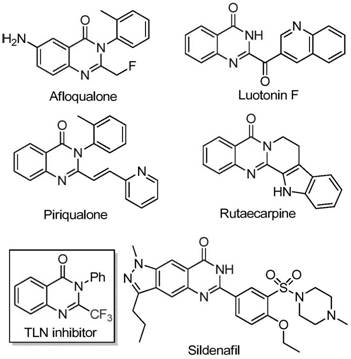
Scalable Iron-Catalyzed Route to 2-Trifluoromethyl Quinazolinones for Advanced Drug Discovery
Introduction to Advanced Quinazolinone Synthesis
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient and cost-effective synthetic routes for bioactive heterocycles. A significant breakthrough in this domain is detailed in patent CN111675662B, which discloses a novel preparation method for 2-trifluoromethyl substituted quinazolinone compounds. These nitrogen-containing fused ring six-membered heterocycles are pivotal scaffolds found in numerous drug molecules exhibiting general anti-cancer, anticonvulsant, anti-inflammatory, antifungal, and antimalarial activities. The strategic introduction of a trifluoromethyl group into these heterocyclic systems confers distinct advantages, such as significantly improved electronegativity, bioavailability, metabolic stability, and lipophilicity of the target molecule. This patent presents a robust methodology that leverages readily available starting materials and a cheap metal iron catalyst to overcome the limitations of prior art, positioning it as a critical technology for reliable pharmaceutical intermediate suppliers aiming to optimize their production pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinones bearing trifluoromethyl functional groups has been fraught with significant technical and economic challenges. Literature reports indicate that traditional methods primarily rely on the cyclization reaction of a synthon containing a trifluoromethyl group with suitable substrates such as anthranilamide, anthranilic acid, and isatoic anhydride. Among the commonly used trifluoromethyl synthons are trifluoroacetic anhydride and ethyl trifluoroacetate. However, these established synthetic pathways are generally limited by severe disadvantages that hinder their widespread industrial adoption. These drawbacks include severe reaction conditions that often require extreme temperatures or pressures, the utilization of expensive reaction substrates that drive up the cost of goods, consistently low yields that waste raw materials, and a narrow substrate range that limits the structural diversity accessible to medicinal chemists. Such inefficiencies create bottlenecks in cost reduction in API manufacturing and complicate the supply chain for high-purity intermediates.
The Novel Approach
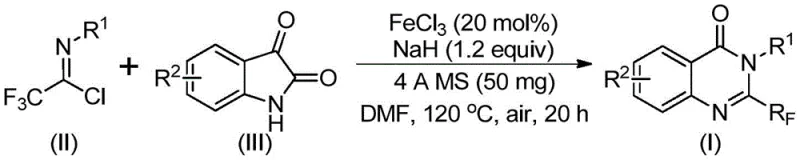
In stark contrast to these conventional struggles, the method disclosed in patent CN111675662B offers a paradigm shift by utilizing readily available trifluoroethylimidoyl chloride and isatin as starting raw materials. This innovative route employs a series of cyclization reactions catalyzed by cheap metal iron, specifically ferric chloride, which represents a drastic departure from precious metal catalysis. The process is characterized by its simple and convenient operation, good functional group tolerance, and wide reaction applicability. Crucially, the method has been demonstrated to be expandable to the gram level, providing a tangible possibility for industrial scale application. By shifting the synthetic strategy to utilize isatin derivatives and imidoyl chlorides under iron catalysis, the process effectively bypasses the need for harsh reagents and expensive precursors, thereby streamlining the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this technological advancement lies in the unique mechanistic pathway facilitated by the ferric chloride catalyst and sodium hydride base. During the reaction, an alkali-promoted carbon-nitrogen bond formation occurs between the trifluoroethylimidoyl chloride and the isatin substrate. This initial step generates a trifluoroacetamidine compound intermediate, which serves as the precursor for the subsequent ring closure. Following this, the iron catalyst mediates a decarbonylation and cyclization reaction, effectively isomerizing the intermediate to obtain the final 2-trifluoromethyl-substituted quinazolinone compound. This mechanism is particularly elegant because it avoids the formation of stable by-products that are common in acid-catalyzed dehydrations, ensuring a cleaner reaction profile. The use of 4A molecular sieves further aids in driving the equilibrium forward by sequestering moisture, which is critical for the stability of the imidoyl chloride and the efficiency of the cyclization step.
From an impurity control perspective, this mechanism offers superior manageability compared to traditional routes. The mild basic conditions provided by sodium hydride, combined with the specific selectivity of the iron catalyst, minimize side reactions such as over-halogenation or polymerization of the sensitive isatin core. The reaction tolerates various substituents on the aryl rings, including alkyl, halogen, methoxy, and nitro groups, without significant degradation in yield. This broad functional group tolerance is essential for R&D directors who require diverse analog libraries for structure-activity relationship (SAR) studies. The ability to synthesize quinazolinone compounds with trifluoromethyl substitution at different positions according to actual needs enhances the practicability of the method, allowing for the rapid generation of high-purity OLED material precursors or pharmaceutical candidates with tailored electronic properties.
How to Synthesize 2-Trifluoromethyl Quinazolinones Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production. The process begins with the precise addition of ferric chloride and sodium hydride into a reaction vessel, followed by the introduction of 4A molecular sieves to maintain anhydrous conditions. Trifluoroethylimidoyl chloride and isatin are then added to an organic solvent, preferably DMF, which has been shown to effectively promote the reaction with high conversion rates. The mixture is stirred uniformly and subjected to a two-stage temperature profile: initially reacting at 40°C for 8-10 hours to facilitate the initial coupling, followed by heating to 120°C for 18-20 hours to drive the cyclization and decarbonylation to completion.
- Mix ferric chloride, sodium hydride, 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin in an organic solvent like DMF.
- React the mixture at 40°C for 8-10 hours, then heat to 120°C for 18-20 hours under air.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this iron-catalyzed methodology represents a strategic opportunity to optimize operational expenditures and mitigate supply risks. The shift away from expensive precious metal catalysts and specialized fluorinating agents towards commodity chemicals like ferric chloride and isatin fundamentally alters the cost structure of production. This change not only lowers the direct material costs but also simplifies the sourcing strategy, as these raw materials are widely available from multiple global suppliers, reducing dependency on single-source vendors. Furthermore, the simplified post-treatment procedure, which involves standard filtration and column chromatography, eliminates the need for complex purification steps often required to remove heavy metal residues, thereby reducing waste disposal costs and environmental compliance burdens.
- Cost Reduction in Manufacturing: The utilization of ferric chloride as a catalyst is a game-changer for cost efficiency, as iron is vastly cheaper and more abundant than palladium, rhodium, or other transition metals typically used in C-H activation or cross-coupling reactions. This substitution leads to substantial cost savings in the catalyst procurement budget. Additionally, the use of readily available aromatic amines and isatin derivatives as starting materials ensures that the raw material bill of materials (BOM) remains low and stable. The high reaction efficiency and yield reported in the patent examples mean that less raw material is wasted per kilogram of product produced, further enhancing the overall economic viability of the process for large-scale manufacturing.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals significantly enhances supply chain resilience. Unlike specialized fluorinated synthons that may have long lead times or limited suppliers, isatin and simple aromatic amines are produced in massive quantities globally. This abundance ensures that production schedules are less likely to be disrupted by raw material shortages. The robustness of the reaction conditions, which tolerate air and moisture to some extent (though molecular sieves are used), also reduces the risk of batch failures due to minor environmental fluctuations in the manufacturing plant. This reliability is crucial for maintaining continuous supply lines to downstream pharmaceutical customers who demand just-in-time delivery of critical intermediates.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, having been successfully demonstrated at the gram level with clear pathways to kilogram and tonne scales. The absence of toxic heavy metals in the catalyst system simplifies the regulatory approval process for the final API, as residual metal limits are easier to meet. Moreover, the simpler workup procedure generates less hazardous waste compared to traditional methods that might require aggressive acidic or basic quenches. This aligns with modern green chemistry principles and helps manufacturing facilities meet increasingly stringent environmental regulations regarding waste discharge and solvent recovery, making the process sustainable for long-term commercial operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, derived directly from the patent specifications and experimental data. Understanding these details is vital for technical teams evaluating the feasibility of adopting this route for their specific projects. The answers reflect the proven capabilities of the method as described in the intellectual property documentation, ensuring that expectations are aligned with the actual performance characteristics of the technology.
Q: What are the key advantages of the iron-catalyzed method over traditional synthesis?
A: The iron-catalyzed method utilizes cheap and abundant ferric chloride instead of expensive precious metals or harsh reagents like trifluoroacetic anhydride. It offers milder reaction conditions, broader substrate tolerance, and higher yields, significantly reducing production costs.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method can be expanded to the gram level and provides possibilities for industrial scale application due to the use of commercially available raw materials and simple post-treatment procedures like filtration and column chromatography.
Q: What is the role of the trifluoromethyl group in these compounds?
A: Introducing a trifluoromethyl group significantly improves the electronegativity, bioavailability, metabolic stability, and lipophilicity of the target molecule, making these quinazolinones highly valuable for developing anti-cancer and anti-inflammatory drugs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the iron-catalyzed synthesis route for 2-trifluoromethyl quinazolinones in accelerating drug discovery programs. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of volume. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate delivered meets the highest industry standards for pharmaceutical applications. We are committed to leveraging advanced synthetic methodologies like the one described in CN111675662B to provide our partners with a competitive edge in the market.
We invite you to collaborate with us to explore how this efficient synthesis route can benefit your specific pipeline. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your project requirements, demonstrating exactly how switching to this methodology can optimize your budget. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom synthesis needs. Let us help you secure a reliable supply of high-quality intermediates while maximizing your R&D efficiency.
