Advanced Three-Step Synthesis of Benzenesulfonamide Anti-Inflammatory Intermediates for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic pathways for novel anti-inflammatory agents, particularly those based on the quinazoline scaffold which has demonstrated significant biological potential. Patent CN108727283B introduces a groundbreaking synthesis method for benzenesulfonamide anti-inflammatory compounds, addressing critical bottlenecks in traditional manufacturing processes. This technology leverages a streamlined three-step reaction sequence that transforms simple, commercially available starting materials into high-value therapeutic intermediates with exceptional efficiency. By integrating a palladium-catalyzed coupling strategy with precise acid-additive control, the method achieves superior yields and purity profiles that are essential for regulatory compliance in drug development. For R&D directors and procurement specialists, this patent represents a viable opportunity to optimize supply chains for next-generation anti-inflammatory drugs, ensuring both technical feasibility and economic viability in a competitive market landscape.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for quinazoline derivatives often rely on harsh reaction conditions that pose significant challenges for industrial scale-up and environmental compliance. Many prior art methods utilize strong oxidizing agents such as DDQ or manganese dioxide to facilitate ring closure, which generates substantial hazardous waste and complicates post-reaction purification processes. Furthermore, conventional metal-catalyzed cyclizations frequently suffer from poor selectivity, leading to complex impurity profiles that require extensive chromatographic separation, thereby driving up production costs and extending lead times. The reliance on expensive transition metals without efficient recovery systems also contributes to unsustainable manufacturing economics, making these routes less attractive for large-volume commercial production. Additionally, the use of unstable intermediates in older methodologies can result in inconsistent batch-to-batch quality, creating supply chain risks for pharmaceutical manufacturers who require stringent quality control standards.
The Novel Approach
In contrast, the methodology disclosed in CN108727283B offers a refined and economically superior alternative that eliminates the need for hazardous oxidizers while maintaining high reaction efficiency. This novel approach utilizes a palladium-catalyzed coupling reaction under mild conditions, significantly reducing energy consumption and operational hazards associated with high-temperature or high-pressure processes. The strategic selection of organic ligands and acidic additives ensures high regioselectivity, minimizing the formation of unwanted byproducts and simplifying the downstream purification workflow. By employing readily available raw materials such as 2-aminobenzonitrile and formic acid derivatives, the process enhances supply chain resilience and reduces dependency on specialized reagents. This streamlined three-step sequence not only improves overall yield but also aligns with green chemistry principles, making it an ideal candidate for sustainable commercial manufacturing of complex pharmaceutical intermediates.
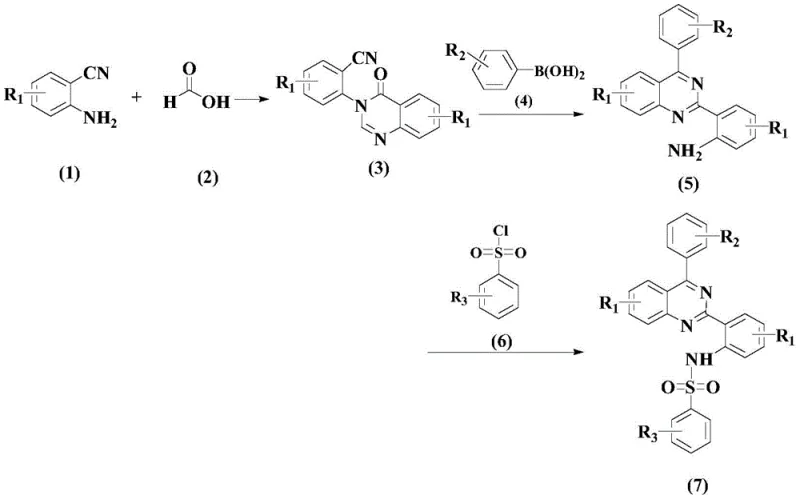
Mechanistic Insights into Pd-Catalyzed Coupling and Cyclization
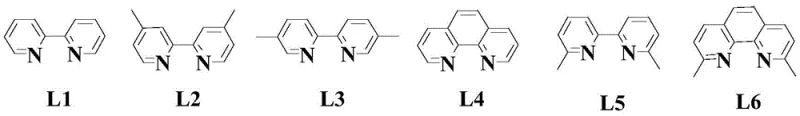
The core innovation of this synthesis lies in the second step, where a palladium-catalyzed coupling reaction constructs the critical carbon-nitrogen bonds necessary for the quinazoline framework. The mechanism involves the oxidative addition of the palladium catalyst to the aryl halide substrate, followed by transmetallation with the amine component and subsequent reductive elimination to form the coupled product. The presence of a specific bipyridine-type ligand, such as L1, stabilizes the palladium center and facilitates the catalytic cycle, ensuring high turnover numbers and consistent reaction performance. This ligand design prevents catalyst deactivation and promotes the desired reaction pathway over competing side reactions, which is crucial for maintaining high purity levels in the final intermediate. The precise tuning of the electronic properties of the ligand allows for optimal reactivity under mild thermal conditions, reducing the risk of thermal degradation of sensitive functional groups.
Impurity control is further enhanced by the inclusion of an acidic additive, specifically p-toluenesulfonic acid monohydrate, which plays a vital role in protonating intermediates and driving the reaction equilibrium towards product formation. This acid additive helps to suppress the formation of basic byproducts that could otherwise coordinate with the catalyst and inhibit its activity. By maintaining an acidic environment, the reaction system ensures that the amine nucleophile remains sufficiently reactive while preventing the hydrolysis of sensitive nitrile or amide groups. The combination of the palladium catalyst, specialized ligand, and acidic promoter creates a synergistic effect that maximizes yield and minimizes impurity generation. This level of mechanistic control is essential for producing pharmaceutical intermediates that meet the rigorous purity specifications required for clinical trials and commercial drug production.
How to Synthesize Benzenesulfonamide Efficiently
The synthesis of the target benzenesulfonamide compound is achieved through a logical three-step sequence that balances reaction efficiency with operational simplicity. The process begins with the cyclization of 2-aminobenzonitrile to form the quinazolinone core, followed by a palladium-catalyzed arylation to introduce the necessary substituents. The final step involves the reaction of the amine intermediate with benzenesulfonyl chloride to install the sulfonamide moiety, which is critical for the compound's anti-inflammatory activity. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- React 2-aminobenzonitrile with formic acid derivatives to form the quinazolinone intermediate via cyclization.
- Perform a palladium-catalyzed coupling reaction using specific organic ligands and acidic additives to introduce aryl groups.
- Complete the synthesis by reacting the amine intermediate with benzenesulfonyl chloride to form the final sulfonamide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthesis route offers substantial strategic benefits that extend beyond mere technical performance. The elimination of hazardous oxidizers and the use of common organic solvents like toluene significantly reduce waste disposal costs and regulatory burdens associated with environmental compliance. The high yields observed in each step of the reaction sequence translate directly into lower raw material consumption per unit of product, driving down the overall cost of goods sold. Furthermore, the robustness of the catalytic system ensures consistent batch quality, reducing the risk of production delays caused by failed batches or out-of-specification results. This reliability is crucial for maintaining continuous supply to downstream drug manufacturers who operate on tight production schedules.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing inexpensive and readily available starting materials such as 2-aminobenzonitrile and formic acid derivatives. The high efficiency of the palladium-catalyzed step minimizes the loss of valuable intermediates, ensuring that the maximum amount of raw material is converted into the final product. By avoiding the use of expensive stoichiometric oxidizers and reducing the need for complex purification steps, the overall manufacturing expense is drastically lowered. This economic advantage allows for more competitive pricing strategies in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals rather than specialized reagents enhances the resilience of the supply chain against market fluctuations and shortages. The simplicity of the reaction conditions means that the process can be easily transferred between different manufacturing sites without significant re-validation efforts. This flexibility ensures that production can be scaled up or shifted geographically to mitigate risks associated with logistics or regional disruptions. Consistent product quality further strengthens relationships with downstream partners who depend on reliable delivery of high-purity intermediates.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of hazardous waste streams make this process highly scalable from laboratory to commercial production volumes. The use of standard equipment and solvents facilitates easy integration into existing manufacturing infrastructure without the need for costly capital investments. Additionally, the reduced environmental footprint aligns with increasingly stringent global regulations on chemical manufacturing, ensuring long-term operational sustainability. This compliance reduces the risk of regulatory penalties and enhances the corporate reputation of manufacturers adopting this green chemistry approach.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation to provide accurate guidance for potential adopters. Understanding these details is essential for evaluating the feasibility of integrating this route into your existing production portfolio.
Q: What are the critical reaction conditions for the Pd-catalyzed step?
A: The process requires a palladium catalyst such as palladium acetate, a specific bipyridine-type ligand (L1), and an acidic additive like p-toluenesulfonic acid monohydrate in toluene solvent at 70-90°C.
Q: How does this method improve impurity control compared to conventional routes?
A: By utilizing a specific acidic additive and ligand system, the reaction minimizes side reactions and byproduct formation, leading to higher purity intermediates that require less rigorous downstream purification.
Q: Is this synthesis route suitable for large-scale manufacturing?
A: Yes, the method uses readily available raw materials and avoids hazardous oxidizers, making it safer and more cost-effective for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzenesulfonamide Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and contract development, offering unparalleled expertise in scaling complex pharmaceutical pathways from 100 kgs to 100 MT/annual commercial production. Our technical team possesses extensive experience in optimizing palladium-catalyzed reactions and managing stringent purity specifications to meet the rigorous demands of global regulatory agencies. With state-of-the-art rigorous QC labs and a commitment to quality, we ensure that every batch of benzenesulfonamide intermediate delivered meets the highest standards of safety and efficacy. We are dedicated to supporting your drug development journey with reliable supply and technical excellence.
We invite you to collaborate with our technical procurement team to explore how this innovative synthesis route can enhance your supply chain efficiency. Request a Customized Cost-Saving Analysis to understand the specific economic benefits for your project. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your unique requirements. Contact us today to initiate a partnership that drives value and innovation in your pharmaceutical manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
