Revolutionizing Tyrosine Derivative Production via Efficient Palladium-Catalyzed Coupling for Commercial Scale-Up
Revolutionizing Tyrosine Derivative Production via Efficient Palladium-Catalyzed Coupling for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking methodologies that can streamline complex synthetic routes while maintaining rigorous purity standards, and the recent disclosure in patent CN115784934A presents a transformative approach to the synthesis of tyrosine derivatives. This specific intellectual property addresses the longstanding inefficiencies associated with traditional multi-step pathways by introducing a robust, one-step palladium-catalyzed coupling strategy that significantly enhances overall process economy. For R&D directors and procurement specialists alike, the ability to condense a four-step sequence into a single operational unit represents a paradigm shift in how high-value amino acid derivatives are manufactured at scale. The technical breakthrough lies in the direct coupling of halogenated acetophenone derivatives with iodopropionate precursors using activated zinc and palladium catalysis, eliminating the need for intermediate isolation and protection-deprotection cycles that typically plague legacy syntheses. By adopting this novel protocol, manufacturers can achieve yields ranging from 67% to 75% with substantially reduced waste generation, positioning this technology as a critical asset for any organization aiming to optimize their supply chain for metabolic and biomedical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex tyrosine derivatives has been hindered by convoluted reaction sequences that introduce significant operational friction and cost volatility into the manufacturing process. As illustrated in the prior art documentation referenced within the patent background, traditional routes often necessitate a minimum of four discrete chemical transformations to arrive at the target molecular architecture, each step requiring its own set of reagents, solvents, and purification protocols. 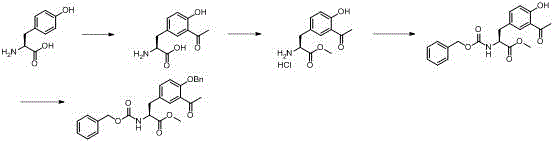 This multiplicity of steps inherently amplifies the risk of yield erosion, as the cumulative loss of material at each stage can render the final process economically unviable for large-scale production. Furthermore, the extensive use of solvents and the generation of intermediate waste streams create substantial environmental compliance burdens and increase the total cost of ownership for the final active ingredient. From a supply chain perspective, relying on such inefficient pathways means longer lead times and greater exposure to raw material price fluctuations, as the demand for various specialized reagents across four different stages creates a fragile logistical network. The complexity also complicates quality control, as impurities generated in early steps can carry through to the final product, necessitating expensive and time-consuming chromatographic purifications that further erode profit margins.
This multiplicity of steps inherently amplifies the risk of yield erosion, as the cumulative loss of material at each stage can render the final process economically unviable for large-scale production. Furthermore, the extensive use of solvents and the generation of intermediate waste streams create substantial environmental compliance burdens and increase the total cost of ownership for the final active ingredient. From a supply chain perspective, relying on such inefficient pathways means longer lead times and greater exposure to raw material price fluctuations, as the demand for various specialized reagents across four different stages creates a fragile logistical network. The complexity also complicates quality control, as impurities generated in early steps can carry through to the final product, necessitating expensive and time-consuming chromatographic purifications that further erode profit margins.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the innovative technique disclosed in the patent data leverages a direct cross-coupling reaction that effectively collapses the entire synthetic timeline into a single, highly efficient operation. 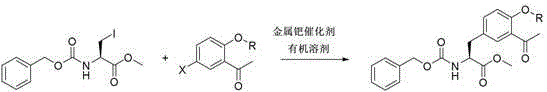 This streamlined approach utilizes a metal palladium catalyst, such as Pd(PPh3)4 or Pd2(dba)3, in conjunction with zinc powder activation to facilitate the bond formation between the aromatic ketone moiety and the amino acid backbone in one pot. The versatility of this method is evidenced by its compatibility with a wide range of organic solvents, including DMF, THF, ethyl acetate, and toluene, allowing process chemists to select the most cost-effective and environmentally benign medium for their specific facility capabilities. By operating at moderate temperatures between 40°C and 80°C, the reaction avoids the energy intensity associated with high-heat processes while still driving the conversion to completion within a reasonable timeframe of approximately 12 hours. This consolidation of steps not only simplifies the operational workflow but also dramatically improves the atom economy of the process, ensuring that a higher proportion of the starting materials end up in the desired product rather than in waste byproducts.
This streamlined approach utilizes a metal palladium catalyst, such as Pd(PPh3)4 or Pd2(dba)3, in conjunction with zinc powder activation to facilitate the bond formation between the aromatic ketone moiety and the amino acid backbone in one pot. The versatility of this method is evidenced by its compatibility with a wide range of organic solvents, including DMF, THF, ethyl acetate, and toluene, allowing process chemists to select the most cost-effective and environmentally benign medium for their specific facility capabilities. By operating at moderate temperatures between 40°C and 80°C, the reaction avoids the energy intensity associated with high-heat processes while still driving the conversion to completion within a reasonable timeframe of approximately 12 hours. This consolidation of steps not only simplifies the operational workflow but also dramatically improves the atom economy of the process, ensuring that a higher proportion of the starting materials end up in the desired product rather than in waste byproducts.
Mechanistic Insights into Pd-Catalyzed Cross-Coupling
The core of this technological advancement rests on the sophisticated interplay between the palladium catalyst and the organozinc species generated in situ, which facilitates a highly selective carbon-carbon bond formation essential for constructing the tyrosine scaffold. The mechanism likely proceeds through a catalytic cycle where the palladium(0) species undergoes oxidative addition with the halogenated acetophenone substrate, forming a reactive organopalladium intermediate that is primed for transmetallation. Simultaneously, the zinc powder, activated by trace amounts of iodine, reacts with the iodopropionate derivative to generate an organozinc reagent that serves as the nucleophilic partner in the coupling event. This transmetallation step transfers the organic group from the zinc to the palladium center, setting the stage for the final reductive elimination that releases the coupled tyrosine derivative and regenerates the active palladium(0) catalyst. Understanding this cycle is crucial for R&D teams, as it highlights the importance of maintaining strict anhydrous conditions and inert atmospheres to prevent the decomposition of the sensitive organometallic intermediates, thereby ensuring consistent batch-to-batch reproducibility. The choice of ligand and catalyst loading can be fine-tuned to accommodate different steric and electronic properties of the substrates, offering a flexible platform for synthesizing a diverse library of tyrosine analogues tailored for specific biological applications.
Beyond the primary reaction pathway, the control of impurity profiles is a critical consideration for any process intended for pharmaceutical grade production, and this one-step method offers inherent advantages in this regard. By eliminating three intermediate isolation and purification steps, the opportunity for the introduction of extraneous contaminants or the formation of side-products derived from harsh reaction conditions is significantly minimized. The use of mild reaction temperatures and standard organic solvents further reduces the likelihood of thermal degradation or unwanted rearrangement reactions that often complicate the purification of sensitive amino acid derivatives. Moreover, the specificity of the palladium-catalyzed coupling ensures that the reaction proceeds with high regioselectivity, targeting the desired position on the aromatic ring without affecting other functional groups present on the molecule. This high level of chemical fidelity translates directly into a cleaner crude reaction mixture, which simplifies the downstream workup and reduces the load on final purification columns, ultimately resulting in a product with superior purity specifications that meet the stringent requirements of global regulatory bodies.
How to Synthesize Tyrosine Derivatives Efficiently
Implementing this synthesis route requires careful attention to the activation of the zinc metal and the precise stoichiometry of the catalyst system to ensure optimal conversion rates. The patent details a robust protocol where zinc powder is first treated with iodine in the chosen solvent to activate the metal surface, followed by the sequential addition of the halogenated ketone and the iodopropionate precursor under a nitrogen atmosphere.
- Activate zinc powder with iodine in an organic solvent such as DMF or THF under inert atmosphere.
- Add the halogenated acetophenone derivative and the iodopropionate precursor to the reaction mixture.
- Introduce the palladium catalyst and heat the mixture to 40-80°C to facilitate the cross-coupling reaction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from a multi-step legacy process to this consolidated one-step methodology offers profound strategic benefits that extend far beyond simple chemical efficiency. The reduction in unit operations directly correlates to a significant decrease in capital expenditure and operational overhead, as fewer reactors, filtration units, and drying ovens are required to produce the same volume of finished goods. This simplification of the manufacturing footprint allows facilities to increase their throughput capacity without the need for major infrastructure investments, effectively unlocking latent production potential within existing plants. Furthermore, the reliance on commodity chemicals such as zinc powder, iodine, and common solvents like ethyl acetate or toluene mitigates the risk of supply disruptions associated with specialized or exotic reagents, ensuring a more resilient and predictable supply chain. The ability to source raw materials from multiple vendors enhances negotiating power and stabilizes input costs, providing a buffer against market volatility that is increasingly common in the global chemical sector.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation and purification steps results in substantial savings on solvent consumption, labor hours, and waste disposal fees, which are often the hidden drivers of high manufacturing costs in fine chemical production. By processing the reaction in a single vessel, the energy demand for heating, cooling, and agitation is drastically reduced, leading to a lower carbon footprint and reduced utility bills for the manufacturing site. The improved overall yield of the process means that less raw material is required to produce a kilogram of the final product, directly improving the gross margin and making the final API or intermediate more competitive in the marketplace. Additionally, the simplified workflow reduces the need for highly specialized operator training, allowing for more flexible staffing arrangements and lower personnel costs associated with complex multi-stage campaigns.
- Enhanced Supply Chain Reliability: Shortening the synthesis from four steps to one inherently reduces the lead time required to fulfill customer orders, enabling a more responsive and agile supply chain that can adapt quickly to fluctuating market demands. With fewer processing stages, the risk of batch failure due to equipment malfunction or human error at intermediate stages is minimized, ensuring a higher rate of successful campaign completions and more reliable delivery schedules. The use of stable and widely available starting materials ensures that production can be sustained even during periods of global supply chain stress, providing a secure source of supply for downstream pharmaceutical manufacturers. This reliability is paramount for long-term supply agreements, where consistency of supply is often valued as highly as the price of the material itself.
- Scalability and Environmental Compliance: The reaction conditions described in the patent are inherently scalable, utilizing standard mixing and heating technologies that are readily available in pilot and commercial-scale reactors, facilitating a smooth transition from laboratory development to full-scale production. The reduction in solvent usage and waste generation aligns perfectly with modern green chemistry principles and increasingly stringent environmental regulations, reducing the permitting burden and potential liability associated with hazardous waste management. The process generates fewer byproducts and effluents, simplifying the wastewater treatment requirements and lowering the environmental impact of the manufacturing operation. This sustainability profile not only meets regulatory compliance but also enhances the brand reputation of the manufacturer as a responsible and forward-thinking partner in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology, drawing directly from the experimental data and beneficial effects outlined in the patent documentation.
Q: How does this new synthesis method improve upon traditional routes?
A: The patented method reduces the synthesis from four distinct steps to a single direct coupling reaction, drastically minimizing solvent consumption, purification burdens, and overall processing time while maintaining high yields between 67% and 75%.
Q: What catalysts are suitable for this tyrosine derivative production?
A: The process effectively utilizes standard palladium catalysts such as tetrakis(triphenylphosphine)palladium(0) or tris(dibenzylideneacetone)dipalladium(0), which are commercially available and compatible with various organic solvents including toluene and ethyl acetate.
Q: Is this process scalable for industrial manufacturing?
A: Yes, the reaction conditions operate at moderate temperatures (40-80°C) and use common industrial solvents, making it highly amenable to scale-up from laboratory benchtop to multi-ton commercial production without requiring exotic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tyrosine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this streamlined synthesis route and possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production necessary to bring this technology to the global market. Our state-of-the-art facilities are equipped to handle the specific requirements of palladium-catalyzed reactions, including rigorous QC labs capable of monitoring trace metal residues to ensure our products meet stringent purity specifications demanded by top-tier pharmaceutical clients. We understand that the successful commercialization of a new process requires more than just a lab recipe; it demands a partner who can navigate the complexities of process safety, regulatory compliance, and supply chain logistics with equal expertise. Our team of process chemists and engineers is dedicated to optimizing this route further, ensuring that every batch delivered represents the pinnacle of quality and consistency.
We invite you to engage with our technical procurement team to discuss how this advanced manufacturing method can specifically benefit your project timelines and cost structures. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages of switching to this one-step protocol for your specific volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments that demonstrate our capability to deliver high-purity tyrosine derivatives reliably and efficiently. Let us collaborate to accelerate your drug development programs with a supply chain solution that is as innovative as the molecules you are creating.
