Advanced Stereoselective Synthesis of Chiral Lactones for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for the production of high-value chiral intermediates, particularly those serving as critical building blocks for active pharmaceutical ingredients (APIs). Patent CN109251207B, published in March 2020, introduces a groundbreaking method for the stereoselective synthesis of chiral lactones that addresses long-standing inefficiencies in resolution processes. This technology represents a significant leap forward in process chemistry, specifically targeting the optimization of reaction selectivity and yield while simultaneously solving the persistent issue of auxiliary recovery. By leveraging a unique quaternary ammonium salt pathway, the inventors have developed a route that eliminates the need for harsh saponification conditions typically associated with expensive lithium hydroxide treatments. For R&D directors and process chemists, this patent offers a compelling alternative to legacy methods, promising not only higher purity profiles but also a more streamlined operational workflow that is inherently safer and more cost-effective. The ability to recover chiral alcohols and organic tertiary amines with high efficiency transforms the economic model of producing these complex heterocyclic structures, making it a highly attractive candidate for commercial scale-up in the competitive landscape of pharmaceutical intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral lactones has been plagued by complex resolution processes that suffer from poor atom economy and difficult downstream processing. Early methods, such as the asymmetric synthesis invented by Sumitomo Corporation in 1973, relied on dehydration reactions with dicarboxylic acids followed by reduction and ring closure. While foundational, these approaches often exhibited poor diastereoselectivity, resulting in low resolution efficiency and yields hovering around only 65%, with significant challenges in recycling the chiral amine auxiliaries. Subsequent advancements, such as those disclosed by Roche in 1985 involving compounds of general formula III and IIIA, attempted to improve selectivity but introduced new complexities regarding reagent preparation and recovery.
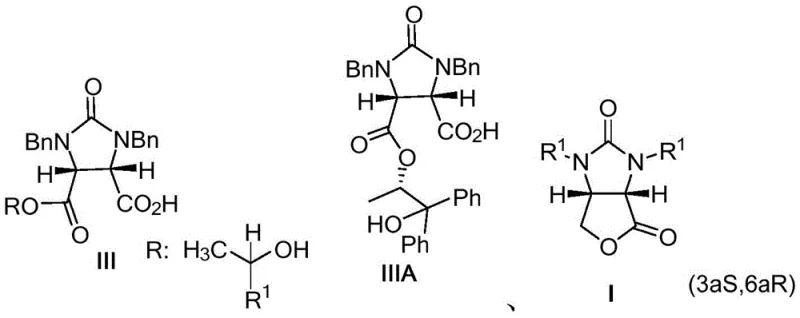
Furthermore, methods utilizing cinchona alkaloids or ephedrine-based amino alcohol derivatives, while capable of achieving high selectivity, often require ligands that are complicated to synthesize, expensive to procure, and notoriously difficult to recover for reuse. A critical bottleneck in many of these prior art processes is the reliance on expensive aqueous lithium hydroxide solutions to treat dicarboxylate esters. This step requires extremely precise control of alkali addition and reaction conditions; even minor deviations can lead to the saponification of the dicarboxylic monoester, reverting materials to starting components and drastically reducing overall yield. These operational sensitivities make conventional methods risky for large-scale manufacturing, where consistency and cost control are paramount.
The Novel Approach
The methodology disclosed in patent CN109251207B fundamentally reimagines the synthetic pathway by utilizing a diastereoselective reaction between a racemic cyclic anhydride and a chiral alcohol in the presence of an organic base to generate a dicarboxylic acid monoester quaternary ammonium salt. This intermediate, represented as formula (3), serves as a pivotal junction in the synthesis, allowing for direct reduction without the need for prior acidification or base treatment that typically triggers saponification. The process flow is elegantly designed to maximize efficiency: the quaternary ammonium salt is subjected to a selective reduction using a borohydride reducing agent to form the alkyd quaternary ammonium salt (formula 4), which is then converted to a metal salt before final ring closure under acidic conditions.
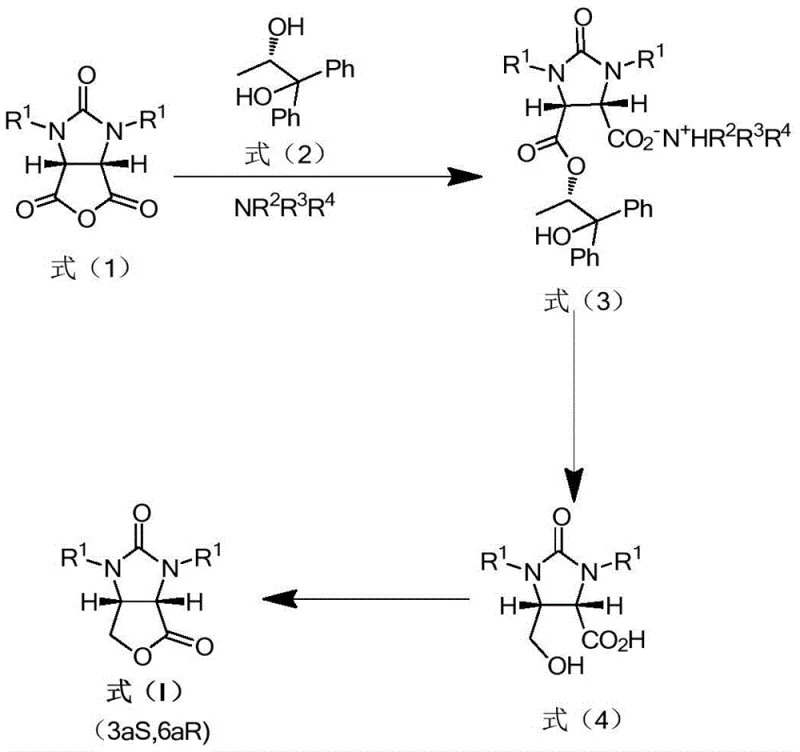
This novel approach effectively decouples the resolution step from the harsh conditions that degrade product quality in traditional routes. By avoiding the use of expensive lithium hydroxide and eliminating the saponification side reaction, the process achieves superior yields, often exceeding 96% in experimental examples, with diastereomeric purity (de value) reaching up to 99% and enantiomeric excess (ee value) of the final lactone product consistently at 99%. Moreover, the design facilitates the straightforward recovery of the chiral alcohol and the organic tertiary amine from the organic phase after the reduction step. These recovered materials can be recycled directly back into the first reaction stage, creating a closed-loop system that significantly lowers the cost of goods sold (COGS) and minimizes waste generation, addressing both economic and environmental concerns inherent in fine chemical manufacturing.
Mechanistic Insights into Diastereoselective Ring Opening and Reduction
The core mechanistic advantage of this synthesis lies in the formation and stability of the quaternary ammonium salt intermediate. In the first reaction step, the cyclic anhydride (formula 1) undergoes a ring-opening reaction with the chiral alcohol (formula 2) catalyzed by an organic tertiary amine (NR2R3R4). This reaction proceeds under mild conditions, typically between -20°C and 60°C, to form the quaternary ammonium salt (formula 3) with high diastereoselectivity. The choice of solvent, such as xylene or toluene, and the specific tertiary amine, such as tributylamine or triethylamine, plays a crucial role in stabilizing this ionic intermediate and ensuring high stereocontrol. Unlike free acid or ester forms which might be prone to racemization or hydrolysis, the quaternary salt structure locks the stereochemistry effectively, setting the stage for the subsequent reduction.
Following the formation of the quaternary salt, the process employs a selective reduction strategy using organoboron hydrides, preferably lithium borohydride, in a mixed solvent system of water and tetrahydrofuran. This step converts one of the carbonyl groups of the imidazolidine-2,4-dione derivative into a hydroxymethyl group, yielding the alkyd quaternary ammonium salt (formula 4). Crucially, because the carboxylate is masked as a quaternary ammonium salt rather than a free acid or a lithium salt susceptible to hydrolysis, the reduction proceeds cleanly without the competing saponification reactions that plague prior art methods. The subsequent treatment with an alkaline substance, such as sodium hydroxide, converts the quaternary salt into a metal salt of the alcohol acid, which partitions into the aqueous phase. This phase separation is key to the purification strategy, allowing the organic phase containing the valuable chiral alcohol and tertiary amine to be easily separated, recovered, and recycled, while the aqueous phase containing the product precursor is carried forward to the final acidification and cyclization step to yield the target chiral lactone (formula I).
How to Synthesize (3aS,6aR)-1,3-dibenzyltetrahydro-1H-furo[3,4-d]imidazole-2,4-dione Efficiently
The synthesis of this high-value chiral lactone involves a carefully orchestrated sequence of four distinct reaction stages that prioritize stereochemical integrity and material efficiency. The process begins with the diastereoselective ring opening of a cyclic anhydride using a chiral alcohol auxiliary in the presence of an organic base, followed by a selective reduction of the resulting quaternary ammonium salt intermediate. The subsequent steps involve the conversion of the reduced intermediate into a metal salt and a final acid-catalyzed cyclization to close the lactone ring. Detailed standardized operating procedures, including specific temperature ramps, solvent ratios, and workup protocols optimized for maximum yield and purity, are outlined in the comprehensive guide below.
- React cyclic anhydride with chiral alcohol and organic tertiary amine to form a quaternary ammonium salt intermediate.
- Perform a selective reduction on the quaternary ammonium salt using a borohydride reducing agent to obtain the alkyd quaternary ammonium salt.
- Treat the reduced intermediate with an alkaline substance to form a metal salt, followed by acidification to induce ring closure and obtain the final chiral lactone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this stereoselective synthesis method offers transformative benefits that extend far beyond simple yield improvements. The primary economic driver is the drastic simplification of the raw material profile and the elimination of costly, hard-to-source reagents. By removing the dependency on expensive lithium hydroxide and replacing it with more common and affordable alkaline substances like sodium hydroxide, the direct material costs are significantly reduced. Furthermore, the ability to recover and recycle the chiral alcohol and organic tertiary amine auxiliaries creates a sustainable supply loop that insulates the manufacturing process from volatility in the pricing of specialized chiral reagents. This closed-loop capability ensures a more predictable cost structure and reduces the overall inventory burden required to maintain continuous production schedules.
- Cost Reduction in Manufacturing: The elimination of the saponification side reaction is a major cost-saving factor, as it prevents the loss of valuable intermediates that would otherwise revert to starting materials in traditional processes. Additionally, the avoidance of expensive lithium hydroxide treatments reduces reagent costs substantially. The high recovery rate of chiral auxiliaries means that the effective consumption of these high-value additives per kilogram of product is minimized, leading to a leaner and more cost-efficient production model that enhances overall profit margins without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes directly to supply chain stability. Because the process tolerates a wider range of operating conditions compared to the precision-demanding lithium hydroxide steps of the past, the risk of batch failure due to minor process deviations is greatly diminished. The use of commercially available solvents like xylene and toluene, along with common reducing agents like borohydrides, ensures that raw material sourcing is not a bottleneck. This reliability allows for more accurate forecasting and shorter lead times for high-purity pharmaceutical intermediates, enabling downstream API manufacturers to plan their production cycles with greater confidence.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process is inherently greener and easier to scale. The reduction in waste generation, driven by the recycling of auxiliaries and the absence of saponification by-products, simplifies wastewater treatment and lowers disposal costs. The phase separation strategy utilized in the workup allows for efficient isolation of the product without the need for complex chromatographic purifications, which are often difficult to scale. This operational simplicity facilitates a smoother transition from pilot plant to commercial scale production, ensuring that the supply of complex chiral lactones can be ramped up rapidly to meet market demand while adhering to strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this stereoselective synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on how this method resolves specific pain points associated with chiral lactone production. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this method improve upon traditional lithium hydroxide treatments?
A: Traditional methods often require expensive lithium hydroxide for saponification, which is difficult to control and leads to side reactions. This novel process avoids expensive lithium hydroxide entirely, preventing saponification of the dicarboxylic monoester and significantly simplifying the purification workflow.
Q: What is the advantage regarding chiral auxiliary recovery?
A: A major bottleneck in prior art was the difficulty in recovering chiral amines and alcohols. This invention enables the high-yield recovery of both the chiral alcohol and the organic tertiary amine, allowing them to be recycled back into the first reaction step, thereby drastically reducing raw material consumption.
Q: What level of stereoselectivity can be achieved with this process?
A: The process demonstrates excellent diastereoselectivity during the initial ring-opening step, with de values reaching up to 99%. The final chiral lactone product is obtained with high enantiomeric purity, typically achieving 99% ee, ensuring suitability for high-specification pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Lactone Supplier
The technological potential of this stereoselective synthesis route is immense, offering a pathway to high-purity chiral lactones that are critical for the development of next-generation therapeutics. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical advantages of this patent can be fully realized in an industrial setting. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of chiral lactone delivered meets the exacting standards required by global pharmaceutical clients. We understand the nuances of handling chiral auxiliaries and optimizing reduction steps to maximize yield and minimize impurities.
We invite you to collaborate with us to optimize your supply chain for these critical intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how switching to this advanced synthesis method can impact your bottom line. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in commercial scale-up of complex pharmaceutical intermediates can support your long-term strategic goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
