Advanced Manufacturing of Ertapenem Intermediates via Mild Hydrolysis
Introduction to Patent CN101376644A and Strategic Value
The pharmaceutical landscape for carbapenem antibiotics demands increasingly efficient and cost-effective synthetic routes to maintain supply chain stability for critical medications like Ertapenem. Patent CN101376644A introduces a groundbreaking methodology for synthesizing the key intermediate, designated as Formula VII, which serves as the essential side chain precursor for Ertapenem. This innovation addresses the longstanding industrial bottlenecks associated with traditional synthesis, specifically the reliance on harsh cryogenic conditions and expensive reagents. By shifting the paradigm to a mild hydrolysis protocol operable at ambient temperatures, this technology offers a compelling value proposition for manufacturers seeking to optimize their production lines. The core of this advancement lies in the conversion of the thioacetate precursor (Formula VI) directly to the free thiol (Formula VII) under controlled acidic or alkaline conditions, eliminating the need for complex purification steps.
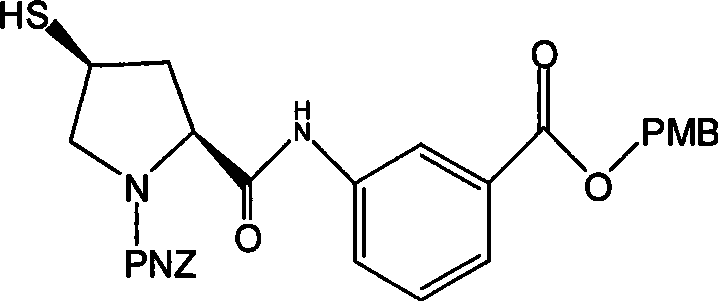
For R&D directors and process chemists, the structural integrity of Formula VII is paramount, as it contains the chiral pyrrolidine scaffold necessary for the biological activity of the final antibiotic. The patent details a robust pathway that preserves stereochemistry while introducing the critical sulfhydryl group with high fidelity. This intermediate is not merely a chemical entity but a strategic asset that enables the downstream assembly of the carbapenem core. The ability to produce this molecule with high yield and purity using simplified protocols represents a significant leap forward in process chemistry, aligning perfectly with the industry's drive towards greener and more economical manufacturing practices.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing Ertapenem intermediates, such as those documented in earlier patents like US5478820, have been plagued by significant operational inefficiencies that hinder large-scale adoption. These legacy methods typically necessitate rigorous low-temperature environments, often requiring reaction vessels to be maintained at minus 20 degrees Celsius or lower. Such cryogenic conditions impose a heavy burden on infrastructure, demanding specialized cooling equipment and resulting in substantial energy expenditures that erode profit margins. Furthermore, these traditional routes frequently rely on costly and hazardous reagents, such as diphenyl phosphoryl chloride and diethyl propyl ethylamine, which complicate waste management and increase the overall cost of goods sold. The multi-step nature of these older processes often mandates column chromatography for purification, a technique that is notoriously difficult to scale and results in significant material loss, thereby reducing overall throughput and yield.
The Novel Approach
In stark contrast, the methodology disclosed in CN101376644A revolutionizes the synthesis by enabling the critical hydrolysis step to proceed under mild, near-ambient conditions ranging from 0 to 45 degrees Celsius. This dramatic relaxation of thermal requirements eliminates the need for energy-intensive cryogenic cooling, allowing the reaction to be performed in standard glass-lined or stainless steel reactors without specialized modifications. The new approach utilizes readily available and inexpensive mineral acids, such as sulfuric acid, or common bases like sodium hydroxide, replacing the exotic and expensive phosphorylating agents of the past. By designing a route that avoids column chromatography and relies on simple crystallization or extraction for isolation, the process achieves a level of operational simplicity that is highly attractive for commercial manufacturing. This shift not only reduces the direct cost of materials but also shortens the production cycle time, enhancing the overall agility of the supply chain.
Mechanistic Insights into Mild Hydrolysis and Thioester Cleavage
The chemical elegance of this process is rooted in the selective cleavage of the thioacetate group to reveal the reactive thiol functionality without compromising the sensitive beta-lactam precursors or the chiral centers of the pyrrolidine ring. The mechanism involves the nucleophilic attack of water or hydroxide ions on the carbonyl carbon of the thioester moiety in Formula VI, leading to the formation of a tetrahedral intermediate that subsequently collapses to release acetate and the free thiol. Under acidic conditions, the protonation of the carbonyl oxygen enhances the electrophilicity of the carbonyl carbon, facilitating the attack by water molecules even at moderate temperatures. This careful balancing of pH and temperature ensures that the hydrolysis proceeds rapidly enough to be practical (often within 10 to 30 minutes) while remaining gentle enough to prevent racemization or degradation of the adjacent amide bonds. The use of solvents like dichloromethane or tetrahydrofuran provides an optimal medium for solubilizing the organic substrate while allowing for effective phase separation during workup.
From an impurity control perspective, the mildness of this hydrolysis protocol is instrumental in maintaining a clean product profile. Harsh conditions often lead to side reactions such as the hydrolysis of the ester protecting groups or the epimerization of the chiral centers, which would generate difficult-to-remove impurities that could compromise the safety and efficacy of the final API. By operating at a controlled pH (preferably pH less than or equal to 1 for acid hydrolysis) and moderate temperatures, the process minimizes these degradation pathways. The patent data suggests that the resulting product can be isolated with high purity, often exceeding 95 percent, simply through crystallization or extraction. This inherent selectivity reduces the burden on downstream purification processes, ensuring that the intermediate meets the stringent quality specifications required for pharmaceutical grade materials without the need for extensive reprocessing.
How to Synthesize Ertapenem Intermediate Efficiently
The synthesis of this high-value intermediate is designed to be straightforward and robust, making it accessible for facilities equipped with standard chemical processing capabilities. The process begins with the preparation of the thioacetate precursor, followed by the pivotal hydrolysis step that defines this patent's innovation. Operators can choose between acidic or alkaline hydrolysis depending on their specific equipment and waste treatment capabilities, with both options yielding the desired thiol product effectively. The reaction times are short, typically ranging from 10 to 30 minutes, which allows for high throughput in batch operations. For a detailed breakdown of the specific molar ratios, solvent volumes, and workup procedures, please refer to the standardized synthesis guide provided below.
- Prepare the thioacetate precursor (Formula VI) via nucleophilic substitution using potassium thioacetate in polar aprotic solvents.
- Conduct acidic hydrolysis using dilute sulfuric acid in dichloromethane or tetrahydrofuran at temperatures between 0°C and 45°C.
- Isolate the final thiol product (Formula VII) through crystallization or extraction, achieving high purity without column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route translates directly into tangible operational efficiencies and risk mitigation. The elimination of cryogenic requirements removes a major bottleneck in production scheduling, as facilities no longer need to compete for limited low-temperature reactor capacity or worry about cooling system failures halting production. The substitution of expensive, specialized reagents with commodity chemicals like sulfuric acid and sodium hydroxide drastically reduces the raw material bill of materials, insulating the manufacturing process from volatility in the pricing of fine chemical reagents. Furthermore, the simplified workup procedure, which avoids time-consuming column chromatography, significantly increases the throughput of the manufacturing suite, allowing for more batches to be produced in the same timeframe. These factors combine to create a more resilient and cost-effective supply chain for this critical antibiotic intermediate.
- Cost Reduction in Manufacturing: The transition to room temperature operations eliminates the substantial energy costs associated with maintaining sub-zero reaction environments, leading to significant utility savings. Additionally, the replacement of high-cost reagents such as diphenyl phosphoryl chloride with inexpensive mineral acids results in a drastic reduction in direct material costs. The avoidance of chromatographic purification further lowers expenses by reducing solvent consumption and silica gel waste, while simultaneously increasing the recovery yield of the product. These cumulative savings contribute to a much lower cost of goods sold, enhancing the competitiveness of the final API in the global market.
- Enhanced Supply Chain Reliability: By relying on widely available commodity chemicals and standard solvents like dichloromethane and THF, the process mitigates the risk of supply disruptions caused by shortages of specialized reagents. The robustness of the reaction conditions means that the process is less sensitive to minor variations in operating parameters, leading to more consistent batch-to-batch quality and fewer rejected lots. This reliability ensures a steady flow of intermediate material to downstream API manufacturers, preventing stockouts and ensuring continuity of supply for life-saving medications. The simplified logistics of handling non-hazardous acids and bases also streamline the procurement and storage processes.
- Scalability and Environmental Compliance: The design of this process is inherently scalable, moving seamlessly from laboratory benchtop to multi-ton commercial production without the need for complex engineering modifications. The reduction in solvent usage and the elimination of silica waste from chromatography significantly lower the environmental footprint of the manufacturing process, aiding in compliance with increasingly strict environmental regulations. The use of common solvents facilitates easier recycling and recovery, further supporting sustainability goals. This alignment with green chemistry principles not only reduces waste disposal costs but also enhances the corporate social responsibility profile of the manufacturing organization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable basis for decision-making. Understanding these nuances is crucial for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the long-term viability of the supply chain. We encourage stakeholders to review these points carefully to fully appreciate the strategic advantages offered by this methodology.
Q: What are the primary advantages of this hydrolysis method over traditional low-temperature routes?
A: Traditional methods often require cryogenic conditions (-20°C) and expensive phosphorylating agents. This novel route operates at room temperature (0-45°C) using common mineral acids, significantly reducing energy consumption and reagent costs while simplifying equipment requirements.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is specifically designed for industrial scalability. It avoids column chromatography, utilizes inexpensive solvents like dichloromethane and THF, and employs robust reaction conditions that are easily controlled in standard reactor vessels.
Q: What is the expected purity profile of the intermediate produced?
A: The patent data indicates that the final product can be isolated as an off-white solid with high purity. The mild conditions minimize side reactions such as epimerization or degradation, ensuring a clean impurity profile suitable for subsequent API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ertapenem Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the modern pharmaceutical landscape. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative technologies like the one described in CN101376644A can be successfully translated into industrial reality. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reaction conditions required for this synthesis, while our rigorous QC labs enforce stringent purity specifications to guarantee the quality of every batch. We are committed to bridging the gap between patent potential and commercial reality, offering our clients a seamless path from process development to full-scale manufacturing.
We invite you to engage with our technical procurement team to discuss how this advanced intermediate can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic benefits applicable to your operation. We are prepared to provide specific COA data and route feasibility assessments to support your internal evaluation processes. Let us collaborate to enhance the efficiency and reliability of your carbapenem production capabilities.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
