Scalable Synthesis of 2-Trifluoromethyl Benzimidazoles Using Recyclable Copper Catalysts
Scalable Synthesis of 2-Trifluoromethyl Benzimidazoles Using Recyclable Copper Catalysts
The pharmaceutical and agrochemical industries continuously demand efficient, scalable, and environmentally benign synthetic routes for nitrogen-containing heterocycles, particularly benzimidazole derivatives which serve as critical scaffolds in drug discovery. A significant technological breakthrough in this domain is detailed in Chinese Patent CN113429349B, which discloses a robust preparation method for 2-trifluoromethyl substituted benzimidazole compounds utilizing heterogeneous catalysis. This innovation addresses long-standing challenges in traditional synthesis by employing a copper-doped carbon nitride (Cu/g-C3N4) catalyst, enabling reactions under mild conditions with high efficiency. For R&D directors and procurement managers seeking reliable pharmaceutical intermediate suppliers, this technology represents a paradigm shift towards sustainable manufacturing, offering a pathway to reduce heavy metal residues and simplify downstream processing while maintaining high yields across a broad substrate scope.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted benzimidazoles has relied on several conventional strategies, each fraught with significant operational and economic drawbacks that hinder large-scale commercialization. Traditional methods often involve the condensation of o-phenylenediamine with trifluoroacetic acid or trifluoroacetaldehyde, which typically requires harsh reaction conditions and generates substantial waste. Alternatively, the reductive cyclization of o-nitroaniline with trifluoroacetic acid or direct trifluoromethylation of existing benzimidazole cores often suffers from narrow substrate tolerance and the use of expensive, hazardous reagents. Furthermore, earlier reports on copper-catalyzed tandem cyclization utilized homogeneous copper systems. While effective, homogeneous catalysts pose severe challenges for industrial application, including difficult removal from the final product to meet stringent regulatory limits on residual metals, and the inability to recycle the expensive metal complex, leading to inflated production costs and environmental burdens.
The Novel Approach
In stark contrast to these legacy methods, the novel approach described in the patent leverages a heterogeneous copper-doped carbon nitride catalyst to drive the tandem cyclization of trifluoroethylimidoyl chlorides and amines. This methodology operates under significantly milder conditions, typically between 70°C and 90°C, utilizing inexpensive potassium carbonate as a base in common organic solvents like DMF. The core innovation lies in the heterogeneity of the catalyst, which allows for simple filtration post-reaction, effectively eliminating the need for complex metal scavenging steps. 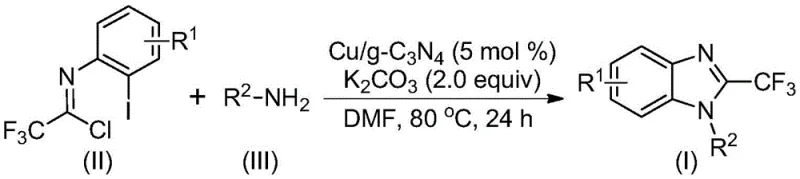 As illustrated in the reaction scheme, this process facilitates the direct formation of the benzimidazole core with high atom economy. The ability to recycle the catalyst further enhances the economic viability, making this route exceptionally attractive for cost reduction in pharmaceutical intermediate manufacturing where margin compression is a constant pressure.
As illustrated in the reaction scheme, this process facilitates the direct formation of the benzimidazole core with high atom economy. The ability to recycle the catalyst further enhances the economic viability, making this route exceptionally attractive for cost reduction in pharmaceutical intermediate manufacturing where margin compression is a constant pressure.
Mechanistic Insights into Cu/g-C3N4 Catalyzed Tandem Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific analogs. The reaction initiates with a nucleophilic addition-elimination sequence between the trifluoroethylimidoyl chloride and the amine substrate, generating an amidine intermediate in situ. Subsequently, the copper species embedded within the carbon nitride matrix plays a pivotal role by inserting into the carbon-iodine bond of the aryl ring. This oxidative addition step forms a transient copper complex, which, under the influence of the base, undergoes deprotonation to eliminate hydrogen iodide. This leads to the formation of a divalent or trivalent cyclocopper intermediate, a key species that dictates the cyclization efficiency. Finally, a reductive elimination step releases the desired 2-trifluoromethyl substituted benzimidazole product and regenerates the active copper catalytic species, completing the cycle. This mechanism highlights the dual functionality of the system in promoting both C-N bond formation and C-C/N cyclization efficiently.
From an impurity control perspective, the heterogeneous nature of the catalyst provides a distinct advantage in managing the impurity profile of the final API intermediate. In homogeneous systems, leached copper ions can catalyze side reactions or form stable complexes with the product, complicating purification. However, the Cu/g-C3N4 system confines the active metal sites within the solid support, minimizing metal leaching into the solution phase. This containment strategy ensures that the crude reaction mixture contains fewer metal-associated impurities, thereby simplifying the subsequent purification workflow. Moreover, the mild basic conditions provided by potassium carbonate minimize the risk of hydrolysis of sensitive functional groups, ensuring high chemical purity. For quality control laboratories, this translates to more consistent batch-to-batch reproducibility and easier compliance with ICH guidelines regarding elemental impurities, a critical factor for any reliable pharmaceutical intermediate supplier.
How to Synthesize 2-Trifluoromethyl Benzimidazole Efficiently
The practical implementation of this synthesis route is designed for scalability and ease of operation, making it suitable for both laboratory optimization and pilot plant production. The protocol involves charging a reactor with the specific trifluoroethylimidoyl chloride, the chosen amine, potassium carbonate, and the Cu/g-C3N4 catalyst in a solvent such as DMF. The mixture is then heated to the optimal temperature range of 80°C for approximately 24 hours, although the patent notes a window of 18 to 30 hours depending on the specific substrate reactivity. 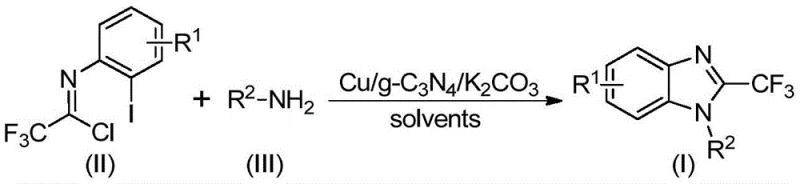 Following the reaction period, the solid catalyst is removed via simple filtration, and the filtrate is concentrated. The crude product is then purified, typically via column chromatography or recrystallization, to afford the high-purity target compound. The detailed standardized synthesis steps, including precise stoichiometric ratios and workup procedures validated across multiple examples, are outlined below to ensure reproducibility.
Following the reaction period, the solid catalyst is removed via simple filtration, and the filtrate is concentrated. The crude product is then purified, typically via column chromatography or recrystallization, to afford the high-purity target compound. The detailed standardized synthesis steps, including precise stoichiometric ratios and workup procedures validated across multiple examples, are outlined below to ensure reproducibility.
- Prepare the reaction mixture by adding copper-doped carbon nitride (Cu/g-C3N4), potassium carbonate, trifluoroethylimidoyl chloride, and the corresponding amine into an organic solvent such as DMF.
- Heat the reaction mixture to a temperature between 70°C and 90°C and maintain stirring for a duration of 18 to 30 hours to ensure complete conversion.
- Upon completion, filter the reaction mixture to recover the heterogeneous catalyst, followed by silica gel mixing and column chromatography purification to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this heterogeneous catalytic process offers transformative benefits that directly impact the bottom line and operational resilience. The shift from homogeneous to heterogeneous catalysis fundamentally alters the cost structure of producing complex fluorinated heterocycles. By eliminating the need for expensive ligand systems and enabling catalyst recovery, the raw material costs are significantly optimized. Furthermore, the simplified workup procedure reduces the consumption of auxiliary materials such as metal scavengers and extensive washing solvents, contributing to substantial cost savings in waste management and processing time. This efficiency gain allows for more competitive pricing strategies in the global market for fine chemical intermediates.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the recyclability of the Cu/g-C3N4 catalyst and the use of commodity chemicals like potassium carbonate. Unlike precious metal catalysts that are lost in the waste stream, this copper-based system can be recovered and reused, drastically lowering the cost per kilogram of the active pharmaceutical ingredient. Additionally, the high reaction efficiency and yields observed (often exceeding 90%) minimize the loss of valuable starting materials, ensuring that the overall process mass intensity is favorable. This logical deduction of cost efficiency makes the technology highly viable for large-scale commercial production where margin preservation is critical.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on specialized, hard-to-source reagents. This method utilizes trifluoroethylimidoyl chlorides and amines that are readily available or easily synthesized from common precursors, reducing the risk of supply bottlenecks. The robustness of the reaction conditions also means that the process is less sensitive to minor fluctuations in operating parameters, leading to higher first-pass yields and more predictable delivery schedules. For supply chain planners, this reliability translates to reduced safety stock requirements and a more agile response to market demand fluctuations for key benzimidazole building blocks.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new safety and environmental challenges, but this heterogeneous system is inherently safer and greener. The absence of pyrophoric reagents or extreme temperatures reduces the hazard profile of the manufacturing facility. Moreover, the ability to filter and reuse the catalyst aligns perfectly with green chemistry principles, reducing the generation of heavy metal waste. This environmental compliance is increasingly important for maintaining operational licenses and meeting the sustainability goals of multinational corporate partners, ensuring long-term viability of the supply chain.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific projects, we have compiled answers to common inquiries based on the patent data. These questions address critical aspects of catalyst performance, substrate compatibility, and process scalability. Understanding these technical nuances is essential for making informed decisions about integrating this synthetic route into your existing manufacturing portfolio. The following insights are derived directly from the experimental results and mechanistic studies presented in the intellectual property documentation.
Q: What are the advantages of using Cu/g-C3N4 over homogeneous copper catalysts?
A: The primary advantage is the ease of separation. Unlike homogeneous catalysts which require complex removal processes to meet ppm limits, the heterogeneous Cu/g-C3N4 can be simply filtered off and reused, significantly reducing metal contamination risks and processing costs.
Q: Can this method tolerate diverse functional groups on the substrate?
A: Yes, the patent demonstrates excellent substrate scope. It successfully synthesizes derivatives with various substituents including halogens (Cl, Br), alkyl groups (methyl, trifluoromethyl), and different amine types (alkyl, benzyl, phenyl), making it highly versatile for library synthesis.
Q: Is the catalyst reusable without significant loss of activity?
A: Experimental data indicates the catalyst is recyclable. While there is a gradual decrease in efficiency over multiple cycles (92%, 80%, 69% over three runs), it remains active, offering substantial economic benefits compared to single-use homogeneous systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Benzimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced catalytic technologies in driving the next generation of pharmaceutical innovations. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. By leveraging the efficiencies of the Cu/g-C3N4 catalyzed route, we can offer our partners a secure and cost-effective supply of 2-trifluoromethyl benzimidazole derivatives, mitigating the risks associated with traditional synthesis methods.
We invite you to collaborate with us to explore how this technology can enhance your drug development pipeline. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact us to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary molecules. Let us be your partner in achieving chemical excellence and supply chain resilience.
