Scalable Synthesis of Quinoxaline-1,4-Dioxide Derivatives for Commercial Veterinary Applications
The pharmaceutical and agrochemical industries are constantly seeking robust synthetic pathways for high-value heterocyclic compounds, particularly those serving as critical intermediates for veterinary growth promoters. Patent CN101016268B introduces a refined chemical synthesis method for 2-formylcyanoacetylhydrazone-quinoxaline-1,4-dioxide, a pivotal structure in the development of quinocetone-related feed additives. This technology addresses long-standing challenges in the manufacturing of quinoxaline-1,4-dioxide derivatives by optimizing reaction conditions and reagent selection to enhance overall yield and operational safety. Unlike earlier methodologies that relied on hazardous reagents or complex multi-step protections, this approach streamlines the construction of the quinoxaline core through a direct condensation strategy. For R&D directors and procurement specialists, understanding the nuances of this patent is essential for securing a reliable veterinary intermediate supplier capable of delivering consistent quality. The process leverages the Beirut condensation reaction, a powerful tool for constructing nitrogen-rich heterocycles, followed by a selective oxidation and final hydrazone formation. By mastering this specific sequence, manufacturers can achieve significant improvements in process efficiency while maintaining stringent purity specifications required for animal health applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinoxaline-N,N-dioxides and their subsequent functionalization has been plagued by inefficiencies that hinder cost reduction in veterinary drug manufacturing. Prior art, such as United States Patent 3660398, disclosed methods utilizing benzofurazan but often resulted in complex mixtures requiring extensive purification protocols that drove up operational expenses. Traditional routes frequently employed harsh oxidizing agents or expensive transition metal catalysts that not only increased the raw material costs but also introduced heavy metal contamination risks, necessitating additional downstream removal steps. Furthermore, many conventional processes operated under extreme temperatures or pressures, posing safety hazards and limiting the feasibility of commercial scale-up of complex quinoxaline derivatives. The reliance on unstable intermediates in older methods often led to inconsistent batch-to-batch yields, creating supply chain volatility for downstream formulators. These technical bottlenecks meant that producing high-purity quinoxaline intermediates was often economically prohibitive for all but the largest chemical enterprises, restricting market availability.
The Novel Approach
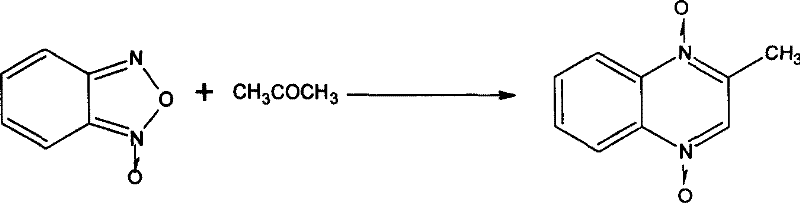
The methodology outlined in CN101016268B represents a strategic evolution in synthetic design, specifically targeting the economic and practical limitations of previous techniques. By utilizing acetone and benzofurazan in the presence of accessible amine catalysts like pyrrolidine or diethylamine, the new route establishes the quinoxaline backbone under significantly milder thermal conditions. This shift eliminates the need for exotic reagents, thereby drastically simplifying the supply chain requirements and reducing lead time for high-purity quinoxaline intermediates. The subsequent oxidation step employs manganese dioxide or chromium trioxide in a dimethyl sulfoxide and alcohol solvent system, which offers superior control over the formylation process compared to older Vilsmeier-Haack type reactions. This precision minimizes the formation of over-oxidized byproducts, directly enhancing the crude purity before the final crystallization. Ultimately, this novel approach transforms a previously cumbersome synthesis into a streamlined, industrially viable process that aligns with modern green chemistry principles and cost-efficiency goals.
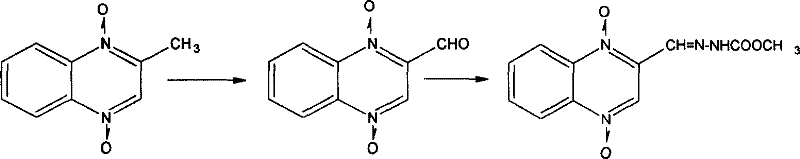
Mechanistic Insights into Beirut Condensation and Oxidative Formylation
To fully appreciate the technical robustness of this synthesis, one must examine the mechanistic underpinnings of the initial ring-closing step. The reaction between benzofurazan and acetone, catalyzed by a secondary amine, proceeds through a nucleophilic attack mechanism that facilitates the opening of the furazan ring and subsequent cyclization. The amine catalyst activates the acetone enolate, allowing it to attack the electron-deficient carbon of the benzofurazan oxide, initiating the rearrangement that forms the N,N-1,4-dioxy-2-methyl quinoxaline core. This step is critical because the stability of the N-oxide bonds formed here dictates the reactivity in subsequent functionalization steps. Controlling the temperature between 45°C and 75°C is vital to prevent the decomposition of the N-oxide moiety while ensuring complete conversion of the starting materials. The choice of solvent and catalyst loading directly influences the kinetics of this transformation, allowing chemists to fine-tune the reaction rate to match reactor capabilities.
Following the formation of the methyl intermediate, the oxidative conversion to the aldehyde is a masterpiece of selective functional group manipulation. The use of oxidants like manganese dioxide in a polar aprotic solvent environment ensures that the methyl group is oxidized to an aldehyde without compromising the sensitive N-oxide groups on the quinoxaline ring. This selectivity is paramount for maintaining the biological activity profile of the final veterinary compound. The mechanism likely involves the formation of a radical intermediate or a metal-alkoxide complex that abstracts hydrogen atoms from the methyl group, progressively introducing oxygen functionality. Once the aldehyde is generated, the final condensation with cyanoacetohydrazide occurs through a nucleophilic addition-elimination sequence. Adjusting the pH of the reaction liquid to a range of 4 to 8 promotes the dehydration step required to form the stable hydrazone linkage, precipitating the product out of the solution. This pH-controlled precipitation acts as an inherent purification step, excluding soluble impurities and ensuring the structural integrity of the final API intermediate.
How to Synthesize 2-Formylcyanoacetylhydrazone-Quinoxaline-1,4-Dioxide Efficiently
Implementing this synthesis requires careful attention to reagent stoichiometry and thermal management to maximize yield and safety. The process begins with the preparation of the reaction vessel, ensuring it is equipped for reflux and controlled addition of catalysts. Operators must monitor the exothermic nature of the initial condensation to maintain the optimal temperature window, preventing runaway reactions that could degrade the sensitive N-oxide structures. Following the isolation of the methyl intermediate, the oxidation step demands anhydrous conditions to prevent hydrolysis of the oxidant, ensuring efficient conversion to the aldehyde. The final coupling reaction should be performed with slow addition of the hydrazide component to control crystal growth and particle size distribution, which impacts downstream filtration efficiency. For detailed standard operating procedures and specific safety guidelines regarding reagent handling, please refer to the technical documentation provided below.
- Condense benzofurazan with acetone using a amine catalyst like pyrrolidine to form N,N-1,4-dioxy-2-methyl quinoxaline.
- Oxidize the methyl intermediate using manganese dioxide or chromium trioxide in a DMSO-alcohol solvent system to generate the aldehyde.
- React the resulting aldehyde with cyanoacetohydrazide under controlled pH and temperature to precipitate the final hydrazone product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented synthesis route offers substantial value propositions for procurement managers and supply chain heads looking to optimize their sourcing strategies for veterinary intermediates. The elimination of expensive noble metal catalysts and the use of commodity chemicals like acetone and ethanol significantly lower the raw material cost base, translating into more competitive pricing for the final active ingredient. This cost structure allows for greater margin flexibility in volatile markets, ensuring long-term supply continuity even during periods of raw material price fluctuation. Furthermore, the mild reaction conditions reduce energy consumption and equipment wear, contributing to overall operational expenditure savings. The robustness of the process means that manufacturing can be scaled up with minimal risk of batch failure, providing reliability that is crucial for meeting the rigorous delivery schedules of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The substitution of complex, high-cost reagents with readily available amines and common oxidants removes significant financial barriers to production. By avoiding the need for specialized heavy metal removal resins or extensive chromatographic purification, the downstream processing costs are drastically simplified. This efficiency gain allows manufacturers to offer high-purity veterinary intermediates at a price point that supports the economic viability of large-scale animal husbandry operations. The streamlined workflow reduces labor hours per kilogram of product, further enhancing the cost-effectiveness of the entire manufacturing campaign.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this process is straightforward, as benzofurazan, acetone, and cyanoacetohydrazide are produced by multiple global suppliers, mitigating the risk of single-source dependency. This diversity in the supply base ensures that production schedules are not held hostage by the availability of niche chemicals. Additionally, the stability of the intermediates allows for potential storage and inventory management strategies that buffer against short-term market disruptions. For supply chain planners, this means a more predictable lead time and the ability to secure long-term contracts with confidence in the manufacturer's ability to deliver.
- Scalability and Environmental Compliance: The process operates at atmospheric pressure and moderate temperatures, making it compatible with standard glass-lined or stainless steel reactors found in most multipurpose chemical plants. This compatibility accelerates the technology transfer from lab to plant, reducing the time to market for new products. Moreover, the reduced use of hazardous solvents and the generation of less toxic waste streams align with increasingly strict environmental regulations. This compliance reduces the burden of waste disposal and permitting, facilitating smoother operations in regions with rigorous environmental oversight.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this quinoxaline derivative. These answers are derived directly from the experimental data and claims presented in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these details helps stakeholders make informed decisions about integrating this intermediate into their own formulation or synthesis pipelines. The responses cover aspects ranging from reaction scalability to impurity profiles, providing a comprehensive overview of the technology's capabilities.
Q: What are the primary advantages of this synthesis route over traditional methods?
A: This method utilizes readily available raw materials like benzofurazan and acetone, avoiding expensive transition metal catalysts often required in older patents, which significantly lowers production costs and simplifies purification.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the reaction conditions are mild, operating between 45°C and 85°C, and utilize common solvents like DMSO and ethanol, making it highly adaptable for commercial scale-up without requiring specialized high-pressure equipment.
Q: How is product purity ensured in this synthetic pathway?
A: The process includes specific pH adjustment steps during the final condensation phase to facilitate precise precipitation of the target compound, effectively separating impurities and ensuring high structural integrity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Formylcyanoacetylhydrazone-Quinoxaline-1,4-Dioxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of effective veterinary medicines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We adhere to stringent purity specifications and operate rigorous QC labs to verify that every batch of 2-formylcyanoacetylhydrazone-quinoxaline-1,4-dioxide meets the highest industry standards. Our commitment to technical excellence means we can adapt the patented synthesis to fit your specific volume requirements while maintaining the cost and quality advantages inherent in the process.
We invite you to collaborate with us to explore how this advanced synthesis route can benefit your product portfolio. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your current manufacturing setup. We are ready to provide specific COA data and route feasibility assessments to demonstrate how partnering with us can drive efficiency and reliability in your supply chain. Let us be your trusted partner in bringing innovative veterinary solutions to the global market.
