Advanced Electrochemical Route for Tazobactam Intermediate Manufacturing and Scale-Up
The pharmaceutical industry continuously seeks more efficient pathways for producing critical beta-lactamase inhibitors, and patent CN113073348B presents a transformative solution for the synthesis of tazobactam key intermediates. This specific intellectual property details a novel electrochemical anodic oxidation method that directly couples disulfide ring-opening compounds with 1,2,3-triazoles, bypassing the cumbersome multi-step sequences traditionally required. For R&D directors and process chemists, this represents a significant leap forward in step economy, offering a route that operates under mild conditions (24-28°C) while utilizing electricity as a clean reagent. The technology addresses the longstanding challenges of low selectivity and poor economic efficiency inherent in previous synthetic strategies, positioning it as a highly attractive candidate for modern API manufacturing pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of diphenylmethyl 2β-triazolylmethylpenicillate has been plagued by inefficient multi-step protocols that rely heavily on hazardous halogenation and expensive metal catalysts. As illustrated in the comparative reaction schemes below, traditional routes often necessitate a preliminary halogenation step to form an intermediate halide, followed by a nucleophilic substitution with triazole, frequently requiring cationic resins or stoichiometric amounts of silver salts to proceed. 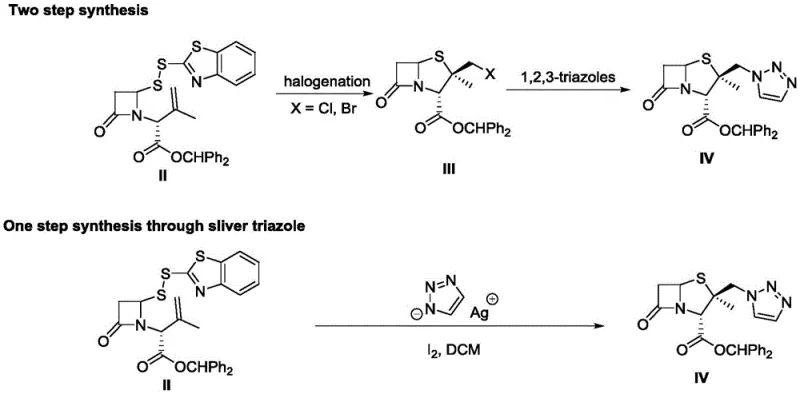 These conventional approaches suffer from significant drawbacks, including poor regioselectivity between five-membered and six-membered ring isomers during halogenation, which complicates purification and drastically reduces overall yield. Furthermore, the reliance on equivalent amounts of silver salts for triazole activation introduces substantial cost burdens and environmental waste, creating a bottleneck for procurement managers aiming to reduce the cost of goods sold (COGS) in high-volume production scenarios.
These conventional approaches suffer from significant drawbacks, including poor regioselectivity between five-membered and six-membered ring isomers during halogenation, which complicates purification and drastically reduces overall yield. Furthermore, the reliance on equivalent amounts of silver salts for triazole activation introduces substantial cost burdens and environmental waste, creating a bottleneck for procurement managers aiming to reduce the cost of goods sold (COGS) in high-volume production scenarios.
The Novel Approach
In stark contrast, the electrochemical method disclosed in the patent utilizes a direct oxidative coupling strategy that merges the disulfide ring-opening substrate and the triazole in a single operational step. 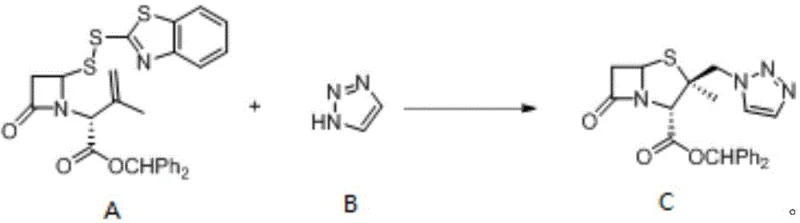 By employing electrochemical anodic oxidation, the process generates active iodine species in situ from inexpensive iodide electrolytes, which serve as efficient electron transfer mediators to drive the cyclization and coupling simultaneously. This innovative approach effectively eliminates the need for pre-functionalization of the triazole and avoids the use of toxic halogenating agents, resulting in a cleaner reaction profile with higher atom economy. For supply chain heads, this simplification translates to a more robust process with fewer unit operations, reduced solvent consumption, and a significantly lower environmental footprint compared to the legacy chemical oxidation methods.
By employing electrochemical anodic oxidation, the process generates active iodine species in situ from inexpensive iodide electrolytes, which serve as efficient electron transfer mediators to drive the cyclization and coupling simultaneously. This innovative approach effectively eliminates the need for pre-functionalization of the triazole and avoids the use of toxic halogenating agents, resulting in a cleaner reaction profile with higher atom economy. For supply chain heads, this simplification translates to a more robust process with fewer unit operations, reduced solvent consumption, and a significantly lower environmental footprint compared to the legacy chemical oxidation methods.
Mechanistic Insights into Electrochemical Anodic Oxidation
The core of this technological breakthrough lies in the precise control of electron transfer at the electrode surface, which facilitates the generation of high-valence iodine intermediates essential for the reaction progression. In this catalytic cycle, iodide ions present in the electrolyte, such as tetrabutylammonium iodide, are oxidized at the anode to form reactive iodine species that act as both oxidants and phase transfer catalysts. These active species interact with the disulfide bond of the penicillanic acid derivative, promoting the formation of a sulfonium ion intermediate that is highly susceptible to nucleophilic attack by the triazole nitrogen. This mechanism ensures high regioselectivity for the desired 2β-position, effectively suppressing the formation of unwanted isomers that typically plague thermal or chemical oxidation methods. The use of a divided or undivided cell allows for fine-tuning of the current density (optimized between 4-30 mA/cm²), providing chemists with a powerful handle to control reaction kinetics and minimize side reactions.
From an impurity control perspective, the electrochemical environment offers a distinct advantage by avoiding the harsh acidic or basic conditions often required in traditional substitution reactions. The mild reaction temperature of 24-28°C prevents the thermal degradation of the sensitive beta-lactam ring, which is a common failure mode in conventional synthesis. Additionally, the continuous regeneration of the iodide mediator at the cathode or through the redox cycle ensures that the concentration of active oxidizing species remains steady, preventing over-oxidation of the product. This stability is crucial for maintaining high purity specifications, as it reduces the formation of polymeric byproducts and sulfone over-oxidation impurities, thereby simplifying the downstream purification workload for quality control teams.
How to Synthesize Diphenylmethyl 2β-Triazolylmethylpenicillate Efficiently
The practical implementation of this electrochemical synthesis is straightforward and adaptable to both batch and continuous flow processing, making it highly suitable for pilot and commercial scale operations. The standard protocol involves dissolving the disulfide ring-opening compound and 1,2,3-triazole in a mixed solvent system of acetonitrile and acetic acid, supplemented with a supporting electrolyte like tetrabutylammonium iodide. The reaction is conducted in an undivided cell equipped with inert electrodes, such as graphite or platinum, under a constant current mode until the theoretical amount of electricity (approximately 5F) is passed. Detailed standardized synthetic steps and specific parameter optimizations for different scales are provided in the guide below.
- Mix disulfide ring-opening compound A and 1,2,3-triazole B with tetrabutylammonium iodide electrolyte in a solvent mixture of acetonitrile and acetic acid.
- Perform electrochemical anodic oxidation in an undivided cell using graphite and platinum/nickel electrodes at a constant current density of 4-30 mA/cm².
- Upon completion of electrolysis (approx. 5F), concentrate the reaction mixture and purify the crude product via extraction and column chromatography to obtain the target intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this electrochemical technology offers compelling economic and logistical benefits that directly impact the bottom line. By replacing expensive stoichiometric reagents like silver salts with catalytic amounts of inexpensive iodides and electricity, the raw material costs are significantly reduced, enhancing the overall margin potential for the final API. The elimination of complex halogenation steps also reduces the dependency on hazardous chemicals, simplifying regulatory compliance and waste disposal procedures, which are often hidden cost drivers in pharmaceutical manufacturing. Furthermore, the streamlined nature of the process shortens the production cycle time, allowing for faster turnaround on orders and improved responsiveness to market demand fluctuations.
- Cost Reduction in Manufacturing: The most immediate financial impact comes from the complete removal of silver salts, which are not only costly but also difficult to recover and recycle efficiently in a GMP environment. By utilizing electricity as the primary oxidant and cheap iodide salts as mediators, the variable cost per kilogram of the intermediate is drastically lowered. Additionally, the one-pot nature of the reaction reduces solvent usage and energy consumption associated with heating and cooling multiple reaction vessels, leading to substantial operational expenditure savings over the lifecycle of the product.
- Enhanced Supply Chain Reliability: Traditional synthesis routes often face bottlenecks due to the limited availability or long lead times of specialized reagents like cationic resins or high-purity silver compounds. This electrochemical method relies on commodity chemicals and standard electrical equipment, ensuring a stable and resilient supply chain that is less vulnerable to geopolitical or market disruptions. The ability to source raw materials locally and the simplified inventory requirements contribute to a more agile manufacturing operation capable of sustaining continuous production schedules without interruption.
- Scalability and Environmental Compliance: The patent data explicitly demonstrates the successful scale-up of this reaction using flow cell apparatuses, achieving high yields even at the 30g scale and beyond, which indicates excellent potential for tonnage production.
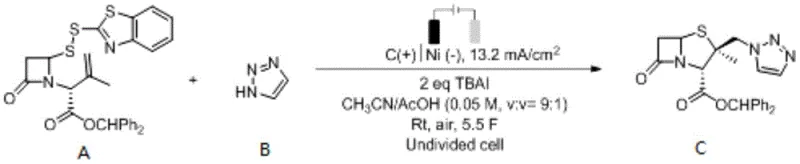 Flow chemistry enhances mass and heat transfer, allowing for safer handling of the electrochemical process and easier integration into existing continuous manufacturing lines. Moreover, the reduction in chemical waste and the absence of heavy metal contaminants align perfectly with green chemistry principles, facilitating easier environmental permitting and reducing the burden on wastewater treatment facilities.
Flow chemistry enhances mass and heat transfer, allowing for safer handling of the electrochemical process and easier integration into existing continuous manufacturing lines. Moreover, the reduction in chemical waste and the absence of heavy metal contaminants align perfectly with green chemistry principles, facilitating easier environmental permitting and reducing the burden on wastewater treatment facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical synthesis route, based on the detailed experimental data provided in the patent documentation. These insights are designed to clarify the operational feasibility and strategic value of adopting this technology for your specific manufacturing needs.
Q: How does this electrochemical method improve upon traditional silver salt methods?
A: Traditional methods require expensive silver salts for triazole pre-functionalization, which creates economic inefficiency and recovery difficulties. This electrochemical approach eliminates the need for silver entirely, using inexpensive iodide electrolytes as electron transfer media to drive the reaction directly.
Q: What yields can be expected from this electrochemical synthesis route?
A: According to the patent data, the method achieves high conversion rates with isolated yields reaching up to 76% in batch processes and maintaining efficiency in flow chemistry setups, significantly outperforming the low conversion rates often seen in conventional halogenation-substitution sequences.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the patent explicitly demonstrates scalability using flow cell apparatuses for 30g-scale preparations, indicating that the technology is robust enough for commercial scale-up of complex pharmaceutical intermediates without the safety risks associated with traditional chemical oxidants.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tazobactam Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes for high-value pharmaceutical intermediates like the tazobactam precursor described in CN113073348B. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can seamlessly translate this innovative electrochemical laboratory method into a robust industrial process. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch against the highest industry standards.
We invite you to collaborate with us to leverage this cutting-edge technology for your supply chain. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can optimize your production costs and secure your supply of high-purity pharmaceutical intermediates.
