Advanced Synthetic Route for Erlotinib Intermediate: Commercial Scale-Up and Technical Insights
The pharmaceutical landscape for non-small cell lung cancer treatment continues to evolve, with Erlotinib Hydrochloride standing as a critical therapeutic agent for patients with advanced metastatic ALK positive conditions. Patent CN106946650B discloses a robust synthetic method for a key intermediate, 2-(4-Ethyl-3-iodoPhenyl)-2-methyl propanoic acid, which addresses significant challenges in current manufacturing protocols. This technical breakthrough offers a streamlined pathway that enhances overall process efficiency while maintaining stringent quality standards required for oncology drug production. The structural integrity of this molecule is paramount for downstream coupling reactions, necessitating a synthesis route that minimizes impurity generation and maximizes yield consistency. As a reliable pharmaceutical intermediates supplier, understanding the nuances of this specific chemical architecture is essential for ensuring supply chain continuity for global drug manufacturers. The disclosed method not only simplifies the operational workflow but also provides a foundation for cost reduction in API manufacturing by utilizing accessible reagents and standard reaction conditions. 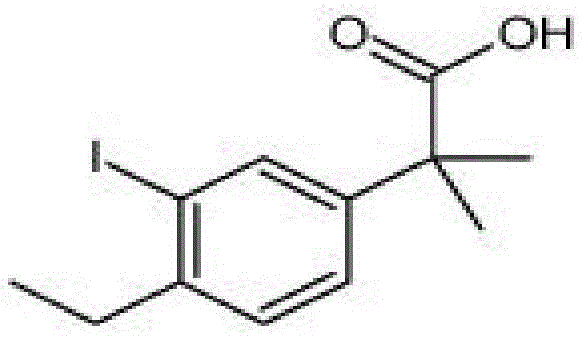
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex pharmaceutical intermediates like the erlotinib precursor has been plagued by convoluted reaction sequences that introduce unnecessary operational risks and economic inefficiencies. Conventional routes often suffer from low overall yields due to cumulative losses across multiple purification stages, leading to substantial material waste and escalated production costs. Many existing methods rely on harsh reaction conditions or expensive catalysts that complicate the removal of trace metal impurities, which is a critical concern for regulatory compliance in drug substance manufacturing. Furthermore, the lack of robust crystallization protocols in older methodologies often results in inconsistent polymorphic forms, potentially affecting the stability and bioavailability of the final active pharmaceutical ingredient. These technical bottlenecks create significant supply chain vulnerabilities, making it difficult for procurement teams to secure consistent volumes of high-purity materials. The industry demand for more sustainable and economically viable synthesis routes has never been more pressing, driving the need for innovation in process chemistry.
The Novel Approach
The methodology outlined in patent CN106946650B represents a paradigm shift by introducing a five-step sequence that prioritizes operational simplicity and high conversion efficiency. This novel approach leverages a strategic combination of chlorination, Friedel-Crafts acylation, and rearrangement reactions to construct the core carbon skeleton with remarkable precision. By optimizing reagent ratios, such as the molar ratio of compound 1 to thionyl chloride at 1:1 to 1:1.5, the process minimizes side reactions and maximizes the utilization of starting materials. The integration of a specific iodination step using iodine and iodic acid ensures the precise introduction of the iodine moiety without compromising the integrity of the sensitive ester functionality. This route is explicitly designed for commercial scale-up of complex pharmaceutical intermediates, offering a clear advantage over legacy methods that struggle with reproducibility at larger batch sizes. The ability to achieve a total yield of 67% in optimized examples demonstrates the practical viability of this chemistry for industrial applications. 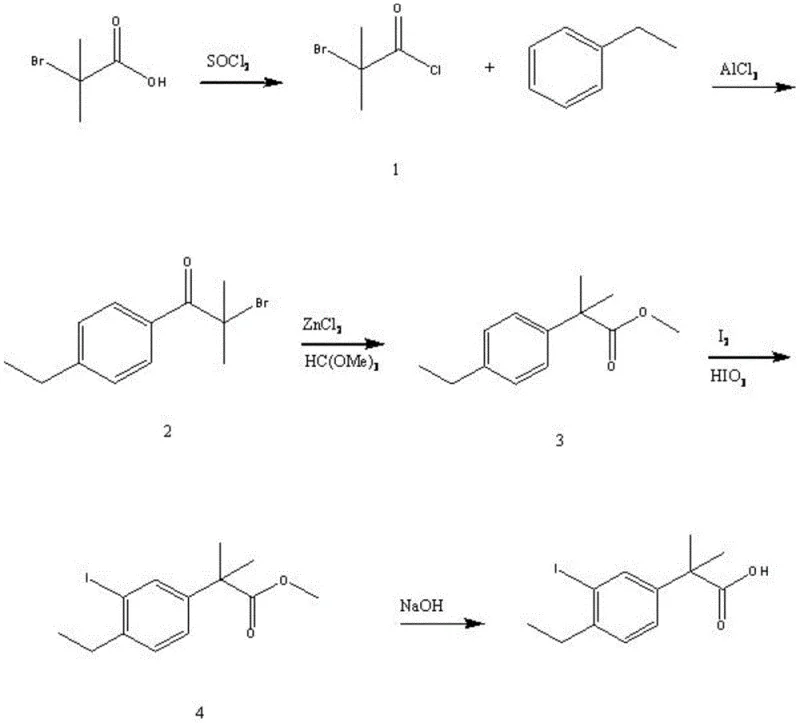
Mechanistic Insights into FeCl3-Catalyzed Cyclization and Iodination
The core of this synthetic strategy lies in the meticulous control of electrophilic aromatic substitution and subsequent functional group transformations. The initial activation of 2-bromo-2-methylpropanoic acid to its acid chloride derivative creates a highly reactive electrophile capable of efficient coupling with ethylbenzene under Lewis acid catalysis. The use of anhydrous aluminum trichloride facilitates the Friedel-Crafts acylation at low temperatures below -5°C, which is critical for controlling regioselectivity and preventing poly-acylation side products. Following the acylation, the zinc chloride catalyzed rearrangement with trimethoxymethane serves as a pivotal step, converting the ketone intermediate into a protected ester form that is more amenable to subsequent halogenation. This rearrangement mechanism effectively masks the reactive carbonyl group, allowing for the selective iodination of the aromatic ring in the presence of other sensitive functionalities. The final hydrolysis step under alkaline conditions cleanly removes the protecting group to reveal the target carboxylic acid, completing the synthesis with high fidelity.
Impurity control is rigorously addressed through the manipulation of crystallization conditions to isolate specific polymorphic forms of the target molecule. The patent details three distinct crystal forms, with forms B and C exhibiting superior stability characteristics compared to form A, as evidenced by their powder X-ray diffraction patterns. By utilizing specific solvent systems such as acetone-n-hexane or ethyl acetate during the recrystallization phase, manufacturers can selectively precipitate the more stable polymorphs, thereby ensuring long-term storage stability and consistent processing behavior. This level of control over solid-state properties is crucial for reducing lead time for high-purity pharmaceutical intermediates, as it eliminates the need for extensive re-processing or stability testing associated with metastable forms. The detailed characterization data provided, including specific diffraction peaks at 11.5, 13.2, and 17.5 degrees, offers a definitive fingerprint for quality assurance teams to verify batch consistency. Such mechanistic depth ensures that the production process remains robust against minor variations in raw material quality or environmental conditions.
How to Synthesize Erlotinib Intermediate Efficiently
The practical implementation of this synthesis route requires careful attention to reaction parameters and workup procedures to ensure optimal outcomes. The process begins with the activation of the starting acid, followed by a controlled acylation that demands precise temperature management to maintain selectivity. Subsequent steps involve reflux conditions for rearrangement and specific catalytic loads for iodination, all of which contribute to the overall efficiency of the transformation. The standardized synthesis steps see the detailed guide below for operational specifics.
- Activate 2-bromo-2-methylpropanoic acid with thionyl chloride and perform Friedel-Crafts acylation with ethylbenzene using aluminum trichloride.
- Execute zinc chloride catalyzed rearrangement with trimethoxymethane followed by iodination using iodine and iodic acid.
- Complete the synthesis via alkaline hydrolysis and recrystallization to obtain stable crystal forms of the target pharmaceutical intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers compelling advantages that directly address the pain points of procurement managers and supply chain directors. The reliance on readily available starting materials such as ethylbenzene and 2-bromo-2-methylpropanoic acid ensures a stable supply base that is not subject to the volatility of exotic reagent markets. This accessibility translates into significant cost savings by reducing the dependency on single-source suppliers and minimizing the risk of production stoppages due to material shortages. Furthermore, the simplified operational workflow reduces the burden on manufacturing facilities, allowing for faster turnaround times and increased throughput without the need for specialized equipment investments. The elimination of complex purification steps associated with transition metal catalysts further streamlines the production process, lowering both waste disposal costs and environmental compliance burdens.
- Cost Reduction in Manufacturing: The process achieves cost optimization through the use of common industrial solvents like carbon tetrachloride, ethyl acetate, and acetonitrile, which are economically priced and easily sourced in bulk quantities. By avoiding the use of expensive transition metal catalysts that require rigorous removal protocols, the method significantly lowers the cost of goods sold associated with purification and waste treatment. The high yield reported in the patent examples indicates efficient atom economy, meaning less raw material is wasted per unit of product produced, which directly impacts the bottom line. Additionally, the ability to operate at relatively mild temperatures for most steps reduces energy consumption, contributing to overall operational expenditure savings.
- Enhanced Supply Chain Reliability: The robustness of this chemical route enhances supply chain reliability by minimizing the number of critical control points where failures could occur. The use of stable intermediates and well-defined reaction conditions reduces the likelihood of batch failures, ensuring a consistent flow of material to downstream customers. This reliability is further bolstered by the flexibility in solvent selection, allowing manufacturers to adapt to regional availability without compromising reaction performance. Consequently, partners can expect more predictable delivery schedules and reduced lead times, which is essential for maintaining just-in-time inventory strategies in the pharmaceutical sector.
- Scalability and Environmental Compliance: The patent explicitly highlights the suitability of this method for large-scale industrial production, citing low requirements on equipment conditions as a key benefit. The straightforward workup procedures, involving standard aqueous washes and distillation, are easily adaptable to multi-tonne scale reactors without significant engineering modifications. Moreover, the generation of waste is minimized through high conversion rates and the potential for solvent recovery, aligning with increasingly stringent environmental regulations. This scalability ensures that the supply can grow in tandem with market demand for erlotinib, securing long-term partnership opportunities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of this erlotinib intermediate based on the patented technology. These insights are derived directly from the experimental data and beneficial effects described in the intellectual property documentation. Understanding these details helps stakeholders make informed decisions regarding process adoption and supply partnerships.
Q: What are the primary advantages of the synthetic method in patent CN106946650B?
A: The method offers simple operation, high total yield reaching up to 67% in optimized examples, and lower production costs compared to conventional routes, making it highly suitable for industrial application.
Q: How does this process address impurity control in erlotinib intermediate manufacturing?
A: The process utilizes specific recrystallization solvents like acetone-n-hexane or ethyl acetate to isolate stable crystal forms B and C, which exhibit superior stability and purity profiles compared to crystal form A.
Q: Is this synthetic route scalable for commercial API production?
A: Yes, the patent explicitly states the method has low requirements on equipment conditions, uses readily available raw materials, and is suitable for large-scale industrial production with good industrial prospects.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(4-Ethyl-3-iodoPhenyl)-2-methyl propanoic acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of this intermediate in the global supply chain for oncology therapeutics. Our CDMO expertise allows us to translate complex laboratory pathways like CN106946650B into robust commercial processes, ensuring that technical potential is fully realized in production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, ensuring that every batch of 2-(4-Ethyl-3-iodoPhenyl)-2-methyl propanoic acid meets the highest industry standards.
We invite you to collaborate with us to optimize your supply chain for this critical molecule. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality expectations. Please contact us to request specific COA data and route feasibility assessments that will demonstrate how our manufacturing capabilities can support your long-term strategic goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
