Revolutionizing Exatecan Intermediate Production: A Deep Dive into Microchannel Nitration Technology
Revolutionizing Exatecan Intermediate Production: A Deep Dive into Microchannel Nitration Technology
The landscape of pharmaceutical intermediate manufacturing is undergoing a transformative shift driven by the need for safer, more efficient, and scalable synthetic routes. A pivotal development in this arena is detailed in patent CN115701419A, which discloses a novel preparation method for Exatecan intermediates, specifically focusing on the synthesis of 1-nitro-3-fluoro-4-methyl-8-oxo-5,6,7,8-tetrahydronaphthalene. Exatecan itself is a potent DNA topoisomerase I inhibitor and a water-soluble camptothecin derivative that has gained immense strategic importance as the cytotoxic payload in next-generation Antibody-Drug Conjugates (ADCs) like Trastuzumab deruxtecan (DS-8201). As global demand for high-purity ADC payloads surges, the ability to produce key precursors reliably becomes a critical supply chain bottleneck. This patent introduces a groundbreaking approach utilizing microchannel reactor technology to execute a hazardous nitration reaction, thereby addressing long-standing safety concerns and efficiency limitations inherent in traditional batch processing methods.
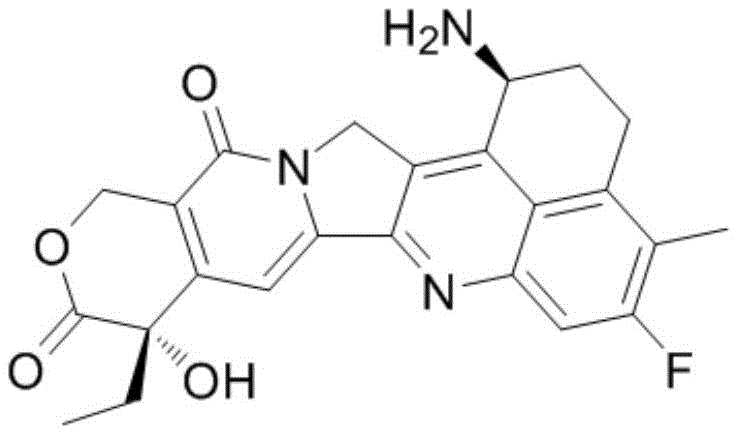
The strategic value of this technology extends beyond mere chemical transformation; it represents a fundamental upgrade in process safety and environmental compliance. By transitioning from conventional batch reactors to continuous flow microchannel systems, manufacturers can achieve intrinsic safety through superior heat management and precise control over reaction parameters. This is particularly vital for nitration reactions, which are notoriously exothermic and prone to generating unstable by-products. For R&D directors and process chemists, understanding the nuances of this flow chemistry approach is essential for designing robust manufacturing processes that can scale from kilogram to multi-ton production without compromising on purity or safety standards. The implications for cost reduction in pharmaceutical manufacturing are profound, as improved yields and reduced waste directly correlate to lower cost of goods sold (COGS).
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
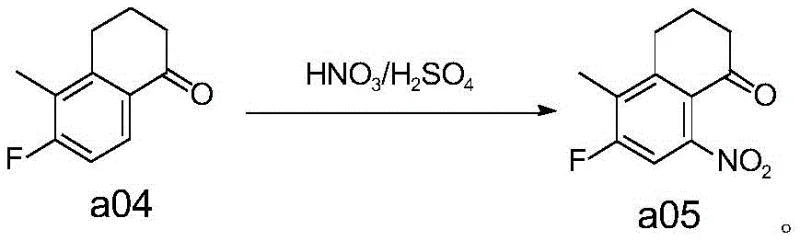
Historically, the synthesis of compound a07, a critical precursor for Exatecan, has been plagued by inefficient and convoluted synthetic pathways. Early methodologies, such as those disclosed in EP0495432B1, relied on a series of repetitive and atom-uneconomical steps including decarbonylation followed by oxidation and re-carbonylation. These legacy routes are characterized by extremely low atom utilization and dismal overall yields, with some reports indicating total yields as low as 3.69 percent. Such inefficiencies are unacceptable in modern commercial manufacturing, where resource optimization is paramount. Furthermore, alternative routes described in documents like WO1996026181A1 and CN111065621A involve complex sequences of ring-closing, ring-opening, oxidation, and reduction reactions. These multi-step processes not only increase the operational complexity and equipment requirements but also accumulate impurities at each stage, making downstream purification increasingly difficult and costly.
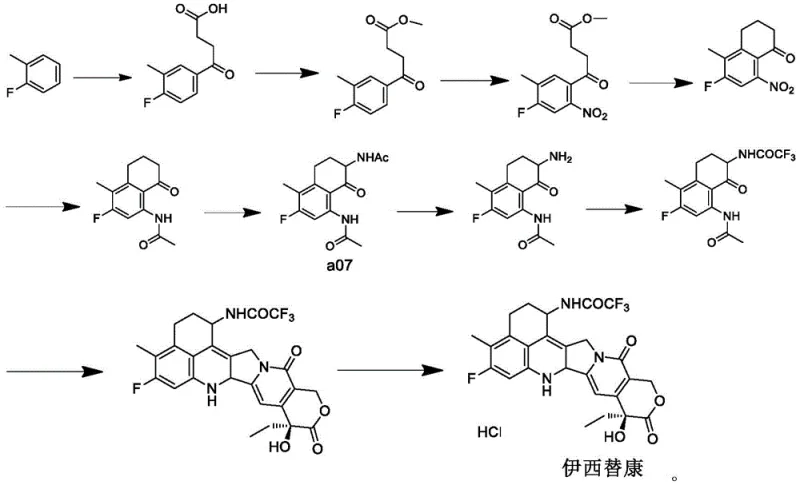
Beyond the economic drawbacks, conventional nitration methods pose severe safety risks that threaten supply chain continuity. Traditional batch nitration often suffers from poor heat dissipation, leading to hot spots that can trigger thermal runaways or even explosions. The formation of polynitro compounds as by-products further exacerbates this instability, as these species can undergo secondary decomposition at elevated temperatures. For supply chain heads, relying on such hazardous processes introduces significant liability and potential for unplanned shutdowns. Additionally, some optimized routes like those in WO2019044946A1, while attempting to improve the synthesis, still rely on expensive starting materials with limited market availability and suffer from low yields in initial bromination steps (around 30 percent). These factors collectively create a fragile supply base that struggles to meet the rigorous demands of the global oncology market.
The Novel Approach
The methodology presented in CN115701419A offers a paradigm shift by leveraging continuous flow chemistry to overcome these historical hurdles. The core innovation lies in the direct nitration of 3-fluoro-4-methyl-8-oxo-5,6,7,8-tetrahydronaphthalene using a mixed acid system within a microchannel reactor. This approach eliminates the need for the tedious decarbonylation-oxidation cycles found in prior art, drastically shortening the synthetic route. By utilizing a microchannel reactor, such as the Corning G1 glass reactor system, the process achieves exceptional mixing efficiency and heat transfer rates. This allows for the precise control of reaction temperature between 25 to 120 degrees Celsius and residence times as short as 30 to 180 seconds. Such tight control prevents the accumulation of heat and suppresses the formation of dangerous polynitro by-products, effectively mitigating the explosion risks associated with batch nitration.
Furthermore, this novel route utilizes readily available starting materials, enhancing the reliability of the supply chain compared to methods requiring scarce or expensive reagents. The streamlined process not only simplifies post-treatment steps—often requiring only separation, washing, and concentration—but also significantly boosts the total yield of the final Exatecan product. For procurement managers, this translates to a more stable and cost-effective sourcing strategy. The ability to run the reaction continuously with high throughput means that commercial scale-up of complex pharmaceutical intermediates becomes far more feasible. The reduction in acid consumption and waste liquid discharge also aligns with increasingly stringent environmental regulations, positioning this technology as a sustainable choice for long-term manufacturing partnerships.
Mechanistic Insights into Microchannel Nitration
The success of this synthetic strategy hinges on the unique physicochemical environment created within the microchannel reactor. Unlike batch vessels where mixing is dependent on agitation and heat removal relies on jacket cooling, microchannels operate on the principle of laminar flow with very high surface-area-to-volume ratios. When the organic solution of the substrate and the mixed acid (comprising nitric acid with mass fraction not lower than 90 percent and sulfuric acid with mass fraction not lower than 70 percent) meet in the microchannels, they form a highly uniform mixture almost instantaneously. This rapid mixing ensures that the concentration of the nitrating agent is consistent throughout the reaction zone, preventing local excesses that could lead to over-nitration or degradation. The molar ratio of nitric acid to the substrate is tightly controlled, typically between 1 to 1.2:1, ensuring stoichiometric efficiency while minimizing waste.
Thermal management is perhaps the most critical mechanistic advantage. Nitration is highly exothermic, and in a batch setting, the heat generated can outpace the cooling capacity, leading to temperature spikes. In the microchannel system, the small channel dimensions allow for near-instantaneous heat exchange with the reactor walls, maintaining the reaction strictly within the optimal temperature window of 30 to 100 degrees Celsius. This isothermal condition is vital for suppressing side reactions and ensuring the stability of the nitro intermediate. Moreover, the short residence time (preferably 50 to 150 seconds) means that the product is quickly moved away from the reactive environment once formed, preventing further decomposition or secondary reactions. This kinetic control is what allows the process to achieve high conversion rates safely, turning a potentially hazardous batch operation into a controlled, continuous manufacturing asset.
How to Synthesize 1-Nitro-3-fluoro-4-methyl-8-oxo-5,6,7,8-tetrahydronaphthalene Efficiently
Implementing this advanced synthesis requires a disciplined approach to process parameters and equipment setup. The procedure begins with the careful preparation of the reaction feed streams. The substrate, 3-fluoro-4-methyl-8-oxo-5,6,7,8-tetrahydronaphthalene, must be fully dissolved in a suitable organic solvent such as dichloromethane, 1,2-dichloroethane, or chloroform, with a solvent-to-substrate volume-to-weight ratio preferably between 8 to 15:1. Simultaneously, the mixed acid is prepared by slowly adding concentrated nitric acid into concentrated sulfuric acid under stirring to ensure homogeneity and manage the heat of mixing. Once the feeds are ready, they are introduced into the microchannel reactor using precision metering pumps. The flow rates are calibrated to achieve the target residence time, which is the key variable determining conversion and selectivity. Following the reaction, the effluent is quenched in cold water to stop the reaction immediately, followed by standard workup procedures including phase separation and washing with sodium bicarbonate to neutralize residual acids.
- Prepare the reaction solution by dissolving 3-fluoro-4-methyl-8-oxo-5,6,7,8-tetrahydronaphthalene in an organic solvent such as dichloromethane or chloroform.
- Generate the mixed acid solution by slowly adding high-concentration nitric acid into concentrated sulfuric acid under stirring conditions.
- Pump both solutions into a microchannel reactor (e.g., Corning G1) controlling flow rates to maintain a residence time between 30 to 180 seconds at temperatures ranging from 25 to 120°C.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for the bottom line and supply continuity, the adoption of this microchannel nitration technology offers compelling strategic advantages. The primary benefit lies in the drastic simplification of the manufacturing process, which directly correlates to substantial cost savings. By eliminating multiple steps such as decarbonylation and repeated oxidation-reduction cycles, the new route reduces the consumption of reagents, solvents, and energy. Fewer unit operations mean less equipment occupancy time and lower labor costs per kilogram of product. Additionally, the significant improvement in overall yield means that less raw material is required to produce the same amount of final API, effectively lowering the material cost basis. This efficiency gain is critical in a competitive market where margin compression is a constant challenge for generic and specialty chemical manufacturers.
- Cost Reduction in Manufacturing: The transition to a continuous flow process eliminates the need for large batch reactors and the associated infrastructure for handling hazardous exothermic reactions on a large scale. The reduction in acid usage and the minimization of waste liquid discharge lower the costs associated with waste treatment and environmental compliance. Furthermore, the higher purity of the crude product reduces the burden on downstream purification steps like chromatography or recrystallization, saving both time and expensive stationary phases. These cumulative efficiencies result in a leaner, more cost-effective production model that enhances competitiveness without sacrificing quality.
- Enhanced Supply Chain Reliability: Relying on readily available starting materials rather than scarce or custom-synthesized precursors mitigates the risk of supply disruptions. The robustness of the microchannel process ensures consistent product quality batch after batch, reducing the likelihood of failed runs that can delay shipments. The inherent safety of the process also means fewer regulatory hurdles and lower insurance premiums, contributing to a more stable and predictable supply chain. For procurement managers, this reliability translates into the ability to secure long-term contracts with confidence, knowing that the supplier has a resilient and scalable manufacturing platform.
- Scalability and Environmental Compliance: One of the greatest strengths of microchannel technology is its linear scalability. Increasing production capacity often involves numbering up (adding more reactor modules) rather than scaling up (increasing vessel size), which preserves the optimized reaction conditions achieved at the lab scale. This facilitates a smoother transition from pilot to commercial production, reducing lead time for high-purity pharmaceutical intermediates. Moreover, the reduced solvent and acid consumption aligns with green chemistry principles, helping companies meet their sustainability goals and comply with increasingly strict environmental regulations regarding hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and process descriptions provided in the patent literature, offering a clear perspective on the feasibility and benefits of the technology. Understanding these details is crucial for technical teams evaluating the potential for technology transfer or licensing.
Q: Why is microchannel technology preferred for the nitration step in Exatecan synthesis?
A: Conventional batch nitration poses significant explosion risks due to heat accumulation and the formation of unstable polynitro by-products. Microchannel reactors provide superior heat exchange and precise residence time control, mitigating these safety hazards while improving reaction efficiency.
Q: How does the new route compare to previous methods in terms of yield?
A: Prior art methods, such as those involving repeated decarbonylation and oxidation cycles, suffered from extremely low total yields (reported as low as 3.69%). The novel microchannel nitration route significantly shortens the synthetic sequence and drastically improves the overall yield of the key intermediate compound a07.
Q: What are the critical reaction parameters for the nitration step?
A: The process requires precise control of the mixed acid ratio (nitric to sulfuric acid weight ratio of 1:1.5 to 1:10) and a strict residence time window of 30 to 180 seconds within the microchannel reactor to ensure complete conversion without thermal runaway.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Exatecan Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development and commercialization of life-saving oncology therapies. Our team of expert process chemists has extensively evaluated the microchannel nitration technology described in CN115701419A and is fully equipped to implement this advanced methodology at scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are designed to handle complex continuous flow reactions safely, adhering to stringent purity specifications and operating within rigorous QC labs to guarantee product integrity. We understand that in the fast-paced world of ADC development, time-to-market is everything, and our agile manufacturing capabilities are tailored to accelerate your project timelines.
We invite you to collaborate with us to leverage this cutting-edge synthesis route for your Exatecan supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this continuous flow process for your operations. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your project requirements. Let us help you optimize your supply chain with a reliable, safe, and cost-effective solution for Exatecan intermediates, ensuring your pipeline remains robust and competitive in the global marketplace.
