Revolutionizing 1,3-Enyne Synthesis: Copper-Catalyzed Process for Commercial-Scale Pharmaceutical Intermediate Production
The patent CN1946670A introduces a groundbreaking copper(I)-catalyzed methodology for synthesizing 1,3-enynes, representing a significant advancement in the field of pharmaceutical intermediate production. This innovative approach addresses critical limitations in conventional palladium-catalyzed Sonogashira coupling reactions by eliminating the need for expensive palladium reagents while maintaining high stereoselectivity and functional group tolerance. The technology enables the production of biologically active compounds such as Terbinafine (Lamisil®) and Calicheamicin γ1, which contain essential 1,3-enyne moieties for their therapeutic properties. By leveraging copper(I) bidentate ligand complexes at catalytic concentrations (1.0-12.0 mol%), this method achieves yields up to 99% while operating under industrially feasible conditions of toluene solvent at 110°C with potassium carbonate as base. The process demonstrates remarkable versatility across diverse substrate combinations, making it particularly valuable for pharmaceutical manufacturers seeking reliable routes to complex intermediates without the cost volatility associated with palladium-based systems.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing 1,3-enynes predominantly rely on palladium-copper catalyzed Sonogashira coupling reactions between acetylenic and vinyl halides, which present significant challenges for industrial-scale pharmaceutical manufacturing. The most pressing issue is the dramatic increase in palladium prices—approximately 900% in recent years—which has severely impacted cost structures and supply chain stability for pharmaceutical intermediates production. Additionally, palladium-based systems often require expensive ligands specifically designed to enhance reactivity, further escalating production costs. Conventional approaches also suffer from poor functional group tolerance, necessitating extensive protection/deprotection strategies that add complexity and reduce overall process efficiency. Many existing methods require stoichiometric amounts of copper reagents or specialized conditions that limit scalability, while others produce unwanted byproducts that complicate purification and reduce final product purity. The need for rigorous palladium removal steps adds additional processing time and cost, creating bottlenecks in commercial manufacturing workflows where high-purity API intermediates are required.
The Novel Approach
The copper(I)-catalyzed method described in patent CN1946670A overcomes these limitations by utilizing catalytic concentrations of copper(I) bidentate ligand complexes that enable efficient cross-coupling between acetylenic compounds and vinyl iodides without palladium assistance. This innovative system maintains complete vinyl stereochemistry while accommodating a wide range of functional groups that would typically be incompatible with other catalytic approaches, including base-labile methyl ketones and methyl esters. The process operates effectively under industrially practical conditions using common solvents like toluene at moderate temperatures (110°C), with reaction times as short as 8 hours for many substrates. By eliminating the need for expensive palladium catalysts and their associated ligands, this method significantly reduces raw material costs while simplifying downstream processing through the elimination of metal removal steps. The catalyst system demonstrates exceptional versatility across diverse substrate combinations, including both electron-rich and electron-poor aryl acetylenes, sterically hindered substrates, and heterocyclic compounds, making it broadly applicable to pharmaceutical intermediate manufacturing.
Mechanistic Insights into Copper(I)-Catalyzed Cross-Coupling
The copper(I)-catalyzed cross-coupling mechanism operates through a well-defined catalytic cycle that begins with oxidative addition of the vinyl iodide to the copper(I) center, forming a copper(III) intermediate. This key step is facilitated by the bidentate ligand system (such as phenanthroline or bipyridine derivatives), which stabilizes the copper center throughout the catalytic cycle while maintaining optimal geometry for substrate coordination. The acetylenic compound then coordinates to the metal center before undergoing transmetalation, followed by reductive elimination to form the desired C-C bond and regenerate the active copper(I) catalyst. This mechanism operates efficiently at catalytic concentrations (typically 10 mol%) without requiring stoichiometric amounts of copper reagents, representing a significant improvement over previous copper-mediated approaches. The bidentate ligand system plays a crucial role in preventing catalyst decomposition and maintaining high turnover numbers, which is essential for commercial-scale implementation where catalyst efficiency directly impacts process economics.
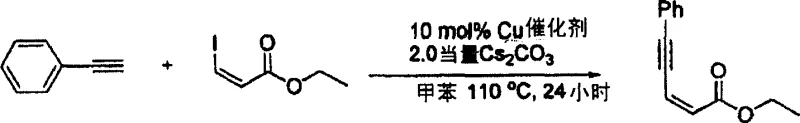
The process demonstrates exceptional control over impurity formation through several key mechanisms that contribute to high product purity essential for pharmaceutical applications. First, the selective activation of vinyl iodides over other potential reaction pathways minimizes side reactions that could generate impurities. Second, the mild reaction conditions (toluene at 110°C) prevent thermal degradation of sensitive functional groups that might otherwise lead to impurity formation. Third, the absence of palladium eliminates potential metal contamination concerns that require additional purification steps in traditional processes. The catalyst system maintains excellent stereoselectivity throughout the reaction sequence, preserving the geometric configuration of the starting vinyl iodide in the final product without isomerization—a critical factor for pharmaceutical intermediates where stereochemistry directly impacts biological activity. This precise control over reaction pathways results in cleaner reaction profiles that simplify downstream purification while ensuring stringent purity specifications required for API intermediates.
How to Synthesize 1,3-Enyne Efficiently
This copper-catalyzed synthesis represents a significant advancement over traditional palladium-based methods for producing high-purity pharmaceutical intermediates. The process has been optimized through extensive experimentation to achieve maximum efficiency while maintaining operational simplicity suitable for commercial scale-up. Detailed standardized synthesis steps are provided below to ensure consistent results across different manufacturing environments.
- Prepare reaction medium containing copper(I) bidentate ligand complex (10 mol%) and appropriate base (2.0 equivalents) in toluene under inert atmosphere
- Add acetylenic compound (2.00 mmol) and vinyl iodide (2.20 mmol) to the reaction mixture while maintaining temperature control
- Stir the reaction mixture at 110°C for 8 hours, monitoring progress through analytical methods to ensure complete conversion
Commercial Advantages for Procurement and Supply Chain Teams
This innovative copper-catalyzed process addresses multiple pain points in pharmaceutical intermediate procurement by offering a more sustainable and economically viable alternative to traditional palladium-dependent methodologies. The elimination of palladium dependency removes a significant source of cost volatility and supply chain vulnerability that has plagued pharmaceutical manufacturers in recent years.
- Cost Reduction in Manufacturing: The elimination of expensive palladium catalysts and their specialized ligands represents substantial cost savings in raw material expenses. By avoiding the need for subsequent palladium removal steps required in traditional processes, this method significantly reduces processing time and associated costs while eliminating potential yield losses during metal purification stages. The use of readily available copper-based catalysts at catalytic concentrations provides a more economically sustainable approach to producing high-value pharmaceutical intermediates without compromising on quality or yield.
- Enhanced Supply Chain Reliability: Copper-based catalysts offer superior supply chain stability compared to palladium due to more consistent global availability and reduced market volatility. The process utilizes common solvents and reagents that are widely accessible from multiple suppliers worldwide, minimizing single-source dependencies that can disrupt production schedules. This enhanced supply chain resilience ensures more predictable lead times for high-purity pharmaceutical intermediates while reducing the risk of production delays caused by raw material shortages or price spikes.
- Scalability and Environmental Compliance: The reaction conditions are inherently scalable from laboratory to commercial production volumes without requiring significant process modifications, making it ideal for technology transfer across different manufacturing scales from 100 kgs to multi-ton annual capacity. The elimination of toxic heavy metals reduces environmental impact while simplifying waste treatment protocols, aligning with increasingly stringent environmental regulations governing pharmaceutical manufacturing processes worldwide.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this innovative copper-catalyzed synthesis method for pharmaceutical intermediate production. These answers are based on extensive experimental data from patent CN1946670A and reflect practical considerations for industrial adoption.
Q: How does this copper-catalyzed method improve upon traditional palladium-based approaches for enyne synthesis?
A: The copper(I)-catalyzed method eliminates the need for expensive palladium catalysts, which have seen approximately 900% price increases in recent years. This approach maintains excellent functional group tolerance while avoiding the need for costly palladium-specific ligands and subsequent metal removal steps required in traditional processes.
Q: What are the key advantages of this process for pharmaceutical intermediate manufacturing?
A: This method provides high stereoselectivity while accommodating a wide range of functional groups that would typically be incompatible with other catalytic systems. The process demonstrates exceptional yield consistency across diverse substrate combinations, with the ability to maintain vinyl stereochemistry throughout the reaction sequence.
Q: How does this technology support supply chain reliability for pharmaceutical manufacturers?
A: The elimination of palladium dependency removes a significant supply chain vulnerability, as copper-based catalysts are more readily available and less subject to market fluctuations. The process utilizes common solvents and reagents that are widely accessible at industrial scale, enhancing production continuity and reducing lead times.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3-Enyne Supplier
Our proprietary copper-catalyzed synthesis technology represents a significant advancement in producing high-purity pharmaceutical intermediates with exceptional efficiency and reliability. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art QC labs and rigorous analytical protocols. Our CDMO expertise ensures seamless technology transfer from laboratory development to full-scale manufacturing, with dedicated process chemistry teams optimizing every aspect of production to meet your specific quality requirements and regulatory standards.
We invite you to request a Customized Cost-Saving Analysis tailored to your specific intermediate requirements by contacting our technical procurement team directly. They will provide detailed information including specific COA data and comprehensive route feasibility assessments to help you evaluate how our innovative copper-catalyzed process can enhance your supply chain reliability while delivering substantial cost benefits compared to traditional palladium-based approaches.
