Advanced Alkylation Technology for High-Purity Benzimidazole Intermediates and Commercial Scale-Up
Advanced Alkylation Technology for High-Purity Benzimidazole Intermediates and Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes that balance high purity with operational safety and economic efficiency. Patent CN101391982B introduces a transformative alkylation reaction method for benzimidazole compounds that addresses critical pain points in traditional synthesis. This technology leverages phase transfer catalysis to facilitate the N-alkylation of benzimidazoles under remarkably mild conditions, utilizing alkyl halides not just as reactants but effectively as solvents. For R&D directors and process chemists, this represents a significant departure from hazardous protocols involving pyrophoric metals, offering a pathway to high-purity pharmaceutical intermediates with simplified downstream processing. The method operates at temperatures between 35-100°C and achieves completion within 3 to 8 hours, demonstrating exceptional versatility across a wide range of substrates including complex bis-benzimidazole derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the alkylation of benzimidazole cores has relied heavily on aggressive dehydrogenation reagents such as sodium metal, potassium, sodium hydride, or sodium amide. These traditional approaches necessitate strictly anhydrous and oxygen-free environments to prevent catastrophic side reactions or ignition, imposing severe constraints on reactor design and operational safety protocols. Furthermore, alternative methods utilizing 50% sodium hydroxide often require substantial volumes of organic solvents to facilitate the reaction, leading to significant environmental burdens due to solvent emissions and difficult post-reaction separation processes. The reliance on flammable and explosive solvent systems increases the risk profile of manufacturing facilities, while the difficulty in removing residual solvents from the final product can compromise the purity specifications required for sensitive pharmaceutical applications. These factors collectively drive up production costs and extend lead times, creating bottlenecks in the supply chain for essential heterocyclic intermediates.
The Novel Approach
In stark contrast, the methodology described in CN101391982B employs a quaternary ammonium salt as a phase transfer catalyst, enabling the reaction to proceed efficiently with solid bases like sodium hydroxide or potassium hydroxide. A key innovation is the use of the alkyl halide reactant in excess, effectively serving as the reaction medium, which eliminates the need for additional volatile organic solvents. This dual-function role of the reagent simplifies the reaction mixture, reduces waste generation, and streamlines the recovery of unreacted materials through distillation. The process tolerates a broad range of temperatures from 35°C to 100°C, allowing for precise control over reaction kinetics without the need for extreme heating or cooling. As illustrated in the general reaction scheme below, this approach facilitates the clean conversion of benzimidazoles to their N-alkylated counterparts with high selectivity.
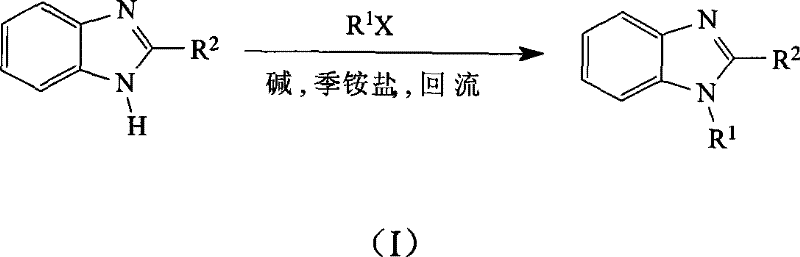
The structural diversity achievable with this method is extensive, accommodating various alkyl and arylmethyl groups ranging from C1 to C12. The patent data highlights the successful synthesis of complex architectures, including bis-benzimidazole ethane derivatives and poly-amine linked structures, which are often challenging to produce using conventional harsh conditions. By avoiding strong reducing agents, the integrity of sensitive functional groups on the benzimidazole ring is preserved, ensuring high product quality. This capability is crucial for manufacturers aiming to produce reliable agrochemical intermediate or pharmaceutical intermediate supplies where batch-to-batch consistency is paramount. The elimination of hazardous reagents also translates to a safer working environment, reducing the regulatory burden associated with handling dangerous chemicals.
Mechanistic Insights into Quaternary Ammonium Salt Catalyzed Alkylation
The core of this technological advancement lies in the efficient phase transfer catalysis mechanism mediated by quaternary ammonium salts. In this system, the solid base (such as NaOH) deprotonates the benzimidazole nitrogen at the interface or within the aqueous micro-environment, generating a nucleophilic benzimidazolide anion. The quaternary ammonium cation acts as a shuttle, transporting this anionic species into the organic phase where the alkyl halide is abundant. Once in the organic phase, the nucleophile attacks the electrophilic carbon of the alkyl halide via an SN2 mechanism, displacing the halide ion and forming the new C-N bond. This cycle continues efficiently, driven by the lipophilicity of the catalyst which ensures high concentration of the reactive anion in the organic layer. The use of excess alkyl halide ensures that the equilibrium is pushed towards product formation while maintaining a homogeneous-like reaction environment despite the heterogeneous nature of the solid base.
Impurity control is inherently superior in this system due to the absence of radical pathways often associated with sodium metal reductions. Traditional methods can lead to over-reduction or coupling byproducts that are difficult to separate, whereas this ionic mechanism is highly selective for N-alkylation. The mild basicity of hydroxide compared to hydride minimizes the risk of base-catalyzed degradation of the benzimidazole ring or other sensitive moieties attached to the scaffold. Furthermore, the ability to recover excess alkyl halide via distillation not only improves atom economy but also removes potential alkylating impurities from the final crude product before crystallization. This results in a cleaner crude profile, reducing the load on purification steps and enhancing the overall yield of high-purity benzimidazole derivatives suitable for downstream drug synthesis.
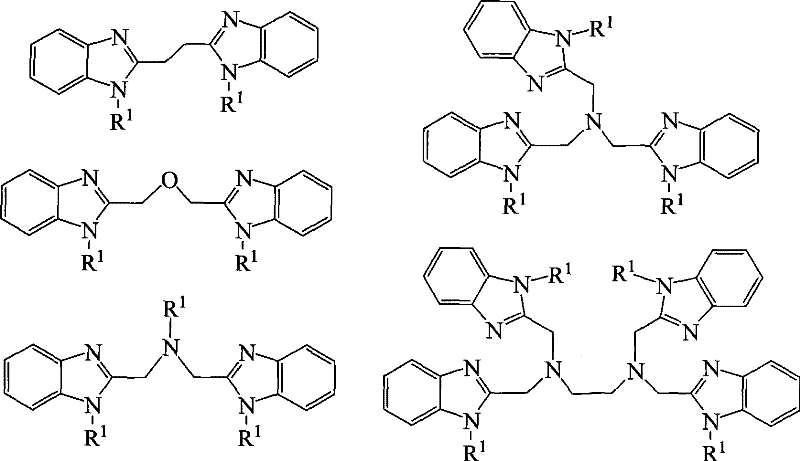
How to Synthesize N-Alkylated Benzimidazoles Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and thermal management to maximize the benefits of the phase transfer system. The patent outlines a straightforward protocol where benzimidazole, alkyl halide, quaternary ammonium salt, and base are combined in a standard reaction vessel. The detailed standardized synthesis steps see the guide below, which covers the precise molar ratios and temperature profiles optimized for different substrates. Operators should note that the molar ratio of benzimidazole to alkyl halide typically ranges from 1:6 to 1:36, ensuring the halide serves effectively as the solvent. Reaction monitoring via TLC is recommended to determine the exact endpoint, usually falling between 3 to 8 hours depending on the steric hindrance of the alkyl group.
- Charge the reactor with benzimidazole substrate, excess alkyl halide (acting as solvent), quaternary ammonium salt catalyst, and solid base (NaOH or KOH).
- Heat the reaction mixture to a temperature range of 35-100°C and maintain stirring for 3 to 8 hours until TLC indicates complete consumption of the starting material.
- Recover excess alkyl halide via distillation, neutralize the residue with dilute acid, filter the solid product, and recrystallize from ethanol to obtain high-purity intermediates.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this alkylation technology offers profound strategic advantages beyond mere chemical efficiency. The shift away from pyrophoric reagents like sodium hydride eliminates the need for specialized storage facilities and hazardous material handling certifications, directly lowering operational overheads. Additionally, the reduction in solvent usage significantly cuts down on raw material costs and waste disposal fees, contributing to substantial cost savings in pharmaceutical intermediates manufacturing. The robustness of the reaction conditions allows for flexible scheduling and reduced downtime, as the process is less susceptible to failures caused by moisture ingress compared to anhydrous methods. This reliability ensures a steady flow of materials, reducing lead time for high-purity benzimidazole intermediates and stabilizing the supply chain against market volatility.
- Cost Reduction in Manufacturing: The elimination of expensive, hazardous reagents and the reduction of solvent volumes lead to a drastically simplified cost structure. By using the alkyl halide as the solvent, the process removes the need for purchasing, storing, and disposing of large quantities of organic solvents, which are often subject to fluctuating market prices and strict environmental taxes. Furthermore, the high yields reported in the patent examples, ranging from 71% to 88%, indicate excellent material efficiency, minimizing the loss of valuable starting materials. This efficiency translates directly to a lower cost of goods sold (COGS), allowing for more competitive pricing strategies in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: The use of stable, commercially available reagents such as sodium hydroxide and common alkyl halides ensures that raw material sourcing is secure and resilient. Unlike specialized reagents that may have limited suppliers or long lead times, the inputs for this process are commodity chemicals with robust global supply chains. The simplicity of the workup procedure, involving filtration and recrystallization, reduces the dependency on complex purification equipment that might become a bottleneck in multi-product facilities. This operational simplicity enhances the overall agility of the manufacturing site, enabling faster response times to urgent customer demands and ensuring consistent delivery schedules.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scale-up, with reaction conditions that are easily manageable in large-scale reactors without requiring exotic materials of construction. The reduced generation of hazardous waste and lower VOC emissions align perfectly with increasingly stringent environmental regulations, mitigating the risk of compliance-related shutdowns. The ability to recycle excess alkyl halide further demonstrates a commitment to circular economy principles, enhancing the sustainability profile of the manufacturing operation. This environmental stewardship is increasingly valued by downstream partners who are under pressure to reduce the carbon footprint of their own supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this benzimidazole alkylation technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of integrating this method into existing production lines or for procurement specialists assessing the quality and reliability of the supply source.
Q: What are the safety advantages of this alkylation method compared to traditional sodium hydride methods?
A: Traditional methods often require pyrophoric reagents like sodium hydride or sodium metal under strict anhydrous conditions. This patented process utilizes stable solid bases like sodium hydroxide and eliminates the need for hazardous anhydrous organic solvents, significantly reducing fire risks and handling complexity.
Q: How does the process handle waste management and environmental compliance?
A: By using the alkyl halide reactant as the solvent, the process drastically reduces the volume of volatile organic compounds (VOCs) typically associated with separate solvent systems. This simplifies downstream recovery and minimizes hazardous waste generation, aligning with modern green chemistry principles.
Q: Is this method suitable for large-scale commercial production of complex bis-benzimidazoles?
A: Yes, the patent demonstrates high yields (71-88%) across various substrates, including complex bis-benzimidazole structures. The mild reaction conditions (35-100°C) and simple workup procedures make it highly scalable for industrial manufacturing without requiring specialized high-pressure or cryogenic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzimidazole Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the phase transfer catalyzed alkylation of benzimidazoles are translated into reliable commercial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the highest standards of quality and consistency required for drug substance manufacturing. Our commitment to technical excellence allows us to navigate complex chemical landscapes, delivering solutions that optimize both performance and cost.
We invite you to collaborate with us to leverage these technological advancements for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality targets. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in benzimidazole chemistry can drive value and efficiency in your supply chain.
