Advanced NFSI Catalysis for Scalable Asymmetric Disulfide Manufacturing
Advanced NFSI Catalysis for Scalable Asymmetric Disulfide Manufacturing
The landscape of fine chemical synthesis is undergoing a significant transformation driven by the urgent need for greener, more cost-effective methodologies, particularly in the production of sulfur-containing organic compounds. Patent CN113563241A introduces a groundbreaking approach for the synthesis of asymmetric disulfide derivatives utilizing N-Fluorobenzenesulfonimide (NFSI) as an efficient organocatalyst. This technology represents a paradigm shift away from traditional transition metal-catalyzed processes, offering a sustainable alternative that addresses critical pain points in pharmaceutical and agrochemical manufacturing. By leveraging a reversible exchange reaction between two distinct symmetric disulfides, this method achieves high atom economy and operational simplicity without the reliance on precious metals or hazardous reagents. For industry leaders seeking a reliable agrochemical intermediate supplier or partners capable of high-purity pharmaceutical intermediate production, understanding the nuances of this catalytic system is essential for optimizing supply chains and reducing overall production costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of asymmetric disulfides has been plagued by significant technical and economic hurdles that hinder large-scale commercialization. The most common traditional route involves the oxidative dehydrogenation of two different thiols; however, this method suffers from severe drawbacks, primarily the intense, unpleasant odor associated with mercaptans, which poses serious safety and environmental challenges in a manufacturing setting. Furthermore, this oxidative approach often lacks selectivity, leading to the formation of symmetrical disulfide byproducts through self-coupling, thereby complicating downstream purification and reducing overall yield. Alternatively, strategies employing transition metal catalysts, such as expensive rhodium complexes, have been developed to facilitate disulfide exchange; yet, the prohibitive cost of rhodium and the stringent regulatory requirements for residual heavy metal content in final API products make these routes less attractive for cost-sensitive applications. These legacy methods create bottlenecks in cost reduction in electronic chemical manufacturing and other high-value sectors where purity and expense are critical factors.
The Novel Approach
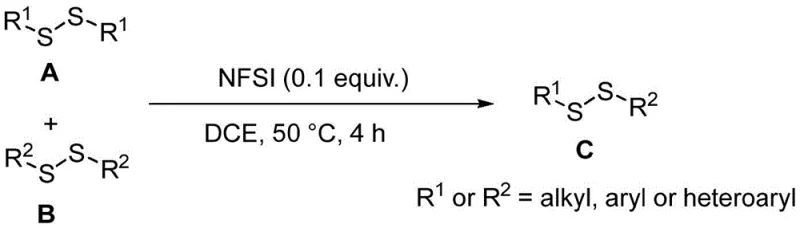
In stark contrast to these conventional limitations, the NFSI-catalyzed method disclosed in the patent offers a streamlined, metal-free pathway that utilizes stable symmetric disulfides as readily available starting materials. This innovative process operates under mild conditions, typically at 50 °C in 1,2-dichloroethane, avoiding the extreme temperatures or pressures often required by older technologies. The use of catalytic amounts of NFSI (0.1 equivalent) drives the reversible equilibrium towards the desired asymmetric product, effectively bypassing the need for stoichiometric oxidants or toxic thiols. This approach not only simplifies the reaction setup but also enhances the safety profile of the manufacturing process, making it highly suitable for the commercial scale-up of complex polymer additives or specialty chemicals. By eliminating the reliance on precious metals, this method inherently lowers the barrier to entry for production, ensuring a more robust and continuous supply chain for key sulfur-containing intermediates.
Mechanistic Insights into NFSI-Catalyzed Disulfide Exchange
The efficacy of this synthesis lies in the unique ability of NFSI to activate the sulfur-sulfur bond through a reversible mechanistic cycle that avoids permanent consumption of the catalyst. The reaction initiates when the catalytic NFSI interacts with one of the symmetric disulfide substrates, generating reactive sulfenyl intermediates that are primed for nucleophilic attack or radical exchange. Unlike irreversible oxidation processes, this system establishes a dynamic equilibrium where the symmetric disulfides A and B continuously exchange sulfur fragments. The presence of NFSI lowers the activation energy for this exchange, allowing the system to thermodynamically favor the formation of the asymmetric disulfide product C over time. This mechanistic feature is crucial for R&D directors focused on impurity control, as the reversibility allows the system to self-correct, minimizing the accumulation of kinetic byproducts that often plague irreversible coupling reactions.
Furthermore, the mechanism ensures high selectivity by preventing the formation of homocoupled side products that typically arise from free thiol intermediates. Since the reaction proceeds directly from disulfide to disulfide without generating free mercaptans in the bulk solution, the characteristic foul odors are completely suppressed, and the risk of self-coupling is drastically reduced. The catalytic cycle concludes with the regeneration of the NFSI species, confirming its role as a true catalyst rather than a stoichiometric reagent. This efficiency translates directly to process economics, as only a small fraction of the promoter is needed to drive the conversion of substantial quantities of starting material. Understanding this mechanism is vital for optimizing reaction parameters such as concentration and temperature to maximize yield and throughput in an industrial reactor setting.
How to Synthesize Asymmetric Disulfides Efficiently
Implementing this synthesis route requires precise control over reaction stoichiometry and thermal conditions to ensure optimal conversion rates and product purity. The general procedure involves combining the two symmetric disulfide precursors in a 1:1 molar ratio with a catalytic loading of NFSI in a chlorinated solvent, followed by heating to promote the exchange equilibrium. While the patent highlights specific examples using 1,2-dichloroethane, the robustness of the catalyst suggests potential adaptability to other solvents depending on solubility profiles of specific substrates. The detailed standardized synthesis steps, including precise workup procedures and purification protocols to achieve pharmaceutical-grade purity, are outlined below for technical teams evaluating process feasibility.
- Prepare the reaction mixture by adding NFSI (0.1 equiv), symmetric disulfide A (1.0 equiv), and symmetric disulfide B (1.0 equiv) into a sealed tube containing 1,2-dichloroethane solvent.
- Heat the reaction system to 50 °C and stir vigorously for 4 hours to allow the reversible exchange reaction to reach equilibrium favoring the asymmetric product.
- Upon completion, concentrate the reaction solution and perform column chromatography separation to isolate the high-purity asymmetric disulfide derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this NFSI-catalyzed technology offers transformative benefits that extend far beyond simple chemical yield improvements. The elimination of rhodium catalysts removes a major volatility risk associated with precious metal pricing and availability, stabilizing the cost structure for long-term production contracts. Additionally, the use of stable, non-volatile disulfide starting materials simplifies logistics and storage requirements compared to handling hazardous thiols, thereby reducing insurance and compliance costs associated with dangerous goods transportation. This process inherently supports sustainability goals by reducing heavy metal waste streams, which simplifies effluent treatment and aligns with increasingly strict environmental regulations in major manufacturing hubs.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the complete removal of expensive transition metal catalysts from the bill of materials. By substituting rhodium with a catalytic amount of inexpensive NFSI, manufacturers can achieve substantial cost savings on raw materials while simultaneously eliminating the costly downstream processing steps required to scavenge trace metals from the final product. This reduction in processing complexity shortens the overall production cycle time and reduces solvent consumption, leading to a leaner, more cost-efficient manufacturing operation that improves margin potential for high-volume intermediates.
- Enhanced Supply Chain Reliability: Relying on symmetric disulfides as feedstocks provides a distinct supply chain advantage, as these compounds are generally more stable and easier to source globally than sensitive thiols or specialized metal catalysts. This stability ensures consistent quality and availability of starting materials, reducing the risk of production delays caused by reagent degradation or supply shortages. Furthermore, the robustness of the reaction conditions allows for flexible manufacturing scheduling, enabling facilities to respond more agilely to fluctuating market demands without compromising product integrity or delivery timelines.
- Scalability and Environmental Compliance: The absence of malodorous thiols and heavy metals makes this process exceptionally scalable, as it mitigates the community and worker safety concerns that often limit batch sizes in traditional sulfur chemistry. The simplified waste profile facilitates easier regulatory approval for new manufacturing sites and reduces the burden on wastewater treatment infrastructure. This environmental compatibility is a strategic asset for companies aiming to expand capacity in regions with rigorous ecological standards, ensuring long-term operational continuity and social license to operate.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology for their specific applications, we have compiled answers to common inquiries regarding the scope, safety, and scalability of the NFSI-catalyzed disulfide synthesis. These insights are derived directly from the experimental data and technical disclosures within the patent literature, providing a factual basis for process assessment. Understanding these details is critical for integrating this methodology into existing production workflows and ensuring alignment with quality and safety standards.
Q: What are the primary advantages of using NFSI over Rhodium catalysts for disulfide synthesis?
A: The NFSI-catalyzed method eliminates the need for expensive transition metal rhodium catalysts, significantly reducing raw material costs and removing the requirement for complex heavy metal removal steps during purification, which aligns with green chemistry principles.
Q: Does this process require the use of odorous mercaptans?
A: No, unlike traditional oxidative dehydrogenation methods that rely on smelly thiols (mercaptans), this novel approach utilizes stable symmetric disulfides as starting materials, greatly improving the working environment and safety profile.
Q: What is the typical yield range for this asymmetric disulfide synthesis?
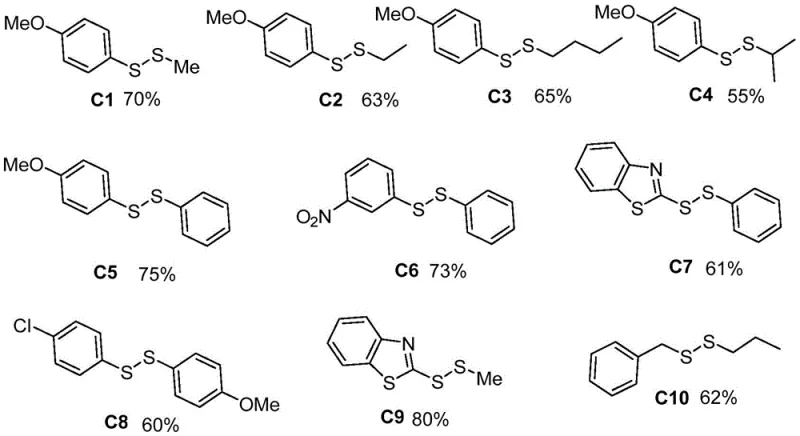
A: According to patent data, the method achieves isolated yields ranging from approximately 55% to 80% across various substrates, including alkyl, aryl, and heteroaryl disulfides, demonstrating robust substrate tolerance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Asymmetric Disulfide Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting innovative catalytic technologies like the NFSI-mediated disulfide exchange to maintain a competitive edge in the global fine chemicals market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory breakthroughs are seamlessly translated into robust industrial processes. We are committed to delivering high-purity asymmetric disulfides that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By partnering with us, clients gain access to a supply chain that prioritizes both technical excellence and operational reliability.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific project requirements. Whether you need a Customized Cost-Saving Analysis for transitioning from metal-catalyzed methods or require specific COA data to validate our quality standards, we are ready to provide comprehensive support. Contact us today to request route feasibility assessments and discover how our expertise in sulfur chemistry can accelerate your development timelines and optimize your manufacturing costs.
