Advanced Double-Oxidation Strategy for High-Purity Tazobactam Manufacturing
Advanced Double-Oxidation Strategy for High-Purity Tazobactam Manufacturing
The pharmaceutical industry constantly seeks robust synthetic routes for beta-lactamase inhibitors, and patent CN109305977B presents a transformative approach to Tazobactam production. This technology addresses critical stability issues inherent in traditional penicillanic acid derivatives by introducing a strategic double-oxidation sequence prior to heterocycle installation. By converting the sulfur atom into a sulfone early in the synthesis, the process fundamentally alters the electronic environment of the molecule, stabilizing the reactive chloromethyl group against premature elimination. This innovation not only mitigates the safety risks associated with high-energy intermediates but also streamlines the purification workflow, offering a compelling value proposition for manufacturers aiming to secure a reliable API intermediate supplier for broad-spectrum antibiotic formulations.
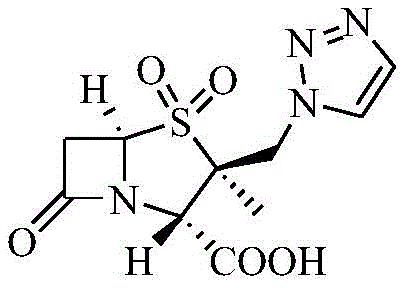
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Tazobactam has been plagued by significant operational hazards and yield inconsistencies, particularly in routes relying on azide chemistry. Traditional methods often involve the use of sodium azide and acetylene, reagents known for their high toxicity and explosive potential, which necessitate expensive containment infrastructure and rigorous safety protocols. Furthermore, the conventional 'upper triazole' processes frequently suffer from the instability of the 2β-chloromethyl group; the lone pair electrons on the sulfur atom in the sulfoxide state can facilitate the departure of the chlorine ion, leading to side reactions and degraded product quality. These factors collectively result in prolonged reaction times, complex waste treatment requirements, and unpredictable batch-to-batch consistency, creating substantial bottlenecks for cost reduction in pharmaceutical manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in CN109305977B circumvents these pitfalls by prioritizing the stabilization of the penicillanic core before functionalization. The novel route employs a double-oxidation step using potassium permanganate and acidic media to generate the 1,1-dioxide derivative immediately. This sulfone formation exerts a strong electron-withdrawing inductive effect, effectively locking the chlorine atom in place and preventing unwanted elimination. Subsequent substitution with triazole is then facilitated by a specialized catalytic system involving crown ethers and potassium iodide, which accelerates the reaction kinetics without requiring extreme temperatures. This strategic reordering of synthetic steps eliminates the need for hazardous azide reagents entirely, simplifying the regulatory compliance landscape and enhancing the overall safety profile of the production facility.
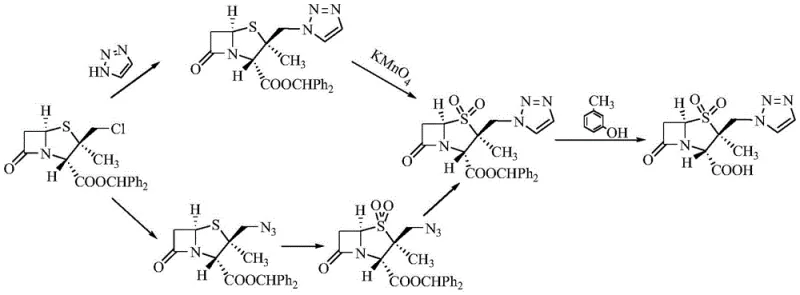
Mechanistic Insights into Double-Oxidation and Phase Transfer Catalysis
The core chemical breakthrough of this patent lies in the precise manipulation of the sulfur oxidation state to control reactivity. In the initial oxidation phase, the use of a mixed solvent system containing glacial acetic acid and concentrated sulfuric acid ensures the complete conversion of the sulfide or sulfoxide to the sulfone (compound 2). Mechanistically, the sulfone group (-SO2-) is a powerful electron-withdrawing group that reduces the electron density on the adjacent carbon bearing the chlorine atom. This reduction in electron density significantly increases the activation energy required for the chlorine to leave as a chloride ion, thereby preserving the integrity of the chloromethyl side chain during the subsequent nucleophilic attack. This stability is crucial for maintaining high stereochemical fidelity and preventing the formation of des-chloro impurities that are difficult to remove downstream.
Following oxidation, the introduction of the triazole ring is achieved through a highly efficient phase transfer catalysis (PTC) mechanism. The addition of 18-crown-6 ether complexes with potassium cations from the potassium iodide catalyst, solubilizing the iodide anion in the organic phase and enhancing its nucleophilicity. This catalytic cycle facilitates the displacement of the stabilized chlorine atom by the triazole nitrogen, proceeding smoothly under reflux conditions. The synergy between the electron-deficient sulfone substrate and the activated nucleophile allows the reaction to reach completion in significantly shorter timeframes compared to non-catalyzed variants. This mechanistic elegance ensures that the final crude product possesses a superior impurity profile, reducing the burden on downstream purification units and maximizing the recovery of high-value active pharmaceutical ingredients.
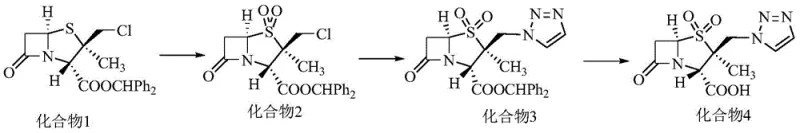
How to Synthesize Tazobactam Efficiently
The implementation of this synthesis route requires careful control of reaction parameters to maximize the benefits of the double-oxidation strategy. The process begins with the preparation of the stable dioxide intermediate, followed by the catalytic triazole coupling, and concludes with a mild deprotection step using m-cresol. Each stage is designed to minimize thermal stress on the beta-lactam ring while ensuring quantitative conversion of intermediates. Operators must adhere to strict temperature controls during the exothermic oxidation phase and utilize the specified solvent ratios to maintain optimal phase transfer efficiency. For a comprehensive breakdown of the specific reagent quantities, temperature profiles, and workup procedures required to replicate this high-yield process, please refer to the standardized technical guide below.
- Perform double oxidation on 2β-chloromethyl penicillanic acid diphenylmethyl ester using potassium permanganate, glacial acetic acid, and concentrated sulfuric acid to form the 1,1-dioxide intermediate.
- React the dioxide intermediate with triazole in the presence of crown ether (18-crown-6) and potassium iodide catalyst to introduce the triazole ring.
- Execute deprotection using m-cresol followed by refining in ethanol to obtain high-purity Tazobactam (>99%).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers tangible strategic benefits beyond mere technical feasibility. By eliminating the reliance on hazardous azide reagents, facilities can drastically reduce the costs associated with specialized safety equipment, hazardous waste disposal, and regulatory reporting. The enhanced stability of the chloromethyl intermediate translates directly into higher process yields and reduced raw material consumption, as fewer batches are lost to decomposition or side reactions. Furthermore, the shortened reaction times and simplified workup procedures enable faster throughput, allowing manufacturing plants to respond more agilely to market demand fluctuations without compromising on product quality or compliance standards.
- Cost Reduction in Manufacturing: The removal of expensive and dangerous reagents like sodium azide significantly lowers the direct material costs and the indirect costs of safety management. Additionally, the improved yield resulting from the stabilized intermediate means that less starting material is required to produce the same amount of final API, driving down the cost of goods sold (COGS) substantially. The simplified purification process also reduces solvent usage and energy consumption during crystallization and drying phases.
- Enhanced Supply Chain Reliability: Sourcing hazardous reagents often involves complex logistics and long lead times due to strict transportation regulations. By shifting to a chemistry based on common oxidants and triazoles, the supply chain becomes more resilient and less prone to disruptions. The robustness of the intermediate also allows for potential storage or transport between steps if necessary, providing greater flexibility in production scheduling and inventory management for global operations.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scale-up, utilizing standard reactor materials and avoiding high-pressure or cryogenic conditions. The reduction in toxic waste streams aligns with increasingly stringent environmental regulations, minimizing the risk of fines and shutdowns. This green chemistry approach not only future-proofs the manufacturing asset but also enhances the corporate sustainability profile, which is increasingly important for partnerships with major pharmaceutical innovators.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this double-oxidation synthesis route. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on why this method outperforms legacy technologies. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for procurement specialists assessing the long-term viability of this supply source.
Q: Why is double oxidation preferred over single oxidation in Tazobactam synthesis?
A: Double oxidation converts the sulfur atom to a sulfone, which strongly withdraws electrons. This stabilizes the adjacent chloromethyl group, preventing premature chlorine loss and significantly improving reaction yield compared to sulfoxide intermediates.
Q: What role does the crown ether play in the triazole substitution step?
A: Crown ether acts as a phase transfer catalyst, facilitating the interaction between the organic substrate and the triazole nucleophile in the biphasic solvent system, thereby shortening reaction time and improving conversion rates.
Q: Does this method avoid hazardous reagents like sodium azide?
A: Yes, unlike traditional 'upper azide' routes that require explosive sodium azide and acetylene, this patent utilizes a direct condensation method with triazole, significantly enhancing operational safety for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tazobactam Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN109305977B are fully realized in a commercial setting. We operate stringent purity specifications and maintain rigorous QC labs equipped to analyze complex beta-lactam impurity profiles, guaranteeing that every batch of Tazobactam intermediate meets the highest global pharmacopoeia standards. Our commitment to quality assurance ensures that your downstream formulation processes remain uninterrupted and compliant.
We invite you to collaborate with us to leverage this innovative chemistry for your antibiotic portfolio. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing platform can become a cornerstone of your supply chain strategy for high-purity pharmaceutical intermediates.
