Revolutionizing Quinoline Production: A Mild, One-Step Catalytic Route for High-Purity Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing privileged scaffolds such as quinolines, which are ubiquitous in bioactive molecules ranging from antimalarials to kinase inhibitors. Patent CN114890944A introduces a transformative preparation method that addresses the longstanding challenges associated with traditional quinoline synthesis by utilizing a difluorocarbene reagent in the presence of a metal additive and base. This innovative approach enables the direct conversion of readily available o-isopropenylphenylaniline derivatives into functionalized quinoline compounds through a streamlined one-step reaction protocol. The significance of this technology lies in its ability to bypass the severe reaction conditions typically required for heterocycle formation, thereby offering a safer and more efficient pathway for generating high-value chemical intermediates. By leveraging sodium difluorochloroacetate as a key building block, the process achieves remarkable yields while maintaining operational simplicity, making it an attractive candidate for industrial adoption.
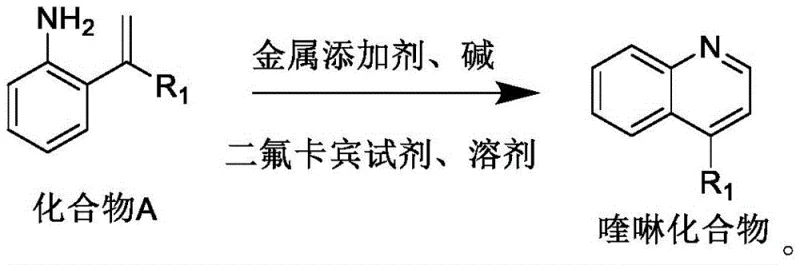
Furthermore, the versatility of this synthetic route allows for the modular introduction of diverse substituents at the 4-position of the quinoline ring, facilitating the rapid exploration of structure-activity relationships in drug discovery programs. The method described in the patent represents a significant leap forward in organic synthesis, providing chemists with a reliable tool for accessing complex nitrogen-containing heterocycles without the need for multi-step sequences or hazardous reagents. As global demand for high-purity pharmaceutical intermediates continues to rise, technologies that offer both economic and environmental advantages become critical assets for forward-thinking chemical manufacturers. This patent not only solves specific synthetic problems but also aligns with the broader industry trend towards greener and more sustainable chemical processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinoline derivatives has relied heavily on classical methodologies such as the Skraup reaction, which involves the condensation of aniline with glycerol in the presence of strong acids and oxidizing agents. These traditional protocols are fraught with significant drawbacks, including the requirement for excessively high temperatures and highly corrosive acidic environments that pose serious safety risks and equipment corrosion issues. The exothermic nature of the Skraup reaction often leads to difficult temperature control, resulting in the formation of numerous by-products and tars that complicate downstream purification efforts. Moreover, the use of concentrated sulfuric acid generates substantial amounts of acidic waste, creating environmental burdens and increasing the cost of waste treatment and disposal. The subsequent separation of the desired quinoline product from the reaction mixture is notoriously labor-intensive, often requiring multiple extraction and neutralization steps that reduce overall process efficiency. Additionally, the substrate scope of these classical methods is frequently limited, struggling to accommodate sensitive functional groups that may degrade under such harsh thermal and acidic conditions.
The Novel Approach
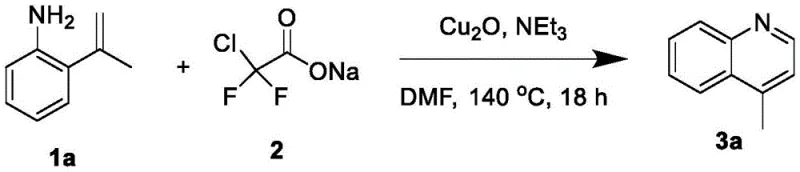
In stark contrast, the method disclosed in patent CN114890944A utilizes a mild copper-catalyzed cyclization strategy that operates under significantly more benign conditions, effectively eliminating the need for strong mineral acids. By employing sodium difluorochloroacetate as a difluorocarbene source, the reaction proceeds through a unique mechanistic pathway that constructs the quinoline core with high atom economy and selectivity. The use of organic bases like triethylamine instead of inorganic acids creates a much safer working environment and simplifies the quenching and workup procedures considerably. This novel approach allows for the direct transformation of o-isopropenylphenylaniline precursors into the target quinolines in a single pot, drastically reducing the number of unit operations required compared to multi-step classical syntheses. The compatibility of this method with a wide range of substituents, including halogens and alkyl groups, demonstrates its superior flexibility for synthesizing diverse libraries of quinoline derivatives. Ultimately, this shift from harsh acid-mediated cyclization to transition-metal catalysis represents a paradigm shift in how these critical intermediates are manufactured.
Mechanistic Insights into Cu-Catalyzed Difluorocarbene Cyclization
The core of this innovative synthesis lies in the generation and utilization of difluorocarbene species mediated by a copper catalyst, specifically cuprous oxide (Cu2O), which plays a pivotal role in activating the difluorocarbene reagent. The reaction initiates with the interaction between the copper species and sodium difluorochloroacetate, likely facilitating the elimination of chloride and carbon dioxide to generate a reactive copper-carbene intermediate or free difluorocarbene in situ. This electrophilic species then engages with the electron-rich double bond of the o-isopropenylphenylaniline substrate, triggering a cascade of cyclization events that forge the new carbon-carbon and carbon-nitrogen bonds essential for the quinoline framework. The presence of the base, such as triethylamine, is crucial for neutralizing the hydrochloric acid by-product and maintaining the catalytic cycle, ensuring that the copper species remains active throughout the prolonged heating period at 140°C. Detailed analysis of the reaction scope suggests that the electronic properties of the substituents on the vinyl group influence the reaction rate, yet the robust nature of the copper catalyst accommodates both electron-donating and electron-withdrawing groups effectively. Understanding this mechanism is vital for process chemists aiming to optimize reaction parameters further, as it highlights the delicate balance between carbene generation and substrate consumption.
From an impurity control perspective, the mildness of this catalytic system inherently suppresses the formation of polymeric tars and oxidation by-products that plague acid-catalyzed routes. The absence of strong oxidants and dehydrating agents minimizes side reactions such as polymerization of the vinyl group or degradation of the aniline moiety, leading to a cleaner crude reaction profile. The primary impurities observed are likely unreacted starting materials or minor regioisomers, which are readily removed through standard silica gel column chromatography as demonstrated in the patent examples. This high level of chemoselectivity ensures that the final quinoline products possess the stringent purity profiles required for pharmaceutical applications, reducing the need for extensive recrystallization or preparative HPLC purification. Furthermore, the stability of the intermediates under the reaction conditions implies that the process is robust against minor fluctuations in temperature or stoichiometry, enhancing its reliability for large-scale production. The mechanistic clarity provided by this patent empowers manufacturers to implement rigorous quality control measures, ensuring consistent batch-to-batch reproducibility.
How to Synthesize Quinoline Derivatives Efficiently
The practical implementation of this synthesis route is straightforward and relies on standard laboratory equipment, making it highly accessible for process development teams aiming to scale up quinoline production. The procedure begins with the careful weighing and mixing of the o-isopropenylphenylaniline substrate, sodium difluorochloroacetate, and the cuprous oxide catalyst in a dry reaction vessel, followed by evacuation and backfilling with nitrogen to create an inert atmosphere. A solution of triethylamine in DMF is then introduced to the mixture, which is subsequently heated in an oil bath to maintain a precise temperature of 140°C for a duration of 18 hours to ensure complete conversion. Upon completion, the reaction mixture is cooled to room temperature and diluted with methylene chloride, allowing for the filtration of insoluble inorganic salts and catalyst residues before concentration. The resulting crude oil is then subjected to flash column chromatography to isolate the pure quinoline product, a process that is significantly less cumbersome than the aqueous workups required for traditional methods. For a comprehensive understanding of the specific stoichiometric ratios and handling precautions, please refer to the detailed standardized synthesis steps outlined below.
- Mix o-isopropenylphenylaniline derivative with sodium difluorochloroacetate and cuprous oxide in a reaction vessel under nitrogen atmosphere.
- Add triethylamine dissolved in DMF solvent to the mixture and stir thoroughly.
- Heat the reaction mixture to 140°C for 18 hours, then cool, dilute with methylene chloride, filter, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed methodology offers tangible benefits that directly impact the bottom line and operational resilience of chemical manufacturing operations. The shift away from hazardous concentrated sulfuric acid and glycerol eliminates the need for specialized acid-resistant reactors and complex neutralization infrastructure, thereby reducing capital expenditure and maintenance costs associated with corrosion management. The use of commercially available and inexpensive reagents like sodium difluorochloroacetate and cuprous oxide ensures a stable supply chain with minimal risk of raw material shortages or price volatility. Moreover, the simplified post-treatment process, which avoids extensive aqueous washing and pH adjustments, significantly reduces the volume of wastewater generated, leading to lower environmental compliance costs and faster throughput times. These operational efficiencies translate into a more agile manufacturing process capable of responding quickly to market demands for quinoline-based active pharmaceutical ingredients.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents combined with the reduction in waste treatment requirements drives a substantial decrease in overall production costs. By avoiding the use of strong acids and oxidants, the process removes the need for costly corrosion-resistant equipment and extensive safety monitoring systems, further lowering overhead expenses. The high yield achieved in the primary step minimizes the loss of valuable starting materials, ensuring that the cost per kilogram of the final quinoline intermediate is optimized for commercial viability. Additionally, the simplified purification workflow reduces the consumption of solvents and stationary phases, contributing to a leaner and more cost-effective manufacturing model.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as DMF, triethylamine, and copper salts ensures that the supply chain is robust and less susceptible to disruptions caused by the scarcity of specialized reagents. Since the starting aniline derivatives can be synthesized from widely available bulk chemicals, manufacturers can secure long-term contracts with multiple suppliers to guarantee continuity of supply. The mild reaction conditions also reduce the risk of batch failures due to thermal runaways or equipment malfunctions, ensuring consistent delivery schedules to downstream customers. This reliability is crucial for pharmaceutical companies that require uninterrupted access to high-quality intermediates to maintain their own production timelines.
- Scalability and Environmental Compliance: The one-pot nature of this reaction facilitates easy scale-up from laboratory to pilot and commercial scales without the need for complex process redesigns or additional safety barriers. The reduced generation of acidic waste and the absence of heavy metal oxidants align with increasingly stringent global environmental regulations, minimizing the regulatory burden on manufacturing sites. The ability to recycle the solvent and recover the copper catalyst further enhances the sustainability profile of the process, making it an attractive option for companies committed to green chemistry principles. This scalability ensures that the method can meet the growing demand for quinoline derivatives in the pharmaceutical and agrochemical sectors efficiently.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this quinoline synthesis technology, providing clarity for stakeholders evaluating its potential integration into their supply chains. These answers are derived directly from the experimental data and technical disclosures within patent CN114890944A, ensuring accuracy and relevance for decision-makers. Understanding these details is essential for assessing the feasibility of adopting this method for specific project requirements and for anticipating any potential challenges during technology transfer.
Q: What are the optimal reaction conditions for this quinoline synthesis?
A: According to patent CN114890944A, the optimal conditions involve using cuprous oxide (Cu2O) as the metal additive, triethylamine as the base, and DMF as the solvent at a temperature of 140°C for 18 hours.
Q: Can this method tolerate different substituents on the starting aniline?
A: Yes, the method demonstrates broad substrate scope, successfully converting various o-isopropenylphenylaniline derivatives with alkyl, cycloalkyl, aryl, and halogen-substituted groups into the corresponding quinolines with moderate to high yields.
Q: How does this method compare to the traditional Skraup reaction?
A: Unlike the Skraup reaction which requires harsh strong acids and high temperatures leading to complex separation, this novel method operates under milder conditions with simpler post-treatment involving only column chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the competitive landscape of pharmaceutical intermediate manufacturing. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are equipped with state-of-the-art facilities and rigorous QC labs capable of meeting stringent purity specifications for complex heterocyclic compounds like the quinolines described in this patent. Our commitment to quality and consistency makes us an ideal partner for organizations seeking to secure a stable supply of high-performance chemical building blocks.
We invite you to contact our technical procurement team to discuss how this innovative copper-catalyzed synthesis can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into the potential economic benefits of switching to this greener and more efficient manufacturing process. We are ready to provide specific COA data and route feasibility assessments to support your R&D and supply chain planning efforts, ensuring that your projects proceed with confidence and precision.
