Advanced Catalytic Synthesis of Losartan Intermediates for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic routes for high-volume antihypertensive agents, and patent CN1915990B presents a transformative approach to the manufacture of Losartan. This intellectual property discloses a novel methodology that fundamentally alters the construction of the critical imidazole-biphenyl scaffold, specifically targeting the elimination of cumbersome protection-deprotection sequences that have historically plagued this synthesis. By leveraging a strategic combination of selective reduction and catalytic tetrazole formation, the inventors have established a pathway that not only simplifies the operational workflow but also enhances the overall safety profile by optimizing the usage of hazardous reagents like sodium azide. For R&D directors and process chemists, this represents a significant opportunity to refine impurity profiles and streamline the production of this essential API intermediate. The core innovation lies in the flexibility to either reduce the aldehyde precursor prior to alkylation or post-alkylation, offering process engineers valuable options for optimizing throughput and yield in a commercial setting.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Losartan has been burdened by inefficient multi-step sequences that rely heavily on the protection of reactive functional groups to prevent side reactions. As illustrated in prior art such as US5138069A, traditional routes often necessitate the protection of the hydroxymethyl group on the imidazole ring, typically using bulky protecting groups like trityl, before proceeding to the tetrazole formation step. 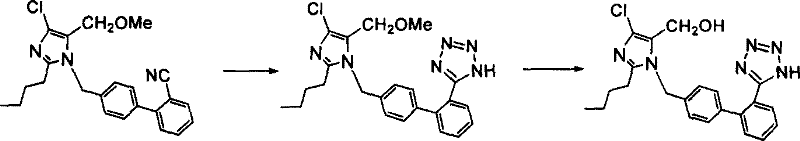 This reliance on protection chemistry introduces significant inefficiencies, including the consumption of additional reagents, the generation of stoichiometric waste, and the requirement for extra purification stages to remove protecting group byproducts. Furthermore, the deprotection steps often require harsh acidic or basic conditions which can compromise the integrity of the sensitive tetrazole ring or lead to the formation of difficult-to-remove impurities. From a supply chain perspective, these extended synthetic sequences result in longer cycle times and increased exposure to potential yield losses at each discrete stage, ultimately inflating the cost of goods sold for the final active pharmaceutical ingredient.
This reliance on protection chemistry introduces significant inefficiencies, including the consumption of additional reagents, the generation of stoichiometric waste, and the requirement for extra purification stages to remove protecting group byproducts. Furthermore, the deprotection steps often require harsh acidic or basic conditions which can compromise the integrity of the sensitive tetrazole ring or lead to the formation of difficult-to-remove impurities. From a supply chain perspective, these extended synthetic sequences result in longer cycle times and increased exposure to potential yield losses at each discrete stage, ultimately inflating the cost of goods sold for the final active pharmaceutical ingredient.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN1915990B achieves a remarkable simplification by completely bypassing the need for hydroxyl protection during the critical coupling and cyclization phases. The inventors have demonstrated that the hydroxymethyl group can remain free throughout the alkylation with 2'-cyano-4-bromomethyl biphenyl and the subsequent conversion to the tetrazole ring without detrimental side reactions. 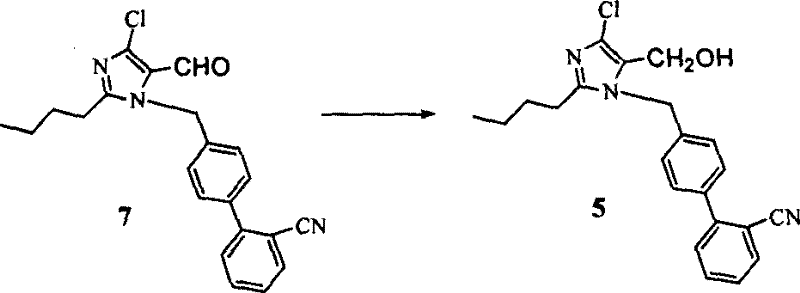 This strategic omission collapses what was previously a three-step sequence (protect-couple-deprotect) into a single, direct alkylation step, drastically reducing the number of unit operations required. The process allows for the direct reaction of 2-butyl-4-chloro-5-hydroxymethylimidazole with the biphenyl bromide under mild basic conditions, or alternatively, the coupling of the aldehyde followed by a highly efficient reduction. This flexibility empowers manufacturers to select the most cost-effective entry point based on raw material availability, while the elimination of protection chemistry translates directly into reduced solvent consumption and simplified downstream processing.
This strategic omission collapses what was previously a three-step sequence (protect-couple-deprotect) into a single, direct alkylation step, drastically reducing the number of unit operations required. The process allows for the direct reaction of 2-butyl-4-chloro-5-hydroxymethylimidazole with the biphenyl bromide under mild basic conditions, or alternatively, the coupling of the aldehyde followed by a highly efficient reduction. This flexibility empowers manufacturers to select the most cost-effective entry point based on raw material availability, while the elimination of protection chemistry translates directly into reduced solvent consumption and simplified downstream processing.
Mechanistic Insights into Catalytic Tetrazole Formation
The cornerstone of this improved synthesis is the optimized conversion of the nitrile intermediate into the pharmacologically active tetrazole ring, a transformation that traditionally requires hazardous conditions and excess azide reagents. The patent details a catalytic system utilizing Lewis acids such as zinc chloride or strong weak base salts like triethylamine hydrochloride to facilitate the [2+3] cycloaddition between the nitrile and sodium azide. 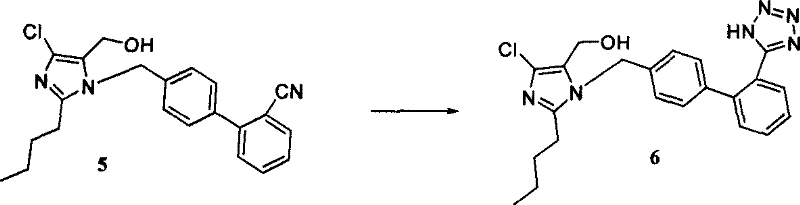 Mechanistically, the catalyst activates the nitrile group towards nucleophilic attack by the azide ion, lowering the activation energy required for the cyclization and allowing the reaction to proceed at moderate temperatures ranging from 110°C to 140°C. This catalytic enhancement is crucial because it permits the use of a lower molar excess of sodium azide compared to uncatalyzed thermal methods, thereby mitigating the safety risks associated with handling large quantities of explosive inorganic azides. For the R&D team, understanding this mechanism is vital for controlling the formation of potential byproducts, ensuring that the final API meets stringent purity specifications without the need for extensive recrystallization.
Mechanistically, the catalyst activates the nitrile group towards nucleophilic attack by the azide ion, lowering the activation energy required for the cyclization and allowing the reaction to proceed at moderate temperatures ranging from 110°C to 140°C. This catalytic enhancement is crucial because it permits the use of a lower molar excess of sodium azide compared to uncatalyzed thermal methods, thereby mitigating the safety risks associated with handling large quantities of explosive inorganic azides. For the R&D team, understanding this mechanism is vital for controlling the formation of potential byproducts, ensuring that the final API meets stringent purity specifications without the need for extensive recrystallization.
Furthermore, the robustness of this catalytic system extends to the choice of solvent, with the patent validating the use of polar aprotic solvents like DMF and N-methylpyrrolidone (NMP), as well as mixed solvent systems involving toluene. The ability to operate effectively in these diverse media provides process chemists with the leverage to optimize solubility parameters and heat transfer characteristics during scale-up. The reaction kinetics are sufficiently fast to achieve high conversions within 48 to 72 hours, yet controlled enough to prevent the degradation of the sensitive imidazole core. By fine-tuning the catalyst loading and temperature, manufacturers can achieve yields exceeding 90% in the final cyclization step, a metric that significantly outperforms many non-catalytic literature precedents. This high level of control over the reaction environment ensures a consistent impurity profile, which is a critical quality attribute for regulatory approval and commercial viability.
How to Synthesize Losartan Intermediate Efficiently
The execution of this synthesis begins with the preparation of the key hydroxymethyl imidazole building block, which can be achieved via the reduction of the corresponding aldehyde using mild hydride donors like potassium borohydride. Following this, the alkylation with the biphenyl bromide is conducted in the presence of a base such as potassium carbonate, a step that proceeds smoothly at room temperature to afford the nitrile intermediate in high yield. The final and most critical transformation involves the cyclization of this nitrile with sodium azide in the presence of the aforementioned catalysts to close the tetrazole ring. For a detailed breakdown of the specific molar ratios, temperature profiles, and workup procedures required to replicate these results in a pilot plant setting, please refer to the standardized synthesis guide below.
- Reduce 2-butyl-4-chloro-5-aldehyde imidazole using KBH4 or NaBH4 to form the hydroxymethyl intermediate, or alternatively alkylate first then reduce.
- React the hydroxymethyl imidazole with 2'-cyano-4-bromomethyl biphenyl under basic conditions (K2CO3) to form the key nitrile intermediate.
- Convert the nitrile group to a tetrazole ring using sodium azide (NaN3) with a Lewis acid or amine salt catalyst in DMF or NMP at elevated temperatures.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers compelling economic and logistical benefits that extend far beyond simple yield improvements. The most significant advantage lies in the drastic simplification of the manufacturing process, which directly correlates to a reduction in capital expenditure and operating costs. By removing the protection and deprotection steps, the facility can reduce the total number of reactor batches required per kilogram of finished product, effectively increasing the throughput of existing infrastructure without the need for new equipment investment. This intensification of the process flow allows for a more agile response to market demand fluctuations, ensuring that supply continuity is maintained even during periods of high global demand for antihypertensive medications.
- Cost Reduction in Manufacturing: The elimination of protecting group reagents, such as trityl chloride, and the associated bases and acids for deprotection, results in a substantial decrease in raw material costs. Additionally, the reduction in the number of isolation and purification steps leads to significant savings in solvent procurement and waste disposal fees, which are often hidden cost drivers in API manufacturing. The catalytic nature of the tetrazole formation further contributes to cost efficiency by minimizing the consumption of sodium azide, a reagent that requires specialized handling and disposal protocols due to its toxicity and explosivity. These cumulative efficiencies create a leaner cost structure that enhances competitiveness in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: The reliance on widely available and commodity-grade starting materials, such as 2-butyl-4-chloro-5-aldehyde imidazole and simple inorganic bases, mitigates the risk of supply disruptions caused by the scarcity of exotic reagents. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent qualities, ensures that production can continue reliably even if there are minor variations in utility supplies or raw material grades. This resilience is critical for maintaining long-term supply contracts with major pharmaceutical partners who prioritize vendor reliability and consistency above all else.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, the process offers a greener alternative to traditional methods by reducing the overall mass intensity and the generation of hazardous waste streams. The ability to run the final cyclization with lower azide loads reduces the potential for energetic incidents, simplifying the safety case for large-scale production. Furthermore, the simplified workup procedures reduce the volume of aqueous and organic waste requiring treatment, aligning the manufacturing process with increasingly stringent global environmental regulations and sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Losartan synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on safety, scalability, and quality control. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for procurement specialists assessing the long-term viability of this supply source.
Q: How does this novel method improve safety regarding sodium azide usage?
A: The patent utilizes specific catalysts like zinc chloride or triethylamine hydrochloride which significantly reduce the required molar ratio of explosive sodium azide, thereby enhancing operational safety and reducing hazardous waste.
Q: What are the primary cost advantages of avoiding hydroxyl protection?
A: By eliminating the protection and deprotection steps required in conventional methods (such as trityl protection), the process reduces raw material consumption, shortens reaction time, and minimizes solvent usage, leading to substantial manufacturing cost reductions.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the method employs readily available raw materials and robust reaction conditions (e.g., room temperature alkylation, standard reflux for cyclization) that are easily scalable from kilogram to multi-ton production without requiring exotic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Losartan Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthetic routes in the modern pharmaceutical landscape. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative strategies outlined in CN1915990B can be seamlessly translated into industrial reality. We are committed to delivering high-purity Losartan intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our facility is equipped to handle the specific safety requirements of azide chemistry, providing a secure and compliant environment for the manufacture of this vital cardiovascular medication.
We invite global partners to collaborate with us to leverage these process advancements for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your next project benefits from the highest standards of chemical manufacturing and supply chain reliability.
