Revolutionizing Capecitabine Production: A Mild Catalytic Glycosylation Strategy for High-Purity Intermediates
Revolutionizing Capecitabine Production: A Mild Catalytic Glycosylation Strategy for High-Purity Intermediates
The global demand for effective antineoplastic agents continues to drive innovation in the synthesis of key pharmaceutical intermediates, particularly for widely used drugs like capecitabine. As the first fluoropyrimidine carbamate antitumor drug, capecitabine represents a critical component in oncology treatment regimens, necessitating robust and efficient manufacturing processes. Recent intellectual property developments, specifically patent CN111100172B, have introduced a groundbreaking methodology utilizing 5-deoxy-D-ribofuranose 1-[2-(1-styryl)benzoate] derivatives. This novel approach addresses long-standing challenges in nucleoside synthesis by employing a specialized glycosyl donor that enables high-selectivity coupling under remarkably mild conditions. The structural integrity and reactivity of these derivatives allow for the avoidance of traditional harsh reagents, marking a significant leap forward in process chemistry. By leveraging this advanced synthetic route, manufacturers can achieve superior purity profiles essential for regulatory compliance. 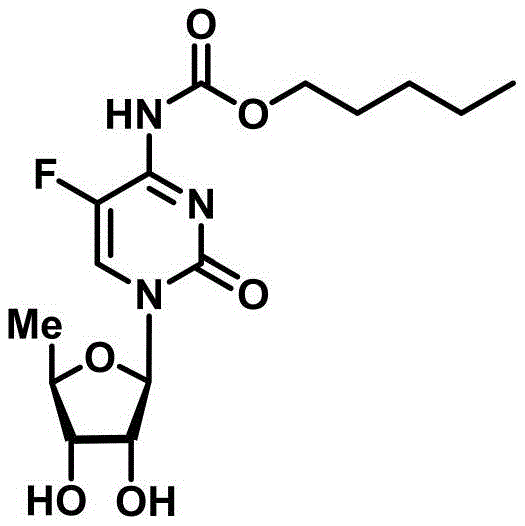
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthetic pathways for capecitabine and its precursors have been plagued by significant operational inefficiencies and chemical hazards. As detailed in the background art of the patent, existing literature describes at least four major synthetic routes, all of which suffer from inherent drawbacks such as the requirement for stoichiometric or excessive amounts of strong Lewis acids. These traditional conditions often lead to uncontrolled side reactions, resulting in complex impurity profiles that are difficult and costly to remove during downstream processing. Furthermore, the harsh reaction environments frequently degrade sensitive functional groups on the nucleobase or sugar moieties, drastically reducing overall yield and complicating scale-up efforts. The reliance on aggressive reagents also poses substantial safety risks in a commercial setting, requiring specialized equipment and rigorous waste management protocols. Consequently, the industry has faced persistent pressure to identify alternative strategies that mitigate these risks while maintaining economic viability.
The Novel Approach
In stark contrast to legacy methods, the technology disclosed in CN111100172B introduces a sophisticated glycosyl donor system based on 5-deoxy-D-ribofuranose 1-[2-(1-styryl)benzoate] derivatives. This innovative donor structure, represented by general formula (I), is uniquely designed to be activated under catalytic conditions rather than stoichiometric ones. The use of a catalytic amount of trimethylsilyl trifluoromethanesulfonate (TMSOTf) combined with N-iodosuccinimide (NIS) creates a reaction system that is exceptionally mild yet highly efficient. This shift eliminates the need for excessive Lewis acids, thereby preventing the degradation of the cytosine base and preserving the stereochemical integrity of the glycosidic bond. The result is a cleaner reaction profile with fewer byproducts, which translates directly into simplified purification workflows. ![General formula (I) of the novel 5-deoxy-D-ribofuranose 1-[2-(1-styryl)benzoate] derivative](/insights/img/capecitabine-intermediate-synthesis-glycosylation-pharma-supplier-20260308063842-06.png)
Mechanistic Insights into TMSOTf/NIS Catalyzed Glycosylation
The core of this technological breakthrough lies in the precise activation mechanism of the glycosyl donor. In the presence of the Lewis acid catalyst TMSOTf and the promoter NIS, the 1-[2-(1-styryl)benzoate] group facilitates the formation of a highly reactive oxocarbenium ion intermediate. Unlike traditional promoters that might require forcing conditions, this system operates effectively at temperatures ranging from 0°C to room temperature, demonstrating exceptional kinetic control. The catalytic cycle ensures that the activation energy barrier is lowered sufficiently to drive the coupling with the silylated fluorocytosine base without inducing thermal decomposition. This mechanistic elegance allows for the formation of the N-glycosidic bond with high regioselectivity and stereoselectivity, crucial for the biological activity of the final drug substance. The compatibility of this system with various protecting groups, such as benzoyl or acetyl moieties, further enhances its versatility across different synthetic contexts.
From an impurity control perspective, the mildness of the reaction conditions is paramount. Harsh acidic environments typically promote depurination or hydrolysis of the ester protecting groups, leading to a cascade of degradation products that complicate isolation. By maintaining a neutral to mildly acidic environment post-quench and avoiding extreme thermal stress, the novel process minimizes the generation of these critical quality attributes (CQAs). The patent data indicates that the reaction proceeds efficiently in anhydrous acetonitrile, a solvent choice that balances solubility with reaction rate. The subsequent workup involves simple quenching with triethylamine and standard extraction, avoiding complex aqueous washes that could hydrolyze the sensitive carbamate linkage. This robust control over the reaction trajectory ensures that the resulting nucleoside derivative meets stringent purity specifications required for pharmaceutical applications. 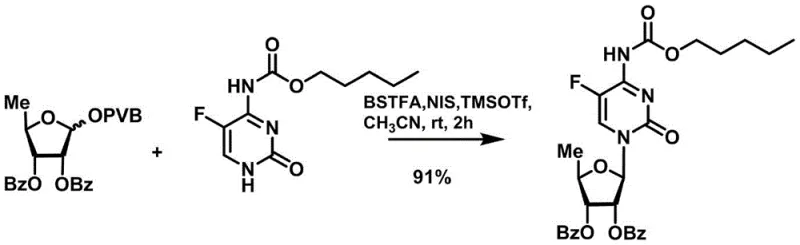
How to Synthesize Capecitabine Efficiently
The practical implementation of this synthesis involves a streamlined sequence that begins with the preparation of the specialized ribose donor, followed by the key glycosylation step, and concludes with a gentle deprotection. The initial preparation of the donor involves converting a protected ribose methyl glycoside into a thioglycoside, followed by oxidative desulfurization and esterification with 2-(1-styryl)benzoic acid. This multi-step precursor synthesis is highly optimized, yielding the active donor in high purity. Once the donor is secured, the coupling with the fluorocytosine base is performed under inert atmosphere to prevent moisture interference, which is critical for the success of the silylation and glycosylation steps. The final transformation involves the removal of the benzoyl protecting groups under basic conditions at low temperatures to preserve the carbamate functionality. For the complete standardized operating procedures and specific molar ratios, please refer to the detailed guide below.
- Prepare the 5-deoxy-D-ribofuranose 1-[2-(1-styryl)benzoate] donor by reacting the protected ribose with p-toluenethiol followed by esterification.
- Perform the glycosylation reaction using catalytic TMSOTf and NIS in anhydrous acetonitrile at 0°C to room temperature to couple with N4-oxycarbonyl-5-fluorocytosine.
- Execute the final deprotection step using sodium hydroxide in methanol at -15°C to yield the final capecitabine API with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers transformative benefits regarding cost structure and operational reliability. The shift from stoichiometric to catalytic reagent usage fundamentally alters the cost of goods sold (COGS) by reducing the consumption of expensive Lewis acids and simplifying the waste disposal burden associated with heavy metal or strong acid residues. This efficiency gain is not merely theoretical; the high yields reported in the patent examples, such as 91% for the glycosylation step and 96% for the final deprotection, indicate a process with minimal material loss. Such high efficiency directly correlates to reduced raw material procurement volumes and lower inventory holding costs. Furthermore, the mild reaction conditions reduce the energy load required for heating or cooling, contributing to a smaller carbon footprint and aligning with modern sustainability goals in chemical manufacturing.
- Cost Reduction in Manufacturing: The elimination of equivalent or excessive Lewis acids removes a significant cost center from the production budget. Traditional methods often require expensive scavengers to remove residual metals or acids, adding both material and time costs to the process. By utilizing a catalytic system, the downstream purification becomes significantly less resource-intensive, allowing for substantial cost savings in solvent usage and chromatography media. Additionally, the high selectivity reduces the formation of hard-to-remove impurities, which often require multiple recrystallization steps or preparative HPLC, further driving down processing expenses.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions enhances the predictability of production schedules. Harsh chemical processes are prone to batch failures due to exotherms or sensitivity to trace moisture, leading to unpredictable lead times. The mild nature of the TMSOTf/NIS system mitigates these risks, ensuring consistent batch-to-batch quality and reliable delivery timelines for downstream API manufacturers. Moreover, the reagents involved are commercially available and stable, reducing the risk of supply disruptions associated with specialty or hazardous chemicals that require strict transportation controls.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety hazards, particularly when dealing with strong acids and high temperatures. This novel route operates at near-ambient temperatures, significantly lowering the thermal risk profile and making it easier to transfer from pilot plant to commercial scale. The reduction in hazardous waste generation simplifies environmental compliance and lowers the cost of waste treatment. The ability to run the reaction in common solvents like acetonitrile and dichloromethane, which are well-understood in industrial recovery systems, further supports sustainable large-scale operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical benefits of the new method. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing frameworks. The answers highlight the specific advantages in terms of yield, purity, and operational safety that distinguish this approach from prior art.
Q: What are the limitations of conventional capecitabine synthesis routes?
A: Conventional methods often rely on stoichiometric or excessive amounts of harsh Lewis acids, leading to severe side reactions, difficult purification processes, and lower overall yields compared to the novel catalytic approach.
Q: How does the novel glycosyl donor improve reaction efficiency?
A: The 5-deoxy-D-ribofuranose 1-[2-(1-styryl)benzoate] derivative acts as a highly reactive glycosyl donor that can be activated under mild conditions using only catalytic amounts of TMSOTf and NIS, significantly reducing impurity formation.
Q: Is this synthesis route scalable for commercial manufacturing?
A: Yes, the process utilizes mild reaction temperatures ranging from -15°C to room temperature and achieves high yields (up to 96% in the final step), indicating excellent potential for large-scale commercial production with reduced safety risks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Capecitabine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries like the one described in CN111100172B can be successfully translated into industrial reality. We are committed to delivering high-purity capecitabine intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our infrastructure is designed to handle complex nucleoside chemistry with the utmost precision, guaranteeing supply continuity for our partners.
We invite you to collaborate with us to leverage this cutting-edge technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how this mild catalytic process can optimize your production economics. Let us partner with you to secure a reliable, cost-effective, and high-quality supply of these vital pharmaceutical intermediates.
