Scalable Production of High-Purity Tazobactam Intermediates via Novel Boc-Protection Strategy
Introduction to Next-Generation Tazobactam Intermediate Synthesis
The pharmaceutical industry continuously seeks robust synthetic pathways for critical beta-lactamase inhibitors, and the recent disclosure in patent CN109678886B represents a significant leap forward in the preparation of tazobactam intermediates. This technology addresses long-standing challenges in the synthesis of penicillanic acid sulfone derivatives, which are pivotal precursors for combination antibiotics widely used in clinical settings. By introducing a strategic amino protecting group, specifically the tert-butyloxycarbonyl (Boc) group, onto the 6-aminopenicillanic acid (6-APA) scaffold, the inventors have unlocked a pathway that dramatically enhances stability and overall process efficiency. This innovation is particularly relevant for global supply chains aiming to secure reliable sources of high-purity pharmaceutical intermediates, as it mitigates the risks associated with unstable reaction intermediates and low-yielding oxidation steps that have plagued previous generations of synthetic methods.
The core breakthrough lies in the manipulation of solubility and chemical stability during the critical oxidation phase. Traditional methods often struggle with the high water solubility of 6-APA derivatives, leading to difficult isolation and purification steps that erode yield. In contrast, the protected intermediate generated in this novel process exhibits poor water solubility but exceptional chemical stability, allowing the oxidation reaction to proceed directly in the same pot without the need for intermediate concentration or drying. This telescoping of steps not only streamlines the workflow but also minimizes material loss, resulting in a combined yield for the protection and oxidation steps that exceeds 80%, a substantial improvement over the roughly 50% yields typical of conventional bromination-based routes. For procurement and technical teams, this translates to a more predictable and cost-effective supply of essential beta-lactam building blocks.
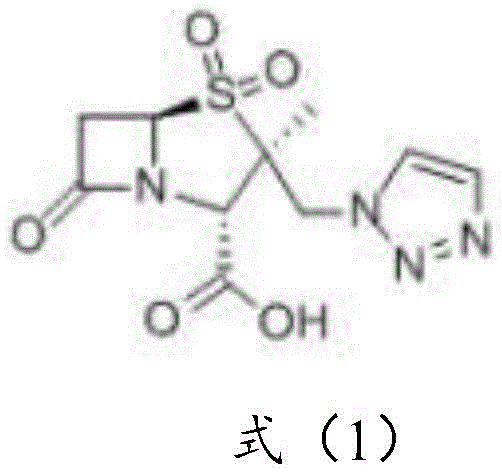
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing tazobactam intermediates have been fraught with inefficiencies and environmental hazards that hinder scalable manufacturing. One prominent prior art route, exemplified by the work of Taniguchi et al., relies on the use of mercury oxide for oxidation, presenting severe environmental pollution risks and requiring complex waste treatment protocols that drive up operational costs. Furthermore, alternative routes starting from penicillin G potassium salt, while utilizing cheaper raw materials, often involve dangerous explosive reagents and suffer from the formation of six-membered ring isomeric by-products during nucleophilic reactions, which drastically lowers the overall yield and complicates purification. The most common industrial route currently in use involves the bromination of 6-APA followed by oxidation, as depicted in the schematic below, but this method is fundamentally limited by the instability of the brominated intermediate.
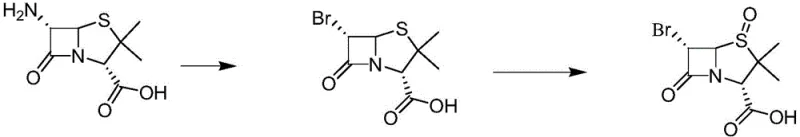
As illustrated in the conventional pathway, the bromination of 6-APA generates an intermediate that possesses high water solubility and poor stability, necessitating immediate oxidation to prevent degradation. However, in practical industrial operations, it is often impossible to coordinate the bromination and oxidation reactions instantaneously, leading to significant decomposition and a cumulative yield for these two steps of only about 50%. This low efficiency creates a bottleneck in the supply chain, forcing manufacturers to process larger volumes of raw materials to achieve the same output, thereby inflating costs and increasing the burden on waste management systems. Additionally, the requirement for immediate processing limits batch flexibility and complicates logistics, making it difficult to maintain a consistent inventory of key intermediates for downstream drug production.
The Novel Approach
The innovative methodology disclosed in the patent data circumvents these pitfalls by employing a protective group strategy that fundamentally alters the physicochemical properties of the intermediate. By introducing a Boc group to the amino functionality of 6-APA prior to oxidation, the resulting molecule becomes significantly less soluble in water and much more chemically robust. This stability allows the reaction mixture to be handled with greater flexibility; specifically, the oxidation with hydrogen peroxide can be performed directly in the aqueous reaction medium without the need to concentrate or isolate the sensitive brominated species found in older routes. The result is a streamlined process where the protection and oxidation steps effectively merge into a highly efficient sequence, boosting the total yield to over 80% and eliminating the need for hazardous mercury reagents.
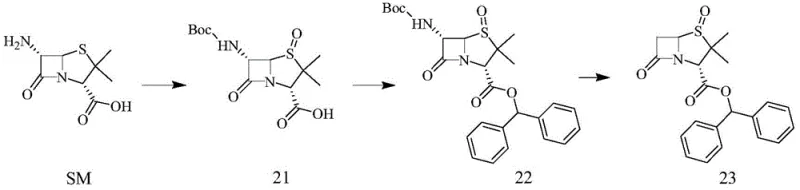
Furthermore, this novel approach offers distinct advantages in the downstream processing stages, particularly regarding the removal of protecting groups. Unlike benzyloxycarbonyl (Cbz) groups which require hydrogenation for removal—a process that introduces safety risks and requires specialized equipment—the Boc group can be cleaved under mild acidic conditions. Remarkably, the patent describes a synergistic effect where the acidic conditions used for Boc removal also facilitate the simultaneous removal of the amino group at the 6-position of the beta-lactam ring using sodium nitrite and hypophosphorous acid. This convergence of deprotection steps simplifies the synthetic tree, reduces the number of unit operations, and significantly lowers the consumption of solvents and reagents, making it an ideal candidate for cost reduction in pharmaceutical intermediates manufacturing on a commercial scale.
Mechanistic Insights into Boc-Protection and Tungstate-Catalyzed Oxidation
The success of this synthetic route hinges on the precise interplay between steric protection and catalytic oxidation mechanisms. The introduction of the bulky tert-butyloxycarbonyl (Boc) group serves a dual purpose: electronically, it moderates the nucleophilicity of the amino nitrogen, preventing unwanted side reactions during the subsequent oxidation phase, and sterically, it imparts hydrophobicity to the molecule. This hydrophobicity is crucial because it causes the protected intermediate to precipitate or remain in a distinct phase upon acidification, facilitating easy separation from the aqueous reaction matrix. The oxidation step itself utilizes hydrogen peroxide as a green oxidant, activated by a catalytic amount of sodium tungstate. This catalytic system is highly selective for the sulfur atom in the penicillane ring, converting the sulfide to the corresponding sulfoxide or sulfone without compromising the integrity of the sensitive beta-lactam ring, which is prone to hydrolysis under harsh conditions.
From an impurity control perspective, the stability conferred by the Boc group is paramount. In unprotected routes, the free amino group can participate in intramolecular cyclization or react with oxidizing agents to form N-oxides, leading to complex impurity profiles that are difficult to purge. By masking this functionality, the reaction pathway is directed exclusively towards sulfur oxidation. Moreover, the use of sodium tungstate accelerates the oxidation rate significantly; without this catalyst, the reaction would take several days to reach completion, whereas with it, the transformation is achieved in mere hours at room temperature. This kinetic enhancement not only improves throughput but also minimizes the time the beta-lactam core is exposed to oxidative stress, thereby preserving the stereochemical integrity of the chiral centers essential for the biological activity of the final tazobactam product.
How to Synthesize Tazobactam Intermediate Efficiently
The practical implementation of this chemistry involves a carefully controlled sequence of protection, oxidation, esterification, and deprotection, optimized for both laboratory and pilot-scale execution. The process begins with the dissolution of 6-aminopenicillanic acid in a mixed solvent system of water and 1,4-dioxane, followed by the addition of a base such as triethylamine and Boc anhydride to install the protecting group. Once the protection is complete, sodium tungstate and hydrogen peroxide are introduced directly to the same vessel to effect oxidation, yielding the stable sulfoxide intermediate which can be isolated by pH adjustment and filtration. The subsequent esterification with benzhydryl alcohol employs standard coupling reagents like DCC and DMAP in dichloroethane, ensuring high conversion to the diphenylmethyl ester, which is a critical functionality for the final drug's pharmacokinetic properties.
- Protect the amino group of 6-aminopenicillanic acid (6-APA) using Boc anhydride and oxidize with hydrogen peroxide catalyzed by sodium tungstate to obtain Compound 1.
- Perform esterification of Compound 1 with benzhydryl alcohol using DCC and DMAP catalysts in DCE solvent to form Compound 2.
- Remove the Boc protecting group and the 6-position amino group under acidic conditions using sodium nitrite and hypophosphorous acid to yield the final intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic methodology offers compelling economic and logistical benefits that extend beyond simple yield metrics. The primary advantage lies in the drastic simplification of the process flow; by eliminating the need for intermediate concentration and enabling a one-pot protection-oxidation sequence, the manufacturing cycle time is significantly reduced. This efficiency gain translates directly into lower utility costs and reduced demand on reactor capacity, allowing facilities to produce more material with the same infrastructure. Furthermore, the replacement of toxic mercury-based oxidants with benign hydrogen peroxide aligns with increasingly stringent environmental regulations, reducing the costs associated with hazardous waste disposal and environmental compliance monitoring, which are often hidden but substantial expenses in fine chemical manufacturing.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents, combined with the high yield of the initial steps, leads to a substantial decrease in the cost of goods sold. By avoiding the low-yielding bromination route, manufacturers can maximize the utilization of the starting material, 6-APA, which is a significant cost driver. Additionally, the ability to perform deprotection and amino-removal in a single step reduces the consumption of acids, bases, and solvents, further driving down variable costs without compromising the quality of the final intermediate.
- Enhanced Supply Chain Reliability: The chemical stability of the Boc-protected intermediate is a game-changer for inventory management. Unlike the unstable brominated species in conventional routes that must be consumed immediately, the protected intermediate can be stored and transported with minimal risk of degradation. This stability provides supply chain planners with greater flexibility to buffer stock against demand fluctuations or raw material shortages, ensuring a continuous supply of critical intermediates to downstream API manufacturers and reducing the risk of production stoppages due to material spoilage.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing common reagents and avoiding extreme conditions that pose engineering challenges at large volumes. The use of water as a primary solvent for the initial steps reduces the fire load and VOC emissions associated with organic solvents, making the facility safer and easier to permit. This environmental friendliness not only future-proofs the supply chain against tightening regulations but also enhances the corporate sustainability profile of the manufacturing partner, a key consideration for multinational pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this technology outperforms legacy methods in terms of yield, safety, and operational simplicity. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: How does the Boc-protection strategy improve yield compared to conventional bromination routes?
A: Conventional routes involving direct bromination of 6-APA suffer from instability and low yields (around 50%) due to the high water solubility of intermediates. The novel Boc-protection method reduces water solubility and stabilizes the molecule, allowing direct oxidation without concentration, thereby increasing the two-step yield to over 80%.
Q: What are the safety advantages of using hydrogen peroxide and Boc groups in this synthesis?
A: This method replaces hazardous reagents like mercury oxide used in older routes with environmentally benign hydrogen peroxide, which produces water as a byproduct. Additionally, the Boc group can be removed under acidic conditions without requiring dangerous hydrogenation processes, significantly enhancing operational safety.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the process is highly scalable. It eliminates the need for intermediate concentration steps, uses stable intermediates that are easy to store, and simplifies the deprotection sequence by combining amino removal and Boc cleavage into a single step, making it ideal for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tazobactam Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in securing the global supply of life-saving antibiotics. Our team of expert chemists has thoroughly analyzed the technological advancements presented in patent CN109678886B and is fully prepared to translate this intellectual property into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields and purity specifications demonstrated in the lab are faithfully reproduced at an industrial scale. Our rigorous QC labs and state-of-the-art manufacturing facilities are equipped to handle the specific requirements of beta-lactam chemistry, including the strict containment and purification protocols necessary for high-purity pharmaceutical intermediates.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis method can optimize your supply chain. By leveraging our expertise in process development, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how the yield improvements and safety enhancements of this route translate into bottom-line value. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred partner for the commercial scale-up of complex pharmaceutical intermediates.
