Advanced Semi-Synthetic Route for Vinblastine Sulfate: Scalability and Purity for Global Oncology Markets
Advanced Semi-Synthetic Route for Vinblastine Sulfate: Scalability and Purity for Global Oncology Markets
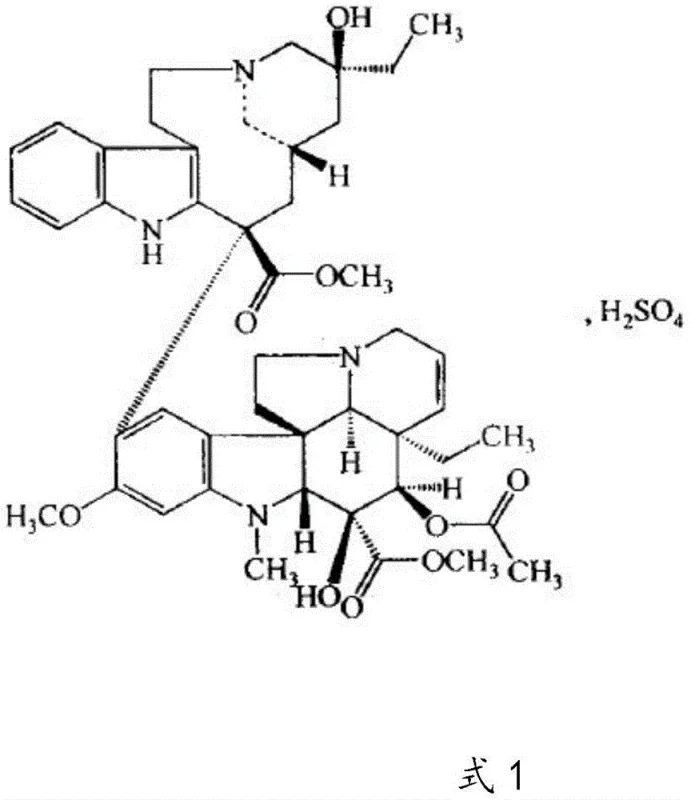
The global demand for effective antineoplastic agents continues to drive innovation in the synthesis of complex bis-indole alkaloids, specifically Vinblastine Sulfate. As a critical microtubule inhibitor used in treating malignant lymphoma, breast cancer, and lung cancer, the supply security and quality consistency of this API are paramount for pharmaceutical manufacturers worldwide. A significant technological breakthrough in this domain is detailed in patent CN112480148B, which discloses a robust semi-synthetic method that fundamentally alters the production landscape. Unlike traditional extraction methods that suffer from extremely low yields and heavy environmental burdens, this novel approach leverages the oxidative coupling of Vindoline and Catharanthine. By utilizing ferric chloride as a key catalyst and optimizing the reduction environment with ferric oxalate, the process achieves a molar yield exceeding 35% with a final purity greater than 98%. This represents a substantial leap forward in process chemistry, offering a viable pathway for reliable vinblastine sulfate supplier networks to secure long-term inventory without relying on fluctuating agricultural outputs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of Vinblastine Sulfate has been bottlenecked by two primary methodologies, both of which present severe logistical and economic challenges for modern supply chains. The dominant method, plant extraction from Catharanthus roseus, is notoriously inefficient; extracting mere grams of high-purity product from tons of plant biomass results in exorbitant raw material costs and generates massive volumes of organic solvent waste that requires expensive treatment. Furthermore, the natural content of Vinblastine in the plant is inherently low, making it impossible to scale production rapidly to meet sudden surges in market demand for cancer therapies. Alternatively, earlier chemical synthesis attempts, such as those referenced in prior art like CN201110378355.0, often relied on dehydrated vinblastine as a starting material. This precursor is itself scarce and expensive, creating a dependency on a limited supply chain. Moreover, these older synthetic routes frequently yielded products with content levels around 60%, necessitating extensive and costly downstream purification to meet pharmacopeial standards, thereby eroding profit margins and extending lead times for high-purity pharmaceutical intermediates.
The Novel Approach
The methodology outlined in patent CN112480148B disrupts these legacy constraints by shifting the synthetic entry point to Vindoline and Catharanthine, which are structurally similar but significantly more abundant and cost-effective precursors found in the same plant source. This strategic pivot allows manufacturers to bypass the scarcity issues associated with direct Vinblastine extraction or the purchase of expensive dehydrated intermediates. The core innovation lies in the seamless integration of the coupling and reduction steps. Instead of isolating the unstable dehydrated vinblastine intermediate, the process utilizes the crude reaction solution directly in the subsequent reduction phase. This telescoping of steps not only simplifies the operational workflow but also minimizes material loss typically associated with isolation and drying procedures. By introducing air and utilizing a ferric oxalate system during the sodium borohydride reduction, the reaction achieves high selectivity, drastically reducing the formation of unwanted byproducts and setting the stage for a much cleaner final product that requires less aggressive purification.
Mechanistic Insights into FeCl3-Catalyzed Oxidative Coupling
From a mechanistic perspective, the success of this synthesis hinges on the precise control of the oxidative coupling between the indole nuclei of Vindoline and Catharanthine. The use of ferric chloride ($FeCl_3$) in an acidic medium (pH 1.0-1.5) facilitates the generation of radical cation species necessary for the C-C bond formation that links the two monomeric units. This step is critical because uncontrolled oxidation can lead to polymerization or degradation of the sensitive indole rings. The patent specifies maintaining the temperature between 20-30°C, a range that balances reaction kinetics with the thermal stability of the intermediates. Following the coupling, the transition to the reduction phase is managed with exceptional care. The addition of the dehydrated vinblastine solution into a ferric oxalate mixture, followed by aeration and the dropwise addition of sodium borohydride, creates a unique redox environment. The ferric oxalate likely acts as a buffer or co-catalyst that moderates the reducing power of the borohydride, preventing over-reduction which could destroy the delicate ester functionalities present in the molecule.
Furthermore, the control of impurities is rigorously addressed through pH regulation during the reduction step. The protocol mandates maintaining the system pH between 3.0 and 5.0 using hydrochloric acid during the addition of the reducing agent. This specific acidity window is crucial; if the pH rises too high due to the basicity of sodium borohydride, side reactions accelerate, leading to complex impurity profiles that are difficult to separate later. By actively titrating with acid, the process ensures that the reduction of the iminium ion intermediate proceeds cleanly to the desired amine without generating alkaline degradation products. This mechanistic precision translates directly to commercial value, as it reduces the burden on the purification columns and ensures that the final crystallization yields a product that consistently meets the stringent requirements for high-purity anticancer intermediates, thereby minimizing batch rejection rates.
How to Synthesize Vinblastine Sulfate Efficiently
The practical execution of this synthesis requires strict adherence to the optimized parameters regarding solvent ratios, temperature gradients, and stoichiometric balances to ensure reproducibility at scale. The process begins with the dissolution of Vindoline in a solvent system such as trifluoroethanol or tetrahydrofuran, followed by the controlled addition of Catharanthine and the ferric chloride catalyst under nitrogen protection to prevent premature oxidation. Once the dehydrated intermediate is formed in situ, it is immediately transferred to the reduction vessel containing chilled ferric oxalate solution. The subsequent workup involves a dual-stage chromatographic purification, first using alkaline alumina to remove bulk impurities and then silica gel for fine polishing, before the final salt formation with sulfuric acid. For a detailed breakdown of the specific reagent quantities, flow rates, and safety protocols required to implement this GMP-compliant workflow, please refer to the standardized operating procedure below.
- Coupling Reaction: React Vindoline and Catharanthine in organic solvent with inorganic acid and ferric chloride at 20-30°C to form dehydrated vinblastine solution.
- Reduction Step: Add the reaction solution to ferric oxalate/water mix, introduce air, and reduce with sodium borohydride while maintaining pH 3.0-5.0.
- Purification and Salting: Purify crude product via dual-column chromatography (alumina then silica) and form sulfate salt with sulfuric acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this semi-synthetic route offers transformative benefits that extend far beyond simple yield improvements. The most significant advantage is the decoupling of production capacity from the volatile yields of direct plant extraction. By utilizing Vindoline and Catharanthine as feedstocks, which are present in the plant in much higher concentrations than Vinblastine, manufacturers can secure raw material contracts at a fraction of the cost of pure Vinblastine. This fundamental shift in the bill of materials leads to substantial cost savings in API manufacturing, allowing pharmaceutical companies to maintain healthier margins even in the face of fluctuating raw commodity prices. Additionally, the elimination of the intermediate refining step means fewer unit operations, less solvent consumption, and reduced energy usage, all of which contribute to a leaner and more cost-effective production model that aligns with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the removal of expensive isolation steps and the use of commodity-grade reagents like ferric chloride and sodium borohydride. By avoiding the need to purchase or synthesize dehydrated vinblastine as a standalone starting material, the overall cost of goods sold (COGS) is significantly lowered. The telescoped nature of the reaction reduces labor hours and equipment occupancy time, further driving down overhead costs. This efficiency makes the production of Vinblastine Sulfate financially viable even in regions with higher operational expenses, providing flexibility in site selection for contract manufacturing organizations.
- Enhanced Supply Chain Reliability: Supply continuity is a critical risk factor for oncology drugs, and this method mitigates that risk by diversifying the raw material base. Since Vindoline and Catharanthine are more abundant and easier to extract in bulk than Vinblastine, the supply chain is less susceptible to agricultural shortages or seasonal variations. The robustness of the chemical synthesis also means that production can be ramped up quickly in response to market demand without the long lead times associated with cultivating and harvesting plant biomass. This reliability ensures that downstream drug product manufacturers can maintain consistent inventory levels, preventing stockouts that could impact patient treatment regimens.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing mild reaction temperatures (0-30°C) and standard pressure conditions that are easily replicated in large-scale stainless steel reactors. The simplified workflow generates less hazardous waste compared to traditional extraction methods, which often require vast quantities of chlorinated solvents for biomass processing. By reducing the environmental footprint and simplifying waste treatment protocols, this method facilitates easier regulatory approval and compliance with increasingly strict environmental regulations. This makes it an ideal candidate for commercial scale-up of complex alkaloids in jurisdictions with rigorous environmental oversight.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of technology transfer or for procurement teams assessing the quality assurances provided by potential suppliers.
Q: What are the primary advantages of this semi-synthetic route over plant extraction?
A: This method bypasses the low-yield and environmentally taxing plant extraction process. It utilizes readily available precursors (Vindoline and Catharanthine) which are more abundant in nature than Vinblastine itself, ensuring better supply chain stability and significantly reduced production costs.
Q: How does the process ensure high purity (>98%) without complex refining?
A: The process employs a strategic two-step column chromatography purification (alkaline alumina followed by silica gel) and precise pH control during the reduction phase. This minimizes byproduct formation and eliminates the need for intermediate refining, directly yielding high-purity final product.
Q: Is this synthesis method suitable for large-scale commercial manufacturing?
A: Yes, the reaction conditions are mild (0-30°C) and utilize common industrial reagents like ferric chloride and sodium borohydride. The elimination of intermediate isolation steps simplifies the workflow, making it highly adaptable for metric-ton scale production in standard pharmaceutical facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vinblastine Sulfate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics of patent CN112480148B can be fully realized in an industrial setting. We are committed to delivering Vinblastine Sulfate that meets stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis development or reliable supply of established intermediates, our infrastructure is designed to support the complex demands of the global oncology market with unwavering consistency and quality.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clearer picture of the potential economic benefits specific to your volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to verify how our manufacturing standards align with your project goals. Let us collaborate to secure a sustainable and cost-efficient supply of this critical life-saving medication.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
