Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Advanced Drug Discovery
Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Advanced Drug Discovery
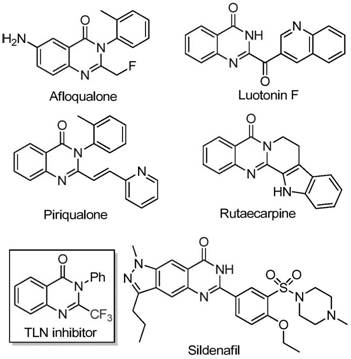
The pharmaceutical industry continuously seeks robust and cost-effective methodologies for constructing nitrogen-containing heterocycles, particularly quinazolinones, which serve as privileged scaffolds in medicinal chemistry. As detailed in the recent patent CN111675662B, a novel preparation method for 2-trifluoromethyl substituted quinazolinone compounds has been disclosed, offering a transformative approach to accessing these bioactive cores. This technology leverages an inexpensive iron catalyst to drive the cyclization of readily available trifluoroethylimidoyl chloride and isatin derivatives, bypassing the limitations of traditional synthetic routes. The introduction of the trifluoromethyl group is strategically critical, as it significantly enhances the electronegativity, metabolic stability, and lipophilicity of the resulting drug candidates, thereby improving their overall bioavailability and therapeutic potential.
The significance of this development extends beyond mere academic interest; it addresses pressing commercial needs for reliable pharmaceutical intermediate supplier networks capable of delivering high-purity building blocks. Quinazolinone derivatives are ubiquitous in modern pharmacopeia, exhibiting a broad spectrum of biological activities including anti-cancer, anticonvulsant, anti-inflammatory, and antifungal properties. By streamlining the synthesis of these fluorinated heterocycles, the patented method facilitates the rapid exploration of chemical space for new drug discovery programs. The ability to efficiently incorporate the trifluoromethyl motif into the quinazolinone core opens new avenues for optimizing lead compounds, ensuring that research and development teams can focus on efficacy rather than struggling with complex synthetic bottlenecks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinones bearing trifluoromethyl functional groups has relied heavily on the cyclization of specific synthons with substrates such as anthranilamide, anthranilic acid, or isatoic anhydride. While these methods are well-documented in literature, they suffer from inherent drawbacks that hinder their utility in large-scale manufacturing. Common trifluoromethylating reagents like trifluoroacetic anhydride or ethyl trifluoroacetate are often expensive and require stringent handling conditions. Furthermore, these conventional pathways are frequently plagued by severe reaction conditions, narrow substrate scope, and unsatisfactory yields, which collectively inflate the cost of goods sold (COGS) for the final active pharmaceutical ingredients. The reliance on precious metal catalysts or harsh reagents also introduces significant challenges in impurity control and environmental compliance, creating friction for procurement managers seeking sustainable supply chains.
The Novel Approach
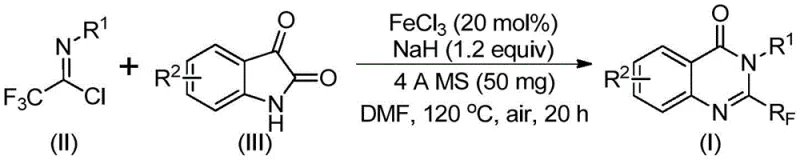
In stark contrast, the methodology outlined in CN111675662B utilizes a synergistic combination of ferric chloride and sodium hydride to catalyze the reaction between trifluoroethylimidoyl chloride and isatin. This innovative route eliminates the need for expensive precious metals, substituting them with abundant and cheap iron salts. The reaction proceeds through a tandem sequence involving initial carbon-nitrogen bond formation followed by iron-catalyzed decarbonylation and cyclization. This mechanistic shift allows for milder reaction conditions and exceptional functional group tolerance, enabling the synthesis of diverse quinazolinone derivatives with substituents at various positions on the aromatic rings. The operational simplicity, combined with the use of commercially available starting materials, positions this method as a superior alternative for cost reduction in API manufacturing, directly addressing the pain points of traditional synthesis.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this technological breakthrough lies in the efficient activation of the substrates by the iron catalyst system. The reaction initiates with the interaction between the trifluoroethylimidoyl chloride and the isatin derivative in the presence of a base, likely forming a transient trifluoroacetamidine intermediate. Subsequently, the ferric chloride catalyst facilitates a decarbonylation event, which is the rate-determining step that drives the cyclization forward to form the stable quinazolinone ring system. This iron-catalyzed pathway is remarkably robust, tolerating electron-withdrawing and electron-donating groups alike, which is crucial for generating diverse libraries of analogs during the lead optimization phase. The use of 4A molecular sieves further enhances the reaction efficiency by sequestering moisture, ensuring that the sensitive imidoyl chloride species remains intact throughout the prolonged heating period.
From an impurity control perspective, this mechanism offers distinct advantages over competing technologies. The high selectivity of the iron catalyst minimizes the formation of side products that typically arise from non-specific acylation or polymerization reactions. The protocol specifies a two-stage temperature profile, starting at 40°C to allow for initial mixing and bond formation, followed by heating to 120°C to drive the cyclization to completion. This controlled thermal ramping prevents the degradation of sensitive functional groups and ensures high conversion rates. For R&D directors focused on purity profiles, the ability to achieve high yields with minimal byproduct formation simplifies downstream processing, reducing the burden on purification teams and ensuring that the final material meets stringent quality specifications required for clinical trials.
How to Synthesize 2-Trifluoromethyl Quinazolinones Efficiently
The practical implementation of this synthesis is straightforward and amenable to standard laboratory equipment, making it an attractive option for process chemists looking to scale up production. The procedure involves charging a reaction vessel with the catalyst system, substrates, and solvent, followed by a defined heating cycle. The use of dimethylformamide (DMF) as the solvent provides excellent solubility for all reactants, ensuring homogeneous reaction conditions that are critical for reproducibility. Detailed standardized synthetic steps see the guide below for precise stoichiometric ratios and workup procedures that guarantee consistent results across different batches.
- Mix ferric chloride (20 mol%), sodium hydride (1.2 equiv), 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin derivative in DMF solvent.
- Stir the reaction mixture at 40°C for approximately 10 hours to initiate the bond formation.
- Heat the mixture to 120°C and maintain for 20 hours under air to complete the decarbonylation and cyclization, followed by purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iron-catalyzed methodology translates into tangible strategic benefits that extend far beyond the laboratory bench. The shift from precious metal catalysts to inexpensive iron salts represents a fundamental change in the cost structure of producing these valuable intermediates. By eliminating the need for costly palladium or rhodium complexes, manufacturers can achieve substantial cost savings on raw materials, which directly improves the margin profile of the final drug product. Furthermore, the reliance on commodity chemicals like isatin and simple aromatic amines ensures a stable and resilient supply chain, mitigating the risks associated with sourcing specialized or scarce reagents that often plague the pharmaceutical industry.
- Cost Reduction in Manufacturing: The replacement of expensive catalysts with ferric chloride drastically lowers the direct material costs associated with the synthesis. Additionally, the reaction operates under air atmosphere rather than requiring rigorous inert gas protection, which simplifies the engineering controls needed for large-scale reactors. This reduction in operational complexity leads to lower energy consumption and reduced capital expenditure on specialized equipment, driving down the overall manufacturing overhead.
- Enhanced Supply Chain Reliability: The starting materials for this process, specifically isatin derivatives and trifluoroethylimidoyl chlorides, are widely available from multiple global suppliers. This abundance reduces the risk of supply disruptions and allows for competitive bidding among vendors. The robustness of the reaction conditions also means that production schedules are less likely to be delayed by sensitivity to environmental factors, ensuring consistent on-time delivery for downstream customers.
- Scalability and Environmental Compliance: The protocol has been demonstrated to be effective on gram scales and is explicitly designed for expansion to industrial levels. The use of DMF, a common industrial solvent, facilitates easy integration into existing manufacturing infrastructure. Moreover, the high atom economy and reduced waste generation associated with this catalytic cycle align with green chemistry principles, helping companies meet increasingly strict environmental regulations and sustainability goals without compromising on output.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of what partners can expect when adopting this route. Understanding these nuances is essential for making informed decisions about process development and vendor selection.
Q: What are the key advantages of using FeCl3 over traditional catalysts for quinazolinone synthesis?
A: The use of ferric chloride (FeCl3) offers a significant cost advantage over precious metal catalysts while maintaining high catalytic efficiency. It enables the use of readily available isatin substrates and tolerates a wide range of functional groups, simplifying the purification process and reducing overall production costs.
Q: Can this synthesis method be scaled for industrial production of API intermediates?
A: Yes, the protocol described in patent CN111675662B is designed for scalability. It utilizes common solvents like DMF and operates under air atmosphere without requiring inert gas protection for the entire duration, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Q: What is the substrate scope for the R1 and R2 positions in this reaction?
A: The reaction demonstrates excellent functional group tolerance. R1 can be various substituted or unsubstituted aryl groups (including methyl, halogen, nitro, methoxy), while R2 on the isatin ring can be alkyl, halogen, or methoxy, allowing for the diverse synthesis of quinazolinone libraries.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug development timelines. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition seamlessly from benchtop discovery to full-scale manufacturing. We are committed to delivering high-purity 2-trifluoromethyl quinazolinones that meet rigorous QC labs standards and stringent purity specifications, providing you with the confidence needed to advance your pipeline. Our state-of-the-art facilities are equipped to handle the specific requirements of iron-catalyzed reactions, guaranteeing consistency and quality in every batch we produce.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our capabilities can optimize your supply chain and reduce your time to market.
