Advanced Rhodium-Catalyzed Synthesis of Indanone Derivatives for Scalable Pharmaceutical Manufacturing
Advanced Rhodium-Catalyzed Synthesis of Indanone Derivatives for Scalable Pharmaceutical Manufacturing
The landscape of organic synthesis for pharmaceutical intermediates is constantly evolving, driven by the need for more sustainable, efficient, and cost-effective methodologies. A significant breakthrough in this domain is documented in Chinese Patent CN112939780B, which outlines a novel synthetic method for indanone derivatives. This technology leverages transition metal-catalyzed C-H bond activation to construct the indanone core directly from readily available benzoic acid compounds and acrylate esters. For R&D directors and process chemists, this represents a paradigm shift away from classical, waste-intensive routes towards a more atom-economical approach. The method utilizes a rhodium catalyst system, specifically pentamethylcyclopentadienylrhodium dichloride, to facilitate ortho-C-H activation followed by intramolecular cyclization. This innovation not only simplifies the synthetic sequence but also enhances the overall yield and purity profile of the resulting 2-substituted indanone compounds, making it highly attractive for the production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the indanone scaffold has relied heavily on classical reactions such as Friedel-Crafts acylation and Nazarov cyclization. While these methods are well-established in academic literature, they present substantial challenges when translated to industrial scale manufacturing. Friedel-Crafts reactions typically require stoichiometric or excess amounts of strong Lewis acids like aluminum chloride, which generate massive quantities of acidic waste and necessitate complex quenching and disposal procedures. Furthermore, these traditional pathways often demand pre-functionalized substrates, adding extra synthetic steps that reduce overall atom economy and increase raw material costs. The harsh acidic conditions can also be incompatible with sensitive functional groups, limiting the scope of substrates that can be utilized without extensive protection-deprotection strategies. Consequently, procurement managers often face higher costs associated with waste treatment and lower throughput due to cumbersome purification requirements inherent to these legacy processes.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN112939780B introduces a streamlined, one-pot synthesis that bypasses the need for pre-functionalization and harsh Lewis acids. By employing a rhodium-catalyzed C-H activation strategy, the process directly couples benzoic acids with acrylates under relatively mild thermal conditions. This approach significantly reduces the number of unit operations required, as the reaction proceeds efficiently in a single vessel with nitrogen protection. The use of sodium acetate as a benign base further mitigates corrosion risks compared to traditional strong bases or acids. This modern catalytic cycle allows for the direct assembly of complex molecular architectures from simple building blocks, thereby drastically simplifying the supply chain for raw materials. For supply chain heads, this translates to a more robust manufacturing process with fewer potential failure points and a reduced dependency on hazardous reagents that are subject to strict regulatory controls.
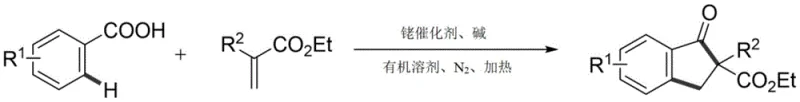
The versatility of this new approach is evident in its broad substrate scope. The reaction tolerates a wide array of substituents on the aromatic ring, including electron-donating groups like methyl and electron-withdrawing groups such as fluoro, chloro, and bromo. This functional group compatibility is crucial for medicinal chemistry applications where diverse analog libraries are needed. Additionally, the system accommodates different acrylate partners, including those with cyano and ethoxycarbonyl groups, allowing for the synthesis of varied 2-substituted indanone derivatives. The ability to introduce these diverse functionalities in a single step without compromising yield is a significant advantage for developing reliable pharmaceutical intermediate supplier capabilities. The reaction conditions are optimized to operate at temperatures around 140 °C in 1,2-Dichloroethane (DCE), ensuring consistent conversion rates across different substrate combinations.
Mechanistic Insights into Rhodium-Catalyzed C-H Activation and Cyclization
The core of this technological advancement lies in the mechanistic efficiency of the rhodium catalyst. The cycle initiates with the coordination of the benzoic acid substrate to the rhodium center, directed by the carboxyl group which acts as a native directing group. This coordination facilitates the selective activation of the ortho-C-H bond, forming a stable rhodacycle intermediate. Subsequently, the acrylate olefin inserts into the rhodium-carbon bond, extending the carbon chain and setting the stage for ring closure. The final step involves an intramolecular nucleophilic attack or reductive elimination that forms the five-membered ring of the indanone skeleton, regenerating the active catalyst species. This mechanism avoids the formation of carbocation intermediates typical of Friedel-Crafts reactions, thereby minimizing side reactions such as polymerization or rearrangement that often plague acid-catalyzed processes. Understanding this mechanism allows process chemists to fine-tune reaction parameters for optimal performance.
Furthermore, the patent details a secondary transformation that adds significant value to the utility of these intermediates. The initial 2-substituted indanone products, which often contain diester or cyano-ester groups at the 2-position, can be subsequently converted into unsubstituted indanones via a decarboxylation protocol. This secondary step utilizes sodium acetate in hexafluoroisopropanol (HFIP) at elevated temperatures (150 °C). The unique solvent properties of HFIP likely stabilize the transition state for decarboxylation, enabling the clean removal of the ester functionality to reveal the parent indanone structure. This two-stage capability provides manufacturers with a flexible platform to produce either functionalized precursors for further derivatization or the core indanone scaffold itself, depending on the specific needs of the downstream API synthesis.

How to Synthesize 2-Substituted Indanone Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and minimize impurities. The standard protocol involves charging a reaction vessel with the benzoic acid derivative, the acrylate coupling partner, the rhodium catalyst, and sodium acetate in DCE solvent. The mixture is then heated under an inert nitrogen atmosphere to ensure oxidative stability of the catalyst and prevent side reactions. Detailed operational guidelines regarding stoichiometry, temperature ramping, and workup procedures are critical for reproducibility. For laboratories aiming to adopt this technology, adhering to the specific molar ratios and purification techniques outlined in the patent is essential to achieve the reported high purity levels. The detailed standardized synthesis steps are provided in the guide below to assist technical teams in rapid method transfer.
- Combine benzoic acid compound, acrylate compound, pentamethylcyclopentadienylrhodium dichloride catalyst, and sodium acetate base in 1,2-Dichloroethane (DCE) solvent.
- Heat the reaction mixture under a nitrogen atmosphere at 140 °C for approximately 12 hours to facilitate C-H activation and cyclization.
- Upon completion, cool the mixture, perform suction filtration, mix with silica gel, and purify via column chromatography to isolate the 2-substituted indanone product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this rhodium-catalyzed methodology offers compelling advantages for cost reduction in pharmaceutical intermediate manufacturing. The elimination of stoichiometric Lewis acids removes a major cost center associated with reagent purchase and, more importantly, hazardous waste disposal. The simplified workup procedure, which often involves basic filtration and standard column chromatography rather than complex aqueous extractions or distillations, reduces solvent consumption and processing time. These factors collectively contribute to a leaner manufacturing process that is both economically and environmentally superior. For procurement managers, this means a more predictable cost structure and reduced exposure to volatile pricing of specialized acidic reagents. The robustness of the reaction also implies fewer batch failures, ensuring a steady supply of critical intermediates.
- Cost Reduction in Manufacturing: The shift from traditional acid-mediated cyclization to catalytic C-H activation fundamentally alters the cost equation by removing the need for expensive and hazardous Lewis acid promoters. Since the catalyst is used in low loading (approximately 5 mol%), the direct material cost is optimized while maintaining high turnover numbers. Additionally, the high atom economy of the reaction ensures that a greater proportion of the starting mass ends up in the final product, reducing the effective cost per kilogram of the active ingredient. This efficiency is further amplified by the ability to use commercially available benzoic acids and acrylates without costly pre-modification steps.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable starting materials like benzoic acids and acrylates enhances supply chain resilience. Unlike sensitive organometallic reagents or unstable acid chlorides required in other methods, these substrates are commodity chemicals with robust global supply networks. The reaction's tolerance to various functional groups also means that supply disruptions for specific substituted precursors can often be mitigated by switching to alternative analogs that still perform well in the catalytic system. This flexibility ensures continuous production schedules and reduces the risk of stockouts for key pharmaceutical building blocks.
- Scalability and Environmental Compliance: Scaling this process is facilitated by the homogeneous nature of the reaction and the absence of exothermic hazards associated with strong acid additions. The use of nitrogen protection and moderate heating is easily manageable in standard stainless steel reactors, allowing for seamless scale-up from pilot to commercial production volumes. Moreover, the reduced generation of acidic wastewater aligns with increasingly stringent environmental regulations, lowering the compliance burden on manufacturing facilities. This green chemistry profile makes the process attractive for companies aiming to improve their sustainability metrics while maintaining high production output.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthetic technology. These insights are derived directly from the experimental data and claims within the patent documentation to provide clarity for technical decision-makers. Understanding these nuances is vital for assessing the feasibility of integrating this route into existing manufacturing pipelines. The answers reflect the specific conditions and outcomes observed during the development of this method.
Q: What are the primary advantages of this Rh-catalyzed method over traditional Friedel-Crafts acylation?
A: Unlike traditional methods requiring harsh Lewis acids and pre-functionalized substrates, this Rh-catalyzed C-H activation utilizes simple benzoic acids and acrylates directly. It offers superior functional group compatibility, milder reaction conditions, and eliminates the need for stoichiometric amounts of corrosive waste-generating reagents.
Q: Can this synthesis protocol tolerate halogen substituents on the aromatic ring?
A: Yes, the protocol demonstrates excellent tolerance for various halogen substituents including fluorine, chlorine, and bromine at the para-position of the benzoic acid substrate. This allows for the direct synthesis of halogenated indanone intermediates which are valuable for further cross-coupling reactions in drug discovery.
Q: Is it possible to convert the diester products into unsubstituted indanones?
A: Absolutely. The patent describes a subsequent decarboxylation step where the 2-substituted indanone diesters are heated with sodium acetate in hexafluoroisopropanol (HFIP) at 150 °C. This efficiently removes the ester groups to yield the corresponding unsubstituted indanone skeleton in high yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indanone Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methods like the one described in CN112939780B for producing high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into reliable industrial processes. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of indanone derivatives meets the exacting standards required for API synthesis. We are committed to leveraging such cutting-edge technologies to deliver superior quality intermediates that accelerate our clients' drug development timelines.
We invite you to collaborate with us to explore how this efficient synthesis route can optimize your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating the economic benefits of switching to this catalytic protocol. Please contact us to request specific COA data for our available indanone inventory or to discuss route feasibility assessments for your custom synthesis projects. Let us be your partner in achieving cost-effective and sustainable chemical manufacturing solutions.
