is significantly cheaper and more commercially available than the expensive oxime precursors required in conventional methods like WO2005085216.
Q: How does the DMF catalysis improve the reaction efficiency?
A: DMF acts as a catalyst during the acyl chloride formation, generating a reactive Vilsmeier-type intermediate that promotes high-yield condensation under mild temperature conditions, reducing energy consumption and side reactions.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights mild reaction conditions, easy control, and simple operation steps, which are critical factors for ensuring safety and consistency during commercial scale-up of complex veterinary intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluralaner Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of this optimized synthetic route to redefine the cost structure of veterinary drug production. As a dedicated CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a practical manufacturing setting. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reaction conditions required for this synthesis, while our rigorous QC labs enforce stringent purity specifications to guarantee product quality. We are committed to leveraging our technical expertise to deliver a reliable supply of high-purity Fluralaner that meets the exacting standards of the global animal health industry.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can optimize your supply chain and reduce your overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic benefits tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will demonstrate our capability to be your trusted partner in veterinary intermediate production.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
