Advanced Photocatalytic Synthesis of Quinazolinones for Scalable Pharmaceutical Manufacturing
Introduction to Next-Generation Quinazolinone Synthesis
The pharmaceutical and agrochemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways for constructing nitrogen-containing heterocycles, particularly quinazolinones, which serve as critical scaffolds in numerous bioactive molecules. A significant breakthrough in this domain is documented in Chinese Patent CN108822046B, which discloses a novel one-pot method for synthesizing quinazolinone compounds. This technology represents a paradigm shift from traditional thermal and metal-catalyzed processes to a green photocatalytic approach. By leveraging visible light and organic photosensitizers, this method achieves high yields under ambient conditions, addressing key pain points related to energy consumption and metal contamination. For R&D directors and procurement strategists, understanding the implications of this patent is crucial for optimizing supply chains and reducing the environmental footprint of complex intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinone derivatives has relied heavily on transition metal catalysis and harsh reaction conditions, which pose significant challenges for industrial scale-up and regulatory compliance. For instance, earlier methodologies reported by groups such as Yuusaku Yokoyama utilized palladium catalysts to effect benzylc-H bond amidation, requiring noble metals that are not only expensive but also difficult to remove to ppm levels required for API production. 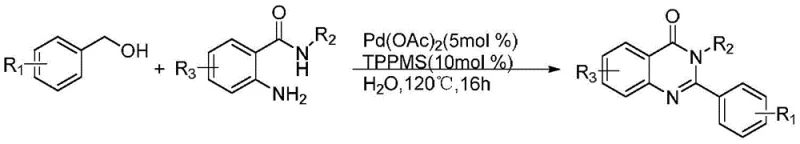 Other approaches, such as those employing copper bromide or iron chloride, often necessitate the use of hazardous oxidants like tert-butyl hydroperoxide (TBHP) and elevated temperatures ranging from 80°C to 130°C. These conditions increase the risk of thermal runaway, complicate safety protocols, and generate substantial waste streams containing heavy metals and peroxide byproducts. Furthermore, the multi-step nature of some traditional routes often requires the isolation of unstable aldehyde intermediates, leading to lower overall throughput and increased operational costs due to additional purification steps.
Other approaches, such as those employing copper bromide or iron chloride, often necessitate the use of hazardous oxidants like tert-butyl hydroperoxide (TBHP) and elevated temperatures ranging from 80°C to 130°C. These conditions increase the risk of thermal runaway, complicate safety protocols, and generate substantial waste streams containing heavy metals and peroxide byproducts. Furthermore, the multi-step nature of some traditional routes often requires the isolation of unstable aldehyde intermediates, leading to lower overall throughput and increased operational costs due to additional purification steps.
The Novel Approach
In stark contrast, the method described in CN108822046B introduces a metal-free, room-temperature strategy that fundamentally simplifies the synthetic landscape. This innovative route utilizes readily available primary alcohols and anthranilamide derivatives as starting materials, bypassing the need for pre-oxidized aldehydes or halogenated precursors. The core of this advancement lies in the use of organic photosensitizers, such as Eosin Y, Rose Bengal, or Fluorescein, which activate molecular oxygen under visible light irradiation to drive the oxidative transformation. This eliminates the dependency on scarce precious metals like palladium, iridium, or rhodium, thereby drastically reducing the raw material cost profile. Moreover, the reaction proceeds efficiently at 20-25°C, removing the energy burden associated with heating reactors to over 100°C and mitigating safety risks linked to high-pressure or high-temperature operations. The one-pot design ensures that the intermediate aldehyde generated in situ is immediately consumed in the subsequent cyclization step, maximizing atom economy and minimizing solvent usage.
Mechanistic Insights into Photocatalytic Oxidative Cyclization
The mechanistic pathway of this transformation is a sophisticated interplay of photo-induced electron transfer and aerobic oxidation, offering a clean alternative to stoichiometric oxidants. In the first stage of the reaction, the primary alcohol substrate undergoes selective oxidation to form the corresponding aldehyde. This process is mediated by the excited state of the photosensitizer, which transfers energy or electrons to molecular oxygen, generating reactive oxygen species that abstract hydrogen from the alcohol.  This step is critical as it avoids the use of toxic chromium or manganese-based oxidants traditionally used for alcohol oxidation. The resulting aldehyde is highly reactive and, in the presence of an acidic promoter, readily condenses with the amino group of the anthranilamide derivative. The second stage involves an intramolecular cyclization where the amide nitrogen attacks the imine carbon, followed by oxidative aromatization to form the stable quinazolinone ring system.
This step is critical as it avoids the use of toxic chromium or manganese-based oxidants traditionally used for alcohol oxidation. The resulting aldehyde is highly reactive and, in the presence of an acidic promoter, readily condenses with the amino group of the anthranilamide derivative. The second stage involves an intramolecular cyclization where the amide nitrogen attacks the imine carbon, followed by oxidative aromatization to form the stable quinazolinone ring system.  The entire sequence is driven by light energy and air, making it a truly green chemical process. The absence of metal catalysts means there is no risk of metal leaching into the final product, a critical quality attribute for pharmaceutical intermediates destined for clinical use. The acidic additives, such as p-toluenesulfonic acid or acetic acid, play a dual role in facilitating the condensation and stabilizing the reaction intermediates, ensuring high conversion rates without the need for exotic reagents.
The entire sequence is driven by light energy and air, making it a truly green chemical process. The absence of metal catalysts means there is no risk of metal leaching into the final product, a critical quality attribute for pharmaceutical intermediates destined for clinical use. The acidic additives, such as p-toluenesulfonic acid or acetic acid, play a dual role in facilitating the condensation and stabilizing the reaction intermediates, ensuring high conversion rates without the need for exotic reagents.
How to Synthesize Quinazolinone Derivatives Efficiently
Implementing this photocatalytic protocol in a laboratory or pilot plant setting requires careful attention to light source selection and oxygen mass transfer, but the operational procedure is remarkably straightforward compared to traditional methods. The process begins by dissolving the alcohol substrate and a catalytic amount of photosensitizer (1-3 mol%) in a suitable solvent such as DMSO, DMF, or acetonitrile. The mixture is then irradiated with a xenon lamp or LED array while bubbling oxygen or air through the solution for approximately 20 hours to ensure complete oxidation to the aldehyde. Following this, the anthranilamide and an acid promoter are added directly to the same vessel, and irradiation continues for another 24 hours to drive the cyclization to completion. The detailed standardized synthesis steps, including specific molar ratios, solvent choices, and workup procedures for various substituents, are outlined below.
- Oxidize the primary alcohol substrate to the corresponding aldehyde using an organic photosensitizer (e.g., Eosin Y) under visible light irradiation and oxygen atmosphere at room temperature.
- Without isolation, add the anthranilamide derivative and an acidic promoter (e.g., PTSA or acetic acid) directly to the reaction mixture.
- Continue irradiation under oxygen or air at room temperature to facilitate cyclization, yielding the target quinazolinone compound after standard workup.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology offers compelling strategic advantages that extend beyond mere technical feasibility. The elimination of noble metal catalysts represents a direct and significant reduction in raw material costs, as palladium and iridium prices are subject to high volatility and geopolitical supply risks. By switching to organic dyes like Eosin Y, which are commodity chemicals, manufacturers can stabilize their bill of materials and achieve substantial cost savings in quinazolinone manufacturing. Furthermore, the mild reaction conditions translate to enhanced supply chain reliability; the process does not require specialized high-pressure reactors or extensive cooling systems for exothermic quenches, allowing for production in standard glass-lined or stainless steel vessels equipped with simple lighting arrays. This flexibility reduces capital expenditure barriers for scaling up production capacity.
- Cost Reduction in Manufacturing: The removal of expensive transition metals and hazardous peroxides from the process workflow leads to a leaner cost structure. Without the need for costly metal scavengers or complex wastewater treatment for heavy metal removal, the downstream processing becomes significantly cheaper. The use of air as the terminal oxidant further reduces the cost of goods sold compared to purchasing stoichiometric chemical oxidants. Additionally, the high yields reported (up to 98%) minimize raw material waste, ensuring that every kilogram of input translates efficiently into saleable product, thereby improving overall margin potential for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Sourcing organic photosensitizers and simple alcohols is far less risky than securing supply chains for specialized organometallic catalysts which often have long lead times and single-source dependencies. The robustness of the reaction at room temperature also means that production is less susceptible to disruptions caused by utility failures, such as steam or chilled water outages, which are critical for high-temperature or cryogenic reactions. This resilience ensures consistent delivery schedules for clients relying on these intermediates for their own API synthesis, reducing the lead time for high-purity quinazolinone derivatives and strengthening the manufacturer's reputation as a dependable partner.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method is superior. The absence of peroxides eliminates the risk of explosion during storage and handling, while the metal-free nature simplifies regulatory filings and environmental audits. Scaling this reaction is primarily a function of light penetration and oxygen availability, which can be engineered using flow chemistry or specialized photoreactors, facilitating the commercial scale-up of complex pharmaceutical intermediates. The reduced generation of hazardous waste aligns with global sustainability goals, making the final product more attractive to multinational corporations with strict green chemistry mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis route. These insights are derived directly from the experimental data and claims within patent CN108822046B, providing a factual basis for evaluating its suitability for your specific production needs. Understanding these details is essential for R&D teams planning technology transfer and for procurement officers assessing vendor capabilities.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the patented process (CN108822046B) utilizes organic photosensitizers such as Eosin Y or Rose Bengal instead of precious metals like Palladium or Iridium, significantly reducing raw material costs and eliminating heavy metal residue concerns.
Q: What are the reaction conditions for this quinazolinone synthesis?
A: The reaction proceeds under mild conditions, specifically at room temperature (20-25°C) using visible light irradiation (such as Xenon lamps or LEDs) and air or oxygen as the oxidant, avoiding the need for high temperatures or hazardous peroxides.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the one-pot nature of the reaction, combined with the use of inexpensive solvents and the absence of harsh conditions, makes it highly scalable and environmentally compliant for commercial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinazolinone Intermediates Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the photocatalytic synthesis route described in CN108822046B for the production of high-value quinazolinone scaffolds. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our facilities are equipped with advanced photoreactors and stringent purity specifications, supported by rigorous QC labs capable of detecting trace impurities down to ppm levels. We are committed to delivering high-purity quinazolinone intermediates that meet the exacting standards of the global pharmaceutical industry, leveraging our expertise in green chemistry to provide sustainable manufacturing solutions.
We invite you to collaborate with us to optimize your supply chain for quinazolinone derivatives. Our technical team is ready to perform a Customized Cost-Saving Analysis tailored to your specific target molecules, evaluating the feasibility of adapting this metal-free protocol to your existing portfolio. Please contact our technical procurement team to request specific COA data, route feasibility assessments, and volume pricing options. By partnering with us, you gain access to a secure, cost-effective, and environmentally responsible supply source for your critical drug development projects.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
