Advanced Manufacturing of Tebipenem Pivoxil: Technical Breakthroughs and Commercial Scalability
The pharmaceutical industry continuously seeks robust synthetic routes for critical antibiotic intermediates, and the technology disclosed in patent CN103059027A represents a significant advancement in the preparation of Tebipenem Pivoxil, also known as L-084. This oral carbapenem antibiotic intermediate requires precise stereochemical control and high purity to meet regulatory standards for downstream API production. The patented method introduces a streamlined three-step synthesis that addresses common inefficiencies found in legacy processes, specifically targeting the elimination of cumbersome purification steps that often hinder throughput. By optimizing reaction conditions and reagent stoichiometry, this approach offers a viable pathway for manufacturers aiming to enhance their production capabilities while maintaining stringent quality specifications required by global health authorities.
Traditional synthetic routes for carbapenem derivatives often suffer from low overall yields and complex purification requirements that escalate manufacturing costs and extend lead times. Conventional methods frequently rely heavily on column chromatography for intermediate purification, a technique that is notoriously difficult to scale, consumes vast quantities of solvents, and introduces bottlenecks in continuous production lines. Furthermore, older protocols may involve harsh reaction conditions that compromise the stability of the sensitive beta-lactam ring, leading to the formation of difficult-to-remove impurities that degrade the final product's quality profile. These limitations create significant challenges for procurement teams and supply chain managers who require consistent, high-volume supply without the risk of batch failures or extended downtime associated with complex downstream processing.
The Novel Approach
In contrast to these legacy methods, the novel approach detailed in the patent utilizes a rational synthetic design that replaces chromatographic purification with efficient crystallization and filtration techniques. The process begins with a highly controlled nucleophilic substitution where MAP and YKTP-1 are reacted in acetonitrile at low temperatures, specifically between -43°C and -37°C, to form the intermediate YKTP-2. This precise thermal management is crucial for maximizing the 66.7% yield reported in the embodiments while minimizing side reactions. The subsequent steps involve catalytic hydrogenation and esterification, both optimized to allow for direct isolation of products through physical separation methods. This shift from chromatographic to crystallization-based purification drastically simplifies the post-processing workflow, making the technology highly suitable for industrial production environments where efficiency and reproducibility are paramount.
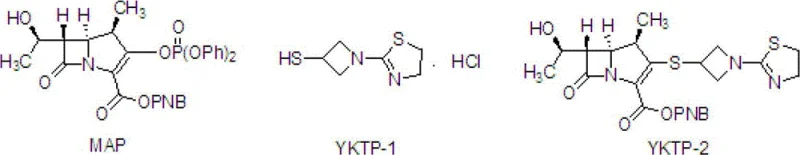
Mechanistic Insights into the Three-Step Synthesis of L-084
The core of this synthetic strategy lies in the meticulous control of reaction kinetics and thermodynamics across three distinct transformation stages. The initial step involves the coupling of the phosphonate intermediate MAP with the thiol-containing YKTP-1 in the presence of diisopropylethylamine (DIPEA) as a base. The reaction is conducted at cryogenic temperatures to ensure the nucleophilic attack occurs selectively without epimerization at the chiral centers, which is a common risk in carbapenem chemistry. Following the reaction, the system is warmed to 0°C to 15°C and quenched with water, allowing the product YKTP-2 to precipitate or be isolated via filtration, bypassing the need for complex extraction sequences. This mechanistic precision ensures that the stereochemical integrity of the molecule is preserved, which is critical for the biological activity of the final antibiotic.
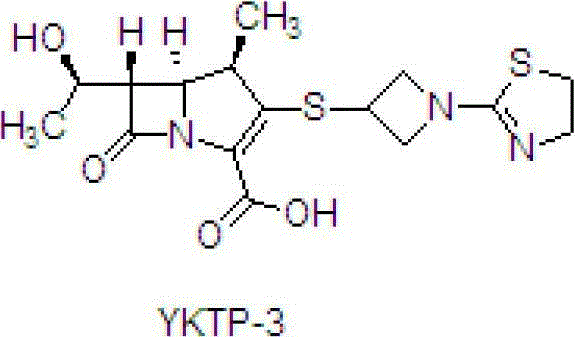
The second and third stages focus on deprotection and functionalization to yield the final pivoxil ester. The hydrogenation step utilizes palladium on carbon (Pd/C) under a hydrogen pressure of 0.8 to 1.0MPa to remove protecting groups, a process that must be carefully monitored to prevent over-reduction of the double bond in the carbapenem core. The final esterification with iodomethyl pivalate is performed in DMF at -10°C to 0°C using a specific base, ensuring high conversion rates. The purification strategy throughout these steps relies on pH adjustments and solvent-induced crystallization, such as using acetone to precipitate YKTP-3 at pH 3.55 to 3.65. This approach effectively manages the impurity profile, ensuring that the final L-084 product meets high-purity specifications without the need for resource-intensive chromatographic columns.
How to Synthesize Tebipenem Pivoxil Efficiently
Implementing this synthesis route requires strict adherence to the specified reaction parameters and workup procedures to achieve the reported yields and purity levels. The process is designed to be operationally simple, relying on standard chemical engineering unit operations that are easily scalable from pilot plant to commercial manufacturing scales. Operators must ensure precise temperature control during the addition of reagents, particularly the dropwise addition of DIPEA and iodomethyl pivalate, to manage exotherms and maintain reaction selectivity. The detailed standardized synthesis steps, including specific solvent volumes, stirring times, and filtration protocols, are outlined in the technical guide below to assist R&D teams in replicating this high-efficiency pathway.
- React MAP with YKTP-1 in acetonitrile at -43°C to -37°C using DIPEA to form intermediate YKTP-2, followed by crystallization.
- Perform catalytic hydrogenation on YKTP-2 using Pd/C at 0.8-1.0MPa hydrogen pressure to remove protecting groups and yield YKTP-3.
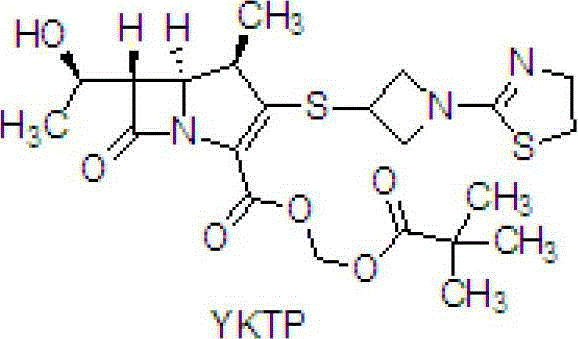
- Esterify YKTP-3 with iodomethyl pivalate in DMF at -10°C to 0°C using artemisinin salt as a base to finalize Tebipenem Pivoxil.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process offers substantial strategic benefits beyond mere technical feasibility. The elimination of column chromatography is a transformative change that directly impacts the cost structure and throughput of the manufacturing facility. By removing this bottleneck, production cycles are significantly shortened, allowing for faster turnaround times and increased annual capacity without the need for capital-intensive equipment upgrades. This efficiency translates into a more resilient supply chain capable of meeting fluctuating market demands for critical antibiotic intermediates, ensuring continuity of supply for downstream pharmaceutical partners.
- Cost Reduction in Manufacturing: The removal of column chromatography operations leads to significant cost savings by reducing solvent consumption and waste disposal expenses associated with large-scale purification. Additionally, the simplified workflow reduces labor hours and equipment occupancy time, allowing the facility to allocate resources more effectively across other production lines. The use of readily available reagents and standard reaction conditions further contributes to a lower cost of goods sold, making the final intermediate more competitive in the global market without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain reliability by minimizing the risk of batch failures associated with complex purification steps. The reliance on crystallization and filtration, which are well-understood and easily controlled unit operations, ensures consistent product quality and yield from batch to batch. This predictability allows supply chain planners to optimize inventory levels and reduce safety stock requirements, knowing that the production process is stable and capable of delivering on schedule even under high-volume demands.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process aligns well with green chemistry principles by reducing the volume of organic solvents required for purification. The simplified post-processing steps generate less hazardous waste, facilitating easier compliance with increasingly stringent environmental regulations. Furthermore, the process is inherently scalable, as the reaction conditions and workup procedures do not rely on laboratory-scale techniques that are difficult to translate to multi-ton production, ensuring a smooth transition from development to commercial manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the patent data and are intended to clarify the operational advantages and technical specifics for stakeholders evaluating this technology for potential adoption or sourcing. Understanding these details is crucial for making informed decisions regarding process integration and supplier qualification.
Q: How does this patent improve upon traditional carbapenem synthesis?
A: The method described in CN103059027A eliminates the need for column chromatography, a bottleneck in traditional synthesis, by utilizing rational crystallization and filtration steps that significantly simplify post-processing and reduce production time.
Q: What are the critical control points for ensuring high purity in L-084 production?
A:
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is designed for industrial suitability, utilizing standard unit operations like filtration and crystallization rather than batch-limited chromatography, facilitating easier commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tebipenem Pivoxil Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for high-value pharmaceutical intermediates like Tebipenem Pivoxil. Our team of expert chemists and engineers possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic challenges are met with robust industrial solutions. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, providing our partners with the confidence they need to advance their own drug development pipelines without supply chain interruptions.
We invite global pharmaceutical companies and procurement leaders to collaborate with us to optimize their supply chains for carbapenem intermediates. By leveraging our technical expertise and manufacturing capabilities, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the most reliable and cost-effective sourcing strategy available in the market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
