Scalable Synthesis of Non-Steroidal Glucocorticoid Receptor Modulator Intermediates for Commercial API Production
Introduction to Advanced Glucocorticoid Receptor Modulator Synthesis
The development of non-steroidal glucocorticoid receptor (GR) modulators represents a significant frontier in treating inflammatory and metabolic disorders, addressing conditions ranging from Cushing's syndrome to Addison's disease. Patent CN1356319A discloses a robust and highly efficient preparation method for compounds of Formula I, which serve as potent modulators of the GR pathway. This technology leverages a sophisticated multi-step synthetic route that ensures high stereochemical fidelity and exceptional purity profiles, essential for downstream pharmaceutical applications. The process begins with readily available tetralone derivatives and progresses through a series of catalytic transformations, including palladium-mediated cyanation and stereoselective hydrogenation, to construct the complex tricyclic core.
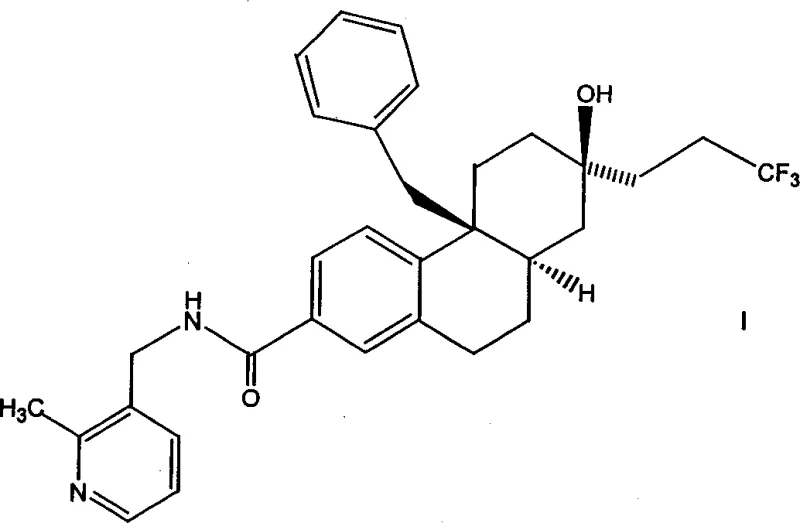
For R&D directors and procurement specialists, understanding the nuances of this synthesis is critical for securing a reliable pharmaceutical intermediate supplier. The disclosed methodology not only optimizes yield but also incorporates strategic purification steps, such as granulation and recrystallization, to eliminate trace impurities and diastereomers. By utilizing established catalytic cycles and avoiding exotic reagents, this process offers a viable pathway for cost reduction in API manufacturing while maintaining the stringent quality standards required for clinical-grade materials. The ability to scale these reactions from gram to kilogram quantities demonstrates the commercial viability of this approach for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing steroidal and non-steroidal GR modulators often suffer from poor stereocontrol, requiring extensive chromatographic purification that drives up costs and reduces overall throughput. Many legacy processes rely on harsh reaction conditions that can degrade sensitive functional groups, leading to complex impurity profiles that are difficult to characterize and remove. Furthermore, conventional methods frequently utilize stoichiometric amounts of chiral reagents or resolving agents, which significantly increases the material cost and waste generation associated with the production of high-purity intermediates. These inefficiencies create bottlenecks in the supply chain, making it challenging to meet the demanding timelines of modern drug development programs.
The Novel Approach
The novel approach detailed in the patent overcomes these hurdles by employing a convergent synthesis strategy that builds complexity early in the sequence while maintaining operational simplicity. Key to this innovation is the use of enamine chemistry to facilitate the introduction of the benzyl group, followed by a Robinson-type annulation that efficiently constructs the tricyclic phenanthrene core with high diastereoselectivity. The integration of palladium-catalyzed cyanation allows for the mild introduction of the nitrile functionality, which serves as a versatile handle for subsequent transformations into the carboxylic acid moiety. This streamlined workflow minimizes the number of isolation steps and maximizes atom economy, providing a distinct advantage for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Palladium-Catalyzed Cyanation and Reduction
A pivotal step in this synthesis is the palladium-catalyzed cyanation of the bromo-intermediate (Compound VII) to form the nitrile (Compound VI). This transformation utilizes tetrakis(triphenylphosphine)palladium(0) and zinc cyanide in a polar aprotic solvent like DMF at elevated temperatures around 80°C. The mechanism involves the oxidative addition of the aryl bromide to the Pd(0) center, followed by transmetallation with the cyanide source and reductive elimination to forge the carbon-nitrogen triple bond. This catalytic cycle is highly efficient, as evidenced by the 96% yield reported in Example 4, and avoids the use of toxic gaseous HCN, enhancing safety profiles for industrial operations.
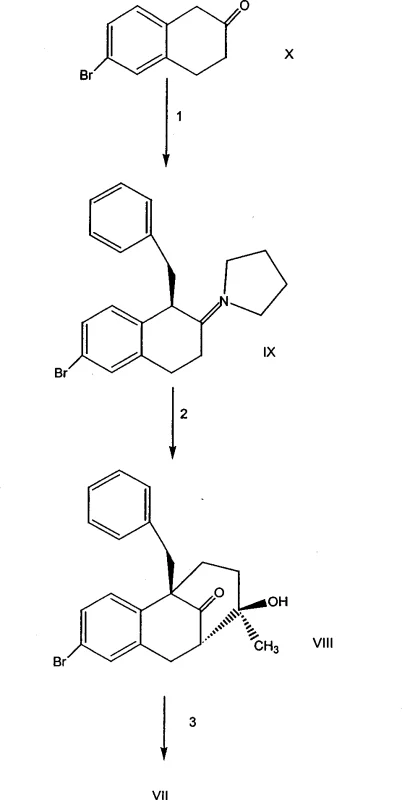
Following the installation of the nitrile group, the process employs stereoselective hydrogenation to reduce the olefinic bonds and introduce the trifluoropropyl side chain. The reduction of the alkyne intermediate (Compound IV) to the saturated propyl chain (Compound III) is carefully controlled using palladium on charcoal under moderate hydrogen pressure (20 psi). This step is crucial for establishing the correct stereochemistry at the C7 position, which is vital for the biological activity of the final modulator. The subsequent hydrolysis of the nitrile to the carboxylic acid is performed under basic conditions, ensuring that the sensitive trifluoromethyl group remains intact while converting the precursor into the active acid form ready for final coupling.
How to Synthesize Formula I Compound Efficiently
The synthesis of the target Formula I compound involves a sequential nine-step process that transforms simple starting materials into a highly functionalized therapeutic candidate. The route begins with the formation of a pyrrolidinium salt from a bromotetralone derivative, followed by chiral annulation to set the core stereochemistry. Subsequent steps include cyanation, hydrogenation, alkyne addition, and final amide coupling. Each stage is optimized for yield and purity, utilizing standard laboratory equipment and commercially available reagents. For detailed operational parameters, temperature controls, and workup procedures, please refer to the standardized synthesis guide below which outlines the critical process parameters derived from the patent examples.
- React bromotetralone derivatives with pyrrolidine and benzyl halide to form the pyrrolidinium intermediate (Compound IX).
- Perform Robinson-type annulation with methyl vinyl ketone and chiral amines to establish the tricyclic core (Compound VIII).
- Execute palladium-catalyzed cyanation followed by stereoselective hydrogenation and alkyne addition to install the trifluoropropyl side chain.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial cost savings by eliminating the need for expensive chiral chromatography columns and reducing the reliance on rare earth catalysts. The use of robust palladium catalysts that can potentially be recovered and recycled further enhances the economic feasibility of the process. Additionally, the starting materials, such as bromotetralone derivatives and pyrrolidine, are commodity chemicals with stable supply chains, mitigating the risk of raw material shortages. This stability ensures enhanced supply chain reliability, allowing manufacturers to plan long-term production schedules without fear of disruption from volatile precursor markets.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing high-yielding catalytic steps that minimize waste and solvent consumption. For instance, the cyanation step proceeds with near-quantitative conversion, reducing the need for extensive recycling of unreacted starting materials. Furthermore, the avoidance of cryogenic conditions in most steps lowers energy consumption, contributing to a lower overall cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: By relying on widely available reagents like zinc cyanide and standard hydrogenation catalysts, the manufacturing process is less susceptible to geopolitical supply constraints. The robustness of the chemistry allows for flexible sourcing of raw materials from multiple vendors, ensuring continuity of supply even if one supplier faces issues. This diversification is critical for maintaining uninterrupted production of high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The synthetic route is designed with scalability in mind, utilizing reaction conditions that are easily transferable from pilot plant to commercial manufacturing scales. The waste streams generated are primarily organic solvents and inorganic salts, which can be managed through standard treatment protocols, ensuring compliance with environmental regulations. The high atom economy of the key coupling reactions further reduces the environmental footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of these glucocorticoid receptor modulator intermediates. The answers are derived directly from the experimental data and process descriptions found in the patent literature, providing accurate insights into the capabilities and limitations of the technology. Understanding these details helps stakeholders make informed decisions about integrating this synthesis route into their existing manufacturing portfolios.
Q: What are the critical purity specifications for the Formula I intermediate?
A: The patent describes achieving high purity through rigorous crystallization steps, such as granulation in ethyl acetate and hexane, resulting in products with >97% purity as confirmed by HPLC analysis.
Q: Is the synthesis route scalable for commercial manufacturing?
A: Yes, the process utilizes standard industrial reagents like zinc cyanide and palladium catalysts, and examples demonstrate reactions on scales up to hundreds of grams, indicating strong potential for metric ton scale-up.
Q: How is stereochemistry controlled during the synthesis?
A: Stereocontrol is achieved through the use of chiral auxiliaries like S-(-)-α-methylbenzylamine during the annulation step and specific hydrogenation conditions that favor the desired diastereomer.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Formula I Compound Supplier
At NINGBO INNO PHARMCHEM, we specialize in translating complex patent methodologies into commercial reality, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at optimizing the palladium-catalyzed steps and stereoselective reductions described in CN1356319A to meet your specific purity requirements. We operate stringent purity specifications and maintain rigorous QC labs equipped with HPLC and NMR capabilities to ensure every batch of Formula I intermediate meets the highest industry standards for potency and impurity profiles.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your project volume. Whether you need specific COA data for regulatory filings or route feasibility assessments for process validation, our experts are ready to provide the support necessary to accelerate your drug development timeline. Partner with us to leverage this advanced synthesis technology and secure a stable, high-quality supply of critical pharmaceutical intermediates for your global operations.
