Revolutionizing Risperidone Intermediate Production: A Green, Scalable Synthetic Route for Global Pharma Supply Chains
Revolutionizing Risperidone Intermediate Production: A Green, Scalable Synthetic Route for Global Pharma Supply Chains
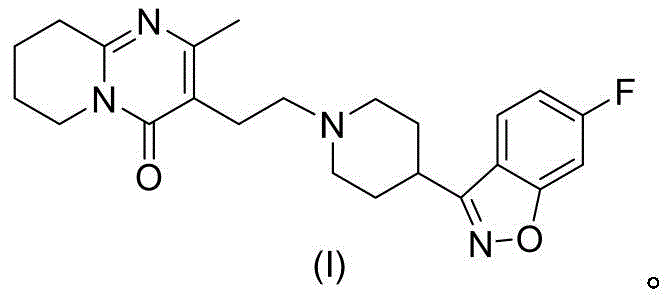
The global demand for high-quality antipsychotic medications continues to drive innovation in the synthesis of key pharmaceutical building blocks. Specifically, the production of risperidone, a widely prescribed atypical antipsychotic, relies heavily on the efficient manufacture of its critical precursor, 3-(2-chloroethyl)-2-methyl-4H-pyrido[1,2-a]pyrimidin-4-one. Recent advancements documented in patent CN112794849B introduce a transformative synthetic methodology that addresses long-standing environmental and efficiency challenges associated with traditional manufacturing protocols. This novel approach replaces hazardous chlorinating agents with a benign catalytic system, offering a robust pathway for the commercial scale-up of complex pharmaceutical intermediates. By leveraging a two-step sequence involving the alkylation of ethyl acetoacetate followed by a specialized cyclocondensation, this technology promises to redefine standards for purity and operational safety in the sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
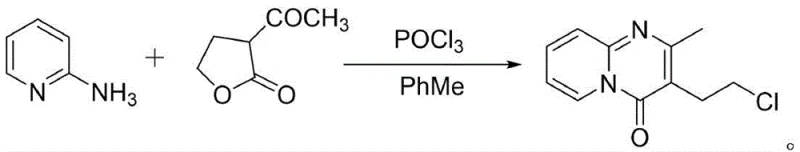
Historically, the industrial synthesis of 3-(2-chloroethyl)-2-methyl-4H-pyrido[1,2-a]pyrimidin-4-one has been plagued by significant environmental and operational hurdles. Traditional routes, such as those described in earlier patents like CN1705661A, typically employ phosphorus oxychloride (POCl3) as a primary chlorinating and cyclizing agent. While chemically effective, the reliance on POCl3 introduces severe safety liabilities, including the generation of corrosive acidic waste streams and toxic phosphorus-containing byproducts that require expensive and energy-intensive neutralization processes. Furthermore, alternative pathways involving multi-step sequences with palladium-catalyzed hydrogenation add unnecessary complexity, increasing the risk of heavy metal contamination and extending production lead times. These legacy methods often struggle to meet the increasingly stringent stringent purity specifications demanded by modern regulatory bodies without incurring prohibitive costs.
The Novel Approach
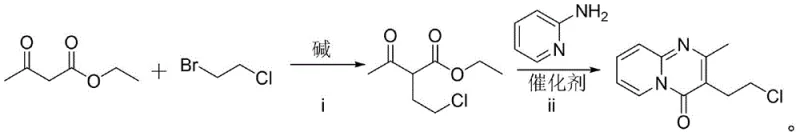
In stark contrast, the methodology outlined in CN112794849B presents a streamlined, two-step strategy that circumvents the need for hazardous reagents entirely. The process initiates with the nucleophilic substitution of ethyl acetoacetate with 1-bromo-2-chloroethane under controlled alkaline conditions to form ethyl 2-acetyl-4-chlorobutyrate. This intermediate is then subjected to a direct cyclization with 2-aminopyridine using imidazole hydrochloride as a mild, organic catalyst. This shift from inorganic halogenating agents to organic salt catalysis not only eliminates the generation of phosphorus waste but also simplifies the downstream purification workflow. The result is a cleaner reaction profile that facilitates easier isolation of the target molecule, thereby supporting cost reduction in API manufacturing through reduced waste treatment expenses and higher overall material throughput.
Mechanistic Insights into Imidazole Hydrochloride-Catalyzed Cyclization
The core innovation of this synthetic route lies in the second step: the condensation of ethyl 2-acetyl-4-chlorobutyrate with 2-aminopyridine. Mechanistically, the use of imidazole hydrochloride serves a dual purpose, acting both as a proton source to activate the carbonyl species and as a buffer to moderate the reaction environment. Unlike strong Lewis acids which can promote uncontrolled side reactions or degradation of the sensitive chloroethyl side chain, this organic salt provides a balanced acidity that favors the formation of the pyrido[1,2-a]pyrimidinone ring system. The reaction proceeds efficiently at temperatures between 100-110°C, where the thermal energy is sufficient to drive the elimination of ethanol and the subsequent aromatization of the heterocyclic core. This precise control over the reaction microenvironment is crucial for minimizing the formation of polymeric impurities or hydrolysis byproducts that often compromise the quality of the final high-purity pharmaceutical intermediate.
Furthermore, the optimization of the initial alkylation step plays a pivotal role in the overall success of the synthesis. The patent data indicates that the activation of the base (sodium hydride) at low temperatures (-20°C) prior to the addition of the alkylating agent is critical for suppressing competitive elimination reactions that could lead to vinyl byproducts. By carefully managing the exotherm during the addition of 1-bromo-2-chloroethane and subsequently raising the temperature to 100°C for the substitution completion, the process achieves exceptional conversion rates. This mechanistic understanding allows for tight control over the impurity profile, ensuring that the intermediate fed into the cyclization step is of sufficient quality to support high yields in the final transformation, ultimately delivering a product suitable for direct use in sensitive medicinal chemistry applications.
How to Synthesize 3-(2-Chloroethyl)-2-methyl-4H-pyrido[1,2-a]pyrimidin-4-one Efficiently
The implementation of this green synthesis protocol requires strict adherence to the optimized reaction parameters identified during the patent's development phase. The process is divided into two distinct stages: the preparation of the chloro-ester intermediate and the subsequent catalytic cyclization. Operators must ensure anhydrous conditions are maintained throughout, particularly during the base-mediated alkylation, to prevent hydrolysis of the ester functionality. The following guide summarizes the critical operational windows derived from the experimental data, providing a roadmap for technical teams aiming to replicate these results in a pilot or production setting. For the complete, standardized operating procedures and safety data sheets, please refer to the detailed technical documentation provided below.
- Perform substitution reaction between ethyl acetoacetate and 1-bromo-2-chloroethane using NaH in DMF at -20°C activation and 100°C reaction temperature to obtain ethyl 2-acetyl-4-chlorobutyrate.
- React the resulting ethyl 2-acetyl-4-chlorobutyrate with 2-aminopyridine using imidazole hydrochloride as a catalyst at 100-110°C for 6 hours.
- Purify the final crude product via recrystallization from ethyl acetate to achieve high-purity 3-(2-chloroethyl)-2-methyl-4H-pyrido[1,2-a]pyrimidin-4-one.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers tangible strategic benefits beyond mere chemical elegance. By eliminating the dependency on phosphorus oxychloride, a reagent subject to strict transportation and storage regulations due to its corrosive nature, facilities can significantly streamline their logistics operations. The removal of this hazardous material reduces the need for specialized containment infrastructure and lowers insurance premiums associated with handling dangerous goods. Moreover, the simplified workup procedure, which avoids complex aqueous washes required to remove phosphorus residues, translates directly into reduced solvent consumption and shorter batch cycle times. These operational efficiencies contribute to a more resilient supply chain capable of responding rapidly to market fluctuations without compromising on quality or compliance standards.
- Cost Reduction in Manufacturing: The economic impact of switching to this catalyst-driven process is substantial. By replacing expensive and waste-generating reagents like POCl3 with inexpensive, commodity-grade chemicals such as imidazole hydrochloride and sodium hydride, the direct material cost per kilogram of product is significantly lowered. Additionally, the high yields reported (up to 97% for the intermediate and 95% for the final product) minimize raw material waste, further enhancing the overall process economics. The reduction in waste disposal costs, stemming from the absence of toxic phosphorus sludge, provides an additional layer of financial optimization that improves the gross margin profile for manufacturers producing this reliable pharmaceutical intermediate supplier grade material.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical concern for long-term API production. The raw materials utilized in this new method, including ethyl acetoacetate and 2-aminopyridine, are widely available bulk chemicals with mature global supply networks. This contrasts sharply with older methods that might rely on specialized or less common reagents. By anchoring the synthesis on readily accessible feedstocks, manufacturers mitigate the risk of supply disruptions caused by geopolitical issues or vendor capacity constraints. This robustness ensures consistent delivery schedules for downstream customers, reinforcing the reputation of the supplier as a dependable partner in the global pharmaceutical value chain.
- Scalability and Environmental Compliance: As regulatory pressure mounts regarding industrial emissions and waste management, the environmental footprint of a chemical process becomes a key differentiator. This synthetic route is inherently greener, generating minimal hazardous waste and avoiding the release of corrosive gases. This alignment with green chemistry principles facilitates easier permitting for plant expansions and reduces the likelihood of regulatory shutdowns due to environmental non-compliance. The scalability of the process is further supported by its thermal robustness; the reactions proceed effectively at moderate temperatures without requiring cryogenic conditions or extreme pressure, making it ideally suited for transfer from laboratory benchtop to multi-ton commercial reactors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental findings and beneficial effects described in the patent literature, aimed at clarifying the practical implications for industrial adoption. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into existing production lines.
Q: Why is the new synthetic route for this risperidone intermediate considered more environmentally friendly?
A: Unlike traditional methods that rely on highly toxic phosphorus oxychloride (POCl3) and generate significant hazardous waste, this novel process utilizes imidazole hydrochloride catalysis under mild conditions, eliminating corrosive chlorinating agents and drastically reducing waste disposal burdens.
Q: What are the critical reaction parameters for maximizing yield in the cyclization step?
A: Optimal results are achieved by maintaining a reaction temperature between 100-110°C for approximately 6 hours using imidazole hydrochloride as the catalyst. Deviating significantly below this temperature range reduces conversion rates, while excessive heat can degrade product quality.
Q: How does this method improve supply chain reliability for API manufacturers?
A: By replacing scarce or heavily regulated reagents like POCl3 with commercially abundant materials like ethyl acetoacetate and simple amine catalysts, the process mitigates raw material procurement risks and simplifies regulatory compliance for large-scale production facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(2-Chloroethyl)-2-methyl-4H-pyrido[1,2-a]pyrimidin-4-one Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is paramount for the future of sustainable pharmaceutical manufacturing. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN112794849B are fully realized in a practical, industrial setting. We are committed to delivering high-purity risperidone intermediate products that meet the most rigorous quality standards, supported by our state-of-the-art rigorous QC labs and comprehensive analytical capabilities. Our facility is equipped to handle the specific requirements of this imidazole-catalyzed process, guaranteeing consistency and reliability for your critical drug development programs.
We invite you to collaborate with us to leverage this advanced technology for your supply chain needs. Whether you require custom synthesis services or bulk supply of this key intermediate, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation can drive value and efficiency in your operations.
