Advanced Pyridine-Free Synthesis of Capecitabine Intermediates for Commercial Scale-up
Advanced Pyridine-Free Synthesis of Capecitabine Intermediates for Commercial Scale-up
The pharmaceutical industry continuously seeks robust manufacturing processes that balance high purity with environmental sustainability, particularly for potent oncology agents like Capecitabine. A pivotal advancement in this domain is detailed in patent CN106478751B, which discloses a novel preparation method for 2',3'-di-O-acetyl-5'-deoxy-5-fluoro-N4-[(pentyloxy)carbonyl]cytosine nucleoside, a critical intermediate in the Capecitabine value chain. This technology addresses long-standing challenges associated with traditional acylation methods by substituting toxic organic bases with inorganic alternatives, thereby streamlining the purification workflow. As a reliable pharmaceutical intermediates supplier, understanding these mechanistic shifts is essential for ensuring the consistent delivery of high-quality active pharmaceutical ingredients (APIs). The structural complexity of Capecitabine requires precise stereochemical control and rigorous impurity management, which this patented route facilitates through optimized reaction conditions.
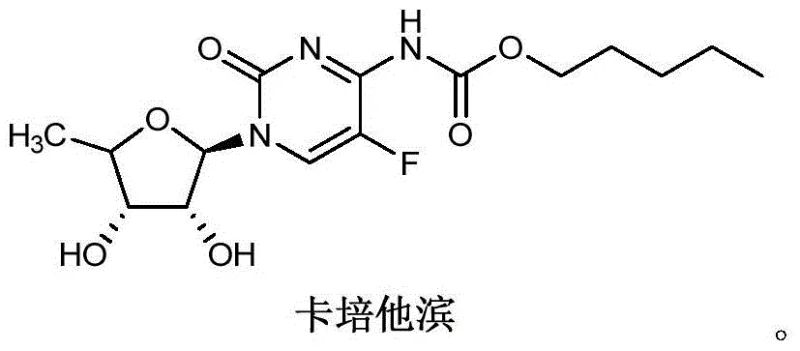
The significance of this innovation extends beyond mere academic interest; it represents a tangible opportunity for cost reduction in pharmaceutical intermediates manufacturing. By eliminating the reliance on pyridine, a solvent notorious for its difficult removal and environmental impact, manufacturers can significantly reduce downstream processing costs. The patent outlines a comprehensive synthetic strategy that begins with the formation of the nucleoside backbone and proceeds through a highly efficient acylation step. This approach not only enhances the overall yield but also ensures that the final product meets stringent regulatory specifications for residual solvents. For procurement teams evaluating supply chain resilience, adopting such advanced synthetic methodologies translates to reduced risk of batch failures and more predictable production timelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the key carbamate intermediate (Compound V) has relied heavily on protocols described in literature such as Bioorganic & Medicinal Chemistry, which utilize pyridine as both a solvent and a condensing agent. While chemically effective, this conventional approach presents severe drawbacks for modern industrial application. Pyridine possesses high toxicity and a distinct, persistent odor, creating significant occupational health hazards for plant operators. Furthermore, its high solubility in both organic and aqueous phases complicates the extraction process, often necessitating multiple washing steps that increase solvent consumption and wastewater volume. The difficulty in completely removing pyridine residues poses a critical quality risk, as even trace amounts can persist into the final API, requiring extensive and costly purification efforts to meet pharmacopoeial standards.
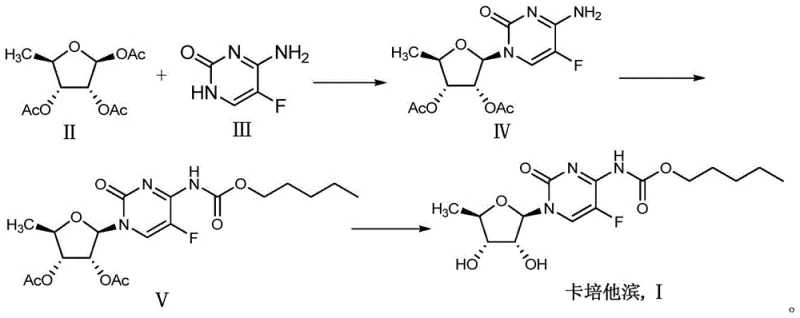
From an environmental compliance perspective, the disposal of pyridine-laden waste streams is increasingly problematic due to tightening global regulations on volatile organic compounds (VOCs). Traditional methods often require specialized incineration or complex treatment protocols to neutralize the basic nitrogenous waste, adding substantial overhead to the manufacturing cost structure. Additionally, the use of pyridine can sometimes lead to the formation of unwanted by-products or adducts that are structurally similar to the target molecule, making chromatographic separation challenging and reducing the overall mass balance of the process. These factors collectively render the conventional pyridine-based route less attractive for large-scale commercial production where efficiency and safety are paramount.
The Novel Approach
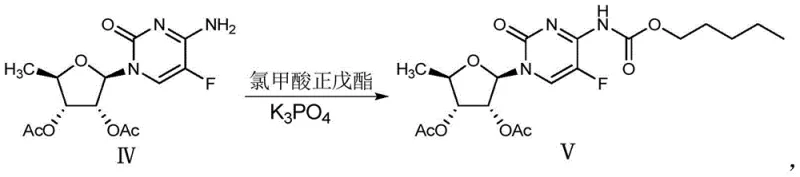
In contrast, the methodology disclosed in patent CN106478751B introduces a paradigm shift by employing potassium phosphate (K3PO4) as the primary condensing agent. This inorganic base offers a superior profile for the acylation of the cytosine nucleoside derivative with n-amyl chloroformate. The reaction proceeds under mild conditions, typically between 0°C and 60°C, which minimizes thermal degradation of the sensitive nucleoside scaffold. By avoiding strong organic bases, the process inherently reduces the formation of side products, leading to a crude reaction mixture with significantly higher purity. This improvement simplifies the workup procedure, often allowing for direct crystallization or simple extraction without the need for complex chromatography.
The operational advantages of this novel approach are manifold. The use of a mixed solvent system, preferably isopropanol and dichloromethane, provides an optimal balance of solubility for the reactants while facilitating easy phase separation during workup. The inorganic nature of K3PO4 ensures that it remains largely insoluble in the organic phase or can be easily washed away with aqueous solutions, leaving no toxic residues in the product. This characteristic is crucial for maintaining the integrity of the supply chain, as it eliminates the need for specialized solvent recovery units dedicated to pyridine. Consequently, this route supports the commercial scale-up of complex pharmaceutical intermediates by offering a safer, cleaner, and more economically viable pathway to the final drug substance.
Mechanistic Insights into K3PO4-Catalyzed Acylation
The core chemical transformation in this process involves the nucleophilic attack of the exocyclic amine at the N4 position of the cytosine ring onto the carbonyl carbon of n-amyl chloroformate. In the absence of a base, this reaction would generate hydrochloric acid as a by-product, which could protonate the amine and halt the reaction or degrade the acid-sensitive glycosidic bond. Potassium phosphate acts as a non-nucleophilic base that effectively scavenges the generated HCl, driving the equilibrium towards the formation of the carbamate bond. Unlike organic amines, which can compete as nucleophiles and form urea by-products, the phosphate anion is sterically hindered and electronically stable, ensuring high chemoselectivity for the desired N-acylation.
Furthermore, the choice of K3PO4 influences the impurity profile of the reaction. The patent data suggests that the inorganic base promotes a cleaner reaction trajectory, minimizing the formation of O-acylated species or bis-acylated impurities that are common when using more reactive organic bases. The reaction kinetics are optimized by controlling the addition rate of the chloroformate at low temperatures (0-10°C), which prevents local exotherms that could trigger decomposition. Following the addition, the reaction is allowed to warm to ambient temperature (20-25°C) to ensure complete conversion of the starting material. This precise control over reaction parameters results in a product with HPLC purity often exceeding 99%, demonstrating the robustness of the mechanistic design.
How to Synthesize Capecitabine Efficiently
Implementing this synthetic route requires careful attention to stoichiometry and solvent selection to maximize efficiency. The process generally involves three distinct stages: the initial glycosylation to form the protected nucleoside, the critical acylation step using the K3PO4 protocol, and the final deprotection to reveal the active drug. Detailed operational parameters, such as the molar ratio of Compound IV to K3PO4 (preferably 1.0:1.5) and the specific solvent volumes, are critical for reproducibility. Manufacturers should note that the use of dry solvents and inert atmosphere conditions during the initial steps helps prevent hydrolysis of the chloroformate reagent. For a comprehensive guide on executing this synthesis with industrial precision, please refer to the standardized protocol below.
- React 5-fluorocytosine with hexamethyldisilazane and tin tetrachloride to form the silylated intermediate, followed by coupling with the sugar moiety to yield Compound IV.
- Treat Compound IV with n-amyl chloroformate in the presence of potassium phosphate (K3PO4) in a mixed solvent system of isopropanol and dichloromethane at 0-25°C to obtain Compound V.
- Deprotect Compound V using sodium hydroxide in methanol, followed by acidification and recrystallization from ethyl acetate and n-hexane to isolate pure Capecitabine.
Commercial Advantages for Procurement and Supply Chain Teams
For strategic decision-makers in the pharmaceutical supply chain, the adoption of this pyridine-free methodology offers compelling economic and operational benefits. The primary advantage lies in the simplification of the purification process, which directly correlates to reduced manufacturing cycle times and lower utility consumption. By eliminating the need for extensive distillation or chromatographic purification to remove pyridine, facilities can increase their throughput capacity without significant capital investment. This efficiency gain is particularly valuable in the context of reducing lead time for high-purity pharmaceutical intermediates, allowing suppliers to respond more agilely to market demand fluctuations.
- Cost Reduction in Manufacturing: The substitution of pyridine with potassium phosphate eliminates the costs associated with handling, storing, and disposing of a hazardous toxic solvent. Since K3PO4 is an inexpensive, commercially available inorganic salt, the raw material cost for the condensing agent is drastically lower compared to high-purity organic bases. Moreover, the simplified workup reduces the volume of organic solvents required for extraction and washing, leading to substantial savings in solvent procurement and waste treatment fees. The higher yield reported in the patent examples further amplifies these savings by maximizing the output per unit of expensive starting materials.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like potassium phosphate reduces dependency on specialized reagent suppliers who may face production bottlenecks. Inorganic salts have a stable and abundant global supply, ensuring continuity of operations even during market disruptions. Additionally, the improved safety profile of the process reduces the regulatory burden and insurance costs associated with handling toxic substances. This stability allows procurement managers to negotiate more favorable long-term contracts and secure a steady flow of materials for API production without the risk of sudden supply interruptions.
- Scalability and Environmental Compliance: The mild reaction conditions and aqueous-compatible workup make this process inherently scalable from pilot plant to multi-ton commercial production. The absence of pyridine in the waste stream simplifies effluent treatment, helping manufacturing sites meet increasingly strict environmental discharge limits. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate sustainability profile of the manufacturer. The ability to scale this process efficiently ensures that the supply of Capecitabine can meet global oncology demands without compromising on quality or environmental stewardship.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the technology's viability. Understanding these nuances is vital for R&D teams planning technology transfer and for quality assurance personnel establishing control strategies.
Q: Why is replacing pyridine critical in Capecitabine intermediate synthesis?
A: Pyridine is toxic and difficult to remove completely, leading to potential residue in the final API which poses safety risks. Additionally, pyridine is miscible with water, complicating wastewater treatment and increasing environmental compliance costs.
Q: What are the yield improvements observed with the K3PO4 method?
A: The patent data indicates that using potassium phosphate as a condensing agent can achieve yields up to 94.2% with HPLC purity exceeding 99%, significantly outperforming traditional amine bases which often result in lower yields and complex purification profiles.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the reaction conditions are mild (0-60°C), utilize commercially available inorganic bases, and avoid hazardous solvents or catalysts, making it highly scalable and compliant with modern GMP and environmental standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Capecitabine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic routes to maintain competitiveness in the global pharmaceutical market. Our technical team has extensively analyzed the pyridine-free methodology described in patent CN106478751B and validated its potential for robust commercial manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory successes are seamlessly translated into industrial reality. Our facilities are equipped with rigorous QC labs capable of detecting trace impurities, guaranteeing that every batch of Capecitabine intermediate meets stringent purity specifications required by international regulatory bodies.
We invite procurement leaders and R&D directors to collaborate with us on optimizing their supply chains for oncology APIs. By leveraging our expertise in nucleoside chemistry, we can help you implement this cost-effective and environmentally friendly synthesis route. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production volumes. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can enhance your project's success and accelerate your time to market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
