Advanced Rhodium-Catalyzed C-H Activation for Scalable Indanone Derivative Manufacturing
Advanced Rhodium-Catalyzed C-H Activation for Scalable Indanone Derivative Manufacturing
The landscape of organic synthesis for complex heterocyclic scaffolds is constantly evolving, driven by the need for more efficient and environmentally benign processes. A significant breakthrough in this domain is detailed in patent CN112939780A, which discloses a novel synthetic method for indanone derivatives. This technology leverages transition metal-catalyzed C-H bond activation to construct the indanone core directly from readily available benzoic acid compounds and acrylate derivatives. For R&D directors and process chemists seeking reliable pathways to access these privileged structures, this one-step synthesis represents a paradigm shift away from multi-step, waste-generating traditional protocols. The method utilizes a rhodium catalyst system that not only simplifies the operational procedure but also delivers high reaction yields and exceptional functional group tolerance, making it an attractive candidate for the commercial scale-up of complex pharmaceutical intermediates.
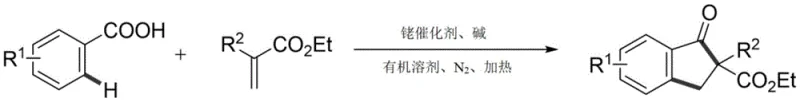
Furthermore, the versatility of this approach extends beyond simple substitution patterns, accommodating a wide range of electronic environments on the aromatic ring. The ability to use the carboxylic acid moiety as a native directing group eliminates the need for installing and removing auxiliary directing groups, thereby enhancing the overall step economy. This innovation addresses critical pain points in modern medicinal chemistry, where speed and efficiency in accessing diverse chemical space are paramount. By enabling the direct coupling of benzoic acids with acrylates under relatively mild thermal conditions, this patent provides a robust foundation for developing cost-effective manufacturing routes for high-purity OLED materials and agrochemical intermediates alike.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the indanone skeleton has relied heavily on classical reactions such as Friedel-Crafts acylation and Nazarov cyclization. While these methods are well-established in academic literature, they suffer from significant drawbacks when applied to industrial-scale manufacturing. Friedel-Crafts reactions typically require stoichiometric amounts of strong Lewis acids, such as aluminum chloride, which generate substantial quantities of corrosive waste and necessitate complex quenching and disposal procedures. Moreover, these reactions often demand harsh conditions that can be incompatible with sensitive functional groups, limiting the scope of substrates that can be utilized. Additionally, traditional approaches frequently require pre-functionalization of the starting materials, adding extra synthetic steps that reduce overall yield and increase production costs. The lack of regioselectivity in some electrophilic aromatic substitutions can also lead to difficult-to-separate isomeric mixtures, complicating downstream purification and impacting the purity profile of the final active pharmaceutical ingredient.
The Novel Approach
In stark contrast, the methodology described in CN112939780A utilizes a rhodium-catalyzed C-H activation strategy that circumvents many of these historical limitations. By employing the carboxylic acid group inherent in benzoic acid derivatives as a directing group, the reaction achieves high ortho-selectivity without the need for external directing auxiliaries. This catalytic system operates under nitrogen atmosphere at elevated temperatures, typically around 140 °C, using 1,2-Dichloroethane (DCE) as the solvent and sodium acetate as a mild base. The use of a catalytic amount of pentamethylcyclopentadienylrhodium dichloride ensures that the transformation is atom-economical and generates significantly less waste compared to stoichiometric Lewis acid promotions. This novel approach not only streamlines the synthetic sequence into a single pot but also expands the chemical space accessible to process chemists, allowing for the incorporation of diverse substituents that would otherwise be unstable under traditional acidic conditions.
Mechanistic Insights into Rhodium-Catalyzed C-H Activation and Cyclization
The core of this transformative synthesis lies in the mechanism of rhodium-catalyzed C-H bond activation. The reaction initiates with the coordination of the rhodium(III) catalyst to the oxygen atoms of the carboxylate group, which acts as a weakly coordinating directing group. This coordination facilitates the cleavage of the ortho C-H bond on the aromatic ring through a concerted metalation-deprotonation (CMD) pathway, forming a stable five-membered rhodacycle intermediate. This organometallic species is highly reactive towards the incoming acrylate olefin, undergoing migratory insertion to extend the carbon chain. Subsequent intramolecular nucleophilic attack by the enolate oxygen onto the carbonyl carbon, followed by reductive elimination or protonolysis, closes the five-membered ring to form the indanone scaffold. The regeneration of the active Rh(III) species completes the catalytic cycle, allowing the turnover to continue efficiently. Understanding this mechanistic nuance is crucial for R&D teams optimizing reaction parameters, as the choice of base and solvent plays a pivotal role in stabilizing the key intermediates and driving the equilibrium towards the desired product.
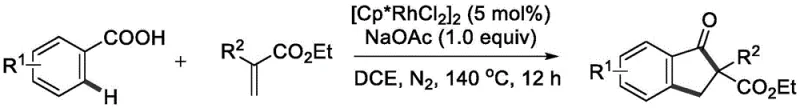
Regarding impurity control, the high selectivity of the C-H activation step inherently minimizes the formation of regioisomers, which are common contaminants in electrophilic aromatic substitutions. The mild basic conditions provided by sodium acetate prevent acid-catalyzed side reactions such as polymerization of the acrylate or decomposition of sensitive functional groups like halogens. Furthermore, the reaction demonstrates remarkable compatibility with electron-withdrawing groups such as fluoro, chloro, and bromo, as well as electron-donating methyl groups, indicating that the electronic nature of the substrate has a manageable impact on the reaction kinetics. This robustness ensures a cleaner crude reaction profile, simplifying the purification process and reducing the burden on quality control laboratories to identify and quantify trace impurities. The ability to tolerate nitrile and ester functionalities simultaneously opens up avenues for further derivatization, making these indanone intermediates highly valuable building blocks for complex molecule synthesis.
How to Synthesize 2-Substituted Indanone Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the specific reaction parameters outlined in the patent data to ensure reproducibility and safety. The process involves charging a reaction vessel with the benzoic acid substrate, the acrylate coupling partner, the rhodium catalyst precursor, and the acetate base in an organic solvent. The mixture is then heated under an inert atmosphere to drive the cyclization to completion. Detailed standard operating procedures regarding stoichiometry, temperature ramping, and workup protocols are essential for achieving the reported yields of up to 78%. For a comprehensive guide on executing this transformation with precision, please refer to the standardized synthesis steps provided below.
- Combine benzoic acid compound, acrylate compound, pentamethylcyclopentadienylrhodium dichloride catalyst, and sodium acetate base in 1,2-Dichloroethane (DCE) solvent.
- Heat the reaction mixture under a nitrogen atmosphere at 140 °C for approximately 12 hours to facilitate the cyclization.
- Upon completion, cool the mixture, perform suction filtration, mix with silica gel, and purify via column chromatography to isolate the 2-substituted indanone product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this rhodium-catalyzed technology offers compelling strategic advantages over legacy manufacturing routes. The simplification of the synthetic sequence from multiple steps to a single direct coupling significantly reduces the consumption of raw materials, solvents, and energy, leading to substantial cost savings in pharmaceutical intermediate manufacturing. By eliminating the need for hazardous Lewis acids and the associated waste treatment infrastructure, facilities can lower their environmental compliance costs and improve their sustainability metrics. The high functional group tolerance means that a wider variety of commercially available starting materials can be utilized without extensive protection-deprotection strategies, enhancing supply chain reliability and reducing lead time for high-purity intermediates. Furthermore, the scalability of transition metal-catalyzed reactions is well-documented, suggesting that this process can be readily transferred from gram-scale discovery to multi-ton commercial production with minimal re-engineering.
- Cost Reduction in Manufacturing: The elimination of stoichiometric Lewis acids and the reduction in synthetic steps directly translate to lower operational expenditures. By avoiding the purchase and disposal of large quantities of corrosive reagents, manufacturers can achieve significant cost optimization. Additionally, the high atom economy of the C-H activation process ensures that a greater proportion of the starting material mass ends up in the final product, minimizing waste generation and maximizing resource efficiency. This lean manufacturing approach is critical for maintaining competitiveness in the global market for fine chemicals.
- Enhanced Supply Chain Reliability: The use of readily available benzoic acids and acrylates as starting materials mitigates the risk of supply disruptions associated with exotic or custom-synthesized precursors. The robustness of the reaction conditions allows for flexibility in sourcing raw materials, as the process can tolerate variations in substrate quality better than sensitive traditional methods. This resilience ensures a consistent supply of critical intermediates, safeguarding production schedules against upstream volatility. Moreover, the simplified workflow reduces the dependency on specialized equipment for handling hazardous reagents, further stabilizing the supply chain.
- Scalability and Environmental Compliance: Transitioning to a catalytic process aligns with green chemistry principles, reducing the environmental footprint of chemical manufacturing. The absence of heavy metal waste streams typical of stoichiometric reactions simplifies effluent treatment and lowers the regulatory burden. The process is amenable to continuous flow chemistry technologies, which can further enhance safety and throughput for large-scale production. This forward-looking approach not only meets current environmental standards but also future-proofs the manufacturing asset against increasingly stringent global regulations on chemical emissions and waste.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthetic methodology. These insights are derived directly from the experimental data and claims within the patent documentation to provide clarity for technical stakeholders. Understanding these nuances is vital for assessing the feasibility of integrating this technology into existing production pipelines.
Q: What are the primary advantages of this Rhodium-catalyzed method over traditional Friedel-Crafts acylation?
A: Unlike traditional methods requiring harsh Lewis acids and pre-functionalized substrates, this Rhodium-catalyzed C-H activation utilizes simple benzoic acids as directing groups, offering milder conditions, better atom economy, and superior functional group compatibility without generating excessive acidic waste.
Q: Can this synthetic route accommodate diverse functional groups on the aromatic ring?
A: Yes, the protocol demonstrates excellent substrate universality, successfully tolerating electron-donating groups like methyl and electron-withdrawing groups such as fluoro, chloro, and bromo at various positions on the benzene ring while maintaining high reaction yields.
Q: Is it possible to convert the diester products into unsubstituted indanones?
A: Absolutely. The patent describes a subsequent decarboxylation step where the 2-substituted indanone diesters are heated in Hexafluoroisopropanol (HFIP) with an inorganic base to efficiently yield the corresponding unsubstituted indanone compounds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indanone Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H activation technologies in accelerating drug discovery and development. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN112939780A can be seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of indanone derivatives delivered meets the highest quality standards required by the global pharmaceutical industry. We are committed to leveraging our technical expertise to solve complex synthetic challenges and deliver value to our partners.
We invite you to collaborate with us to explore how this efficient synthetic route can optimize your supply chain and reduce costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can support your long-term strategic goals in the competitive landscape of fine chemical manufacturing.
