Scalable Synthesis of Vinblastine Sulfate via Iron-Catalyzed Coupling for Commercial API Production
Scalable Synthesis of Vinblastine Sulfate via Iron-Catalyzed Coupling for Commercial API Production
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective routes for critical oncology agents, and the synthesis of Vinblastine Sulfate represents a prime example of such technological evolution. As detailed in patent CN112480148A, a novel preparation method has been developed that fundamentally shifts the production paradigm from resource-intensive plant extraction to a streamlined semi-synthetic approach. This method leverages the oxidative coupling of vindoline and catharanthine, utilizing an inorganic acid and ferric chloride system to generate the key dehydrated vinblastine intermediate in situ. By bypassing the need for isolating this unstable intermediate and proceeding directly to a controlled reduction phase, the process achieves exceptional purity levels exceeding 98% while significantly simplifying the operational workflow. This technical breakthrough addresses the chronic supply shortages and high costs associated with traditional extraction methods, offering a viable pathway for reliable pharmaceutical intermediates supplier networks to meet global demand.
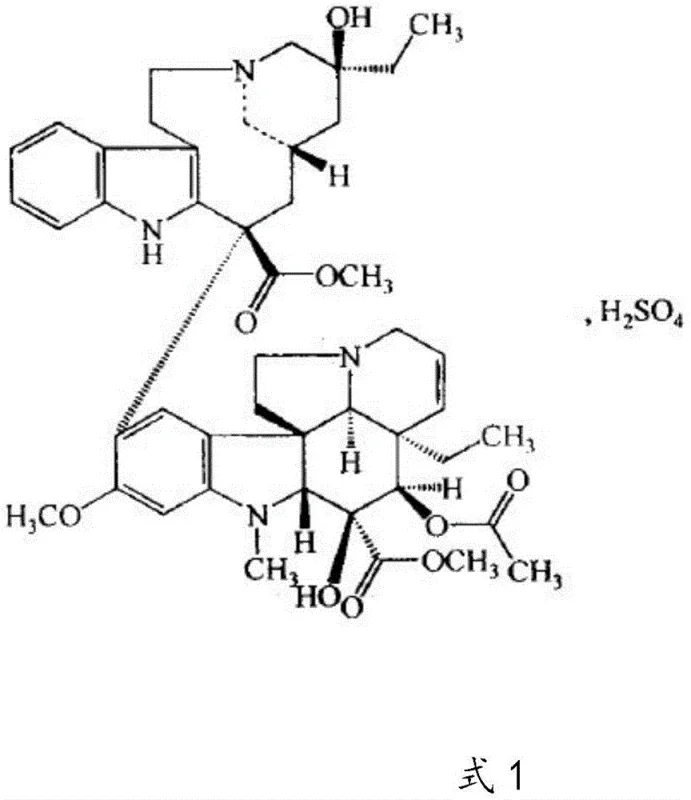
The strategic importance of this synthesis lies in its ability to utilize readily available raw materials that are far more abundant than the final API itself. Vindoline and catharanthine, extracted from the periwinkle plant, exist in much higher concentrations than vinblastine, making them economically superior starting points for cost reduction in pharmaceutical intermediates manufacturing. The process described involves a sophisticated interplay of oxidation and reduction steps, meticulously controlled by pH and temperature parameters to ensure high fidelity in the molecular assembly. For R&D directors and procurement managers alike, understanding the nuances of this iron-catalyzed pathway is essential for evaluating the long-term viability and supply security of this critical anticancer ingredient.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of Vinblastine Sulfate has been plagued by severe inefficiencies inherent to direct plant extraction and early-generation synthetic attempts. The traditional extraction process from Catharanthus roseus is notoriously inefficient, yielding only a few grams of high-purity product from a single ton of plant material, which creates a massive bottleneck for commercial scale-up of complex polymer additives and similar high-value chemicals. Furthermore, this method requires vast quantities of organic solvents for extraction and refining, generating substantial hazardous waste streams that pose significant environmental compliance challenges. On the synthetic front, prior art methods, such as those disclosed in patent CN201110378355.0, relied on dehydrated vinblastine as a direct starting material. However, this precursor is itself scarce and expensive, and the resulting processes often yielded products with content levels as low as 60%, necessitating extensive and costly downstream purification that eroded profit margins and compromised supply continuity.
The Novel Approach
In stark contrast, the methodology outlined in CN112480148A introduces a transformative strategy that circumvents these bottlenecks by synthesizing the dehydrated intermediate directly from abundant precursors. By reacting vindoline and catharanthine under the catalytic influence of ferric chloride and inorganic acid, the process generates the reactive dehydrated species in solution, eliminating the need for its isolation and purification. This "one-pot" generation of the intermediate not only reduces unit operations but also minimizes material loss, a critical factor in reducing lead time for high-purity pharmaceutical intermediates. The subsequent reduction step, utilizing sodium borohydride in the presence of ferric oxalate and air, is conducted under mild conditions that preserve the delicate stereochemistry of the molecule. This approach ensures that the final product meets stringent quality specifications with a molar yield exceeding 35% relative to vindoline, representing a substantial improvement in both efficiency and economic feasibility for large-scale manufacturers.
Mechanistic Insights into FeCl3-Catalyzed Oxidative Coupling
The core of this synthesis relies on a precise oxidative coupling mechanism facilitated by ferric chloride (FeCl3) in an acidic medium. In the initial stage, vindoline and catharanthine are dissolved in a mixed solvent system of tetrahydrofuran or trifluoroethanol and water, with the pH rigorously adjusted to between 1.0 and 1.5 using hydrochloric acid. The introduction of ferric chloride acts as a Lewis acid and oxidant, promoting the formation of a carbocation intermediate on the catharanthine moiety, which subsequently attacks the electron-rich indole ring of vindoline. This coupling reaction is exothermic and is maintained at a controlled temperature of 20-30°C to prevent degradation of the sensitive alkaloid structures. The result is the formation of dehydrated vinblastine, a crucial bridge molecule that retains the necessary structural integrity for the subsequent reduction to the final saturated bond found in vinblastine.
Following the coupling, the reaction mixture undergoes a critical reduction phase where the dehydrated intermediate is converted to vinblastine. This step involves the addition of the reaction solution to a chilled mixture of ferric oxalate and water, followed by the introduction of air and the dropwise addition of sodium borohydride. The presence of ferric oxalate and the controlled influx of oxygen likely serve to modulate the redox potential of the system, preventing over-reduction or side reactions. Crucially, the pH is maintained between 3.0 and 5.0 during the addition of the reducing agent using hydrochloric acid; this specific acidity window is vital to neutralize the alkalinity generated by sodium borohydride decomposition, thereby ensuring the stability of the acid-sensitive indole nuclei. The final purification employs a dual-column chromatography strategy, first using alkaline alumina to remove bulk impurities and then silica gel for fine polishing, ensuring the removal of trace isomers and byproducts to achieve the target purity of ≥98%.
How to Synthesize Vinblastine Sulfate Efficiently
Executing this synthesis requires strict adherence to the specified stoichiometric ratios and environmental controls to maximize yield and purity. The process begins with the preparation of the coupling reaction solution, followed immediately by the reduction step without intermediate workup, which is a key efficiency driver. Detailed operational parameters, including solvent volumes, reagent grades, and specific elution gradients for the chromatography steps, are critical for reproducibility. For a comprehensive breakdown of the standardized operating procedures and safety protocols required for this synthesis, please refer to the technical guide below.
- Prepare dehydrated vinblastine reaction solution by reacting vindoline and catharanthine with ferric chloride and inorganic acid at 20-30°C.
- Generate crude vinblastine by adding the reaction solution to ferric oxalate, introducing air, and reducing with sodium borohydride at controlled pH 3.0-5.0.
- Purify the crude product through a two-stage column chromatography process using alkaline alumina followed by silica gel.
- Form the final sulfate salt by reacting pure vinblastine with sulfuric acid in an organic solvent at low temperature.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this semi-synthetic route offers profound strategic advantages over traditional sourcing models. The primary benefit stems from the decoupling of production capacity from the volatile yields of direct plant extraction, which are subject to agricultural variables and seasonal fluctuations. By utilizing vindoline and catharanthine as feedstocks, manufacturers can leverage a more stable and abundant supply chain, as these precursors are present in the plant in significantly higher quantities than the final API. This shift effectively mitigates the risk of raw material shortages that have historically plagued the vinblastine market, ensuring a more predictable and continuous flow of goods for downstream drug formulation.
- Cost Reduction in Manufacturing: The elimination of the intermediate refining step for dehydrated vinblastine represents a significant operational saving. In conventional synthetic routes, isolating and purifying this intermediate incurs substantial solvent and labor costs, alongside yield losses. By processing the crude reaction solution directly into the reduction phase, this novel method drastically reduces solvent consumption and processing time. Furthermore, the use of inexpensive inorganic reagents like ferric chloride and ferric oxalate, instead of scarce or proprietary catalysts, lowers the overall bill of materials, driving down the cost of goods sold (COGS) without compromising quality.
- Enhanced Supply Chain Reliability: The reliance on chemically synthesized intermediates rather than direct extraction from tons of biomass reduces the logistical footprint and storage requirements. Direct extraction necessitates the handling and disposal of massive amounts of plant waste, creating logistical bottlenecks and environmental liabilities. In contrast, this chemical synthesis is compact and efficient, allowing for production in standard chemical facilities rather than specialized extraction plants. This flexibility enhances supply chain resilience, enabling manufacturers to respond more agilely to market demand spikes for this critical oncology medication.
- Scalability and Environmental Compliance: The process conditions are inherently scalable, operating at near-ambient temperatures and atmospheric pressure, which simplifies reactor design and safety requirements for commercial scale-up of complex pharmaceutical intermediates. The reduction in organic solvent usage, particularly in the extraction phase which is bypassed, aligns with modern green chemistry principles and stricter environmental regulations. The simplified waste profile, characterized by fewer organic byproducts and the use of recoverable solvents like dichloromethane and methanol in closed loops, facilitates easier waste treatment and regulatory approval, smoothing the path for large-scale commercial deployment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear picture of the technology's capabilities and limitations for potential adopters.
Q: Why is the Vindoline and Catharanthine route preferred over direct plant extraction for Vinblastine Sulfate?
A: Direct plant extraction yields extremely low quantities (grams per ton of plant material) and generates significant organic waste. The semi-synthetic route using Vindoline and Catharanthine utilizes raw materials that are more abundant in nature and cheaper, allowing for consistent, large-scale production with higher purity (>98%) and significantly reduced environmental impact.
Q: How does the novel iron-catalyzed method improve upon previous chemical synthesis patents?
A: Previous methods often relied on scarce dehydrated vinblastine as a starting material, resulting in low content (~60%) and difficult purification. This novel method generates the dehydrated intermediate in situ using inexpensive ferric chloride, eliminates the need for intermediate refining, and employs a precise pH-controlled reduction step to minimize byproducts, achieving yields over 35% with purity exceeding 98%.
Q: Is this synthesis method suitable for industrial scale-up?
A: Yes, the process is designed for scalability. It operates under mild reaction conditions (20-30°C and 0-10°C), uses common reagents like ferric oxalate and sodium borohydride, and avoids complex biotechnology equipment. The simplified workflow, which removes the need for intermediate isolation, makes it highly viable for commercial manufacturing from 100 kgs to multi-ton scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vinblastine Sulfate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that Vinblastine Sulfate plays in the global oncology landscape and are committed to delivering this essential API with uncompromising quality and reliability. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We employ stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets or exceeds the pharmacopoeial standards for identity, assay, and impurity profiles.
We invite pharmaceutical partners to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance your competitive edge in the marketplace.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
