Advanced Iridium Coordination Compounds for Next-Generation OLED Manufacturing
Advanced Iridium Coordination Compounds for Next-Generation OLED Manufacturing
The rapid evolution of the organic light-emitting diode (OLED) industry demands materials that not only exhibit high quantum efficiency but also possess the thermal stability and processability required for commercial-scale production. Patent CN101759685A introduces a groundbreaking class of organic electroluminescent iridium coordination compounds that address the critical bottlenecks of traditional phosphorescent emitters. By integrating hole-transport functionalities directly into the ligand structure via covalent bonding, this technology effectively creates a host-guest integrated system within a single molecule. This architectural innovation solves the persistent issues of phase separation and concentration quenching found in conventional blended systems. Furthermore, the strategic modification with alkyl chains ensures excellent solubility, paving the way for solution-processable manufacturing methods that are far more cost-effective than vacuum deposition. As a reliable OLED material supplier, understanding these molecular advancements is crucial for developing next-generation display technologies.
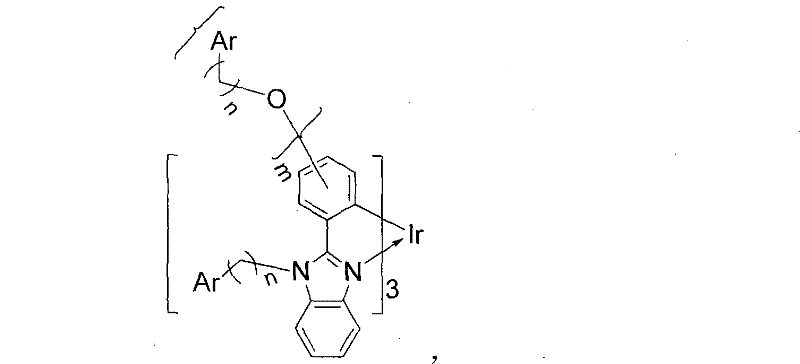
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional phosphorescent OLED devices typically rely on a doping strategy where a small molecule guest emitter is dispersed within a host matrix. While effective in laboratory settings, this approach suffers from significant drawbacks when scaled for industrial application. The physical blending of host and guest molecules often leads to phase separation over time, causing instability in the emissive layer and reducing the operational lifetime of the display. Additionally, small molecule emitters generally exhibit poor solubility in common organic solvents, necessitating the use of vacuum thermal evaporation for film formation. This vacuum-based process is inherently expensive, energy-intensive, and results in low material utilization rates, as a significant portion of the source material is lost during deposition. Moreover, the lack of covalent linkage between the host and guest can lead to inefficient energy transfer and exciton confinement issues, ultimately limiting the external quantum efficiency of the device.
The Novel Approach
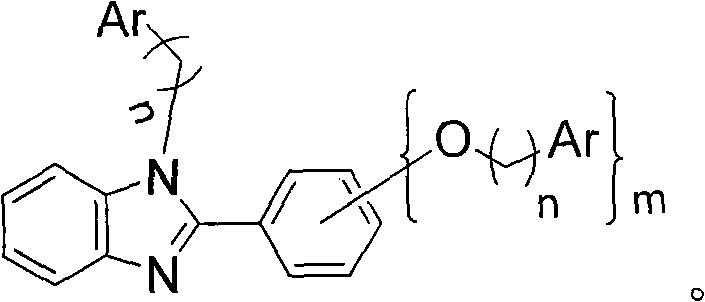
The methodology outlined in the patent data presents a paradigm shift by synthesizing a single-molecule system that embodies both the emissive center and the charge transport capabilities. By covalently attaching hole-transport groups, such as carbazole or triarylamine derivatives, to the phenyl benzoglyoxaline ligand, the material inherently prevents the aggregation of luminescent centers that causes concentration quenching. This "subjective and objective integral structure" ensures uniform dispersion of the emitting species without the need for a separate host matrix. The inclusion of flexible alkyl chains between the functional groups dramatically improves the solubility of the complex, enabling high-quality film formation through solution processing techniques like spin coating and inkjet printing. This transition from vacuum deposition to solution processing represents a massive leap forward in cost reduction in electronic chemical manufacturing, allowing for the fabrication of large-area displays with significantly lower capital expenditure.
Mechanistic Insights into Hole-Transport Modified Iridium Coordination
The core of this technological advancement lies in the precise molecular engineering of the ligand system surrounding the trivalent iridium ion. The phenyl benzoglyoxaline moiety serves as the primary cyclometalating ligand, providing a rigid framework that facilitates strong spin-orbit coupling, which is essential for harvesting triplet excitons and achieving high phosphorescence efficiency. However, the true innovation is the peripheral modification with arylamine-based hole-transport groups. These groups, such as carbazole, possess high triplet energy levels (up to 3.7 eV), which prevents reverse energy transfer from the iridium center to the ligand, thereby confining the excitons effectively on the metal center for radiative decay. The covalent linkage ensures that the hole injection and transport properties are intrinsic to the emitter molecule itself, balancing the charge carrier flux within the emissive layer and reducing the driving voltage required for operation.
Furthermore, the control of impurities and morphological stability is achieved through the steric bulk provided by the peripheral substituents. The alkyl chains, typically ranging from 2 to 6 carbon atoms, act as insulating spacers that physically isolate the bulky iridium cores from one another. This steric hindrance minimizes intermolecular interactions that could lead to non-radiative decay pathways or excimer formation, which are detrimental to color purity and efficiency. The resulting amorphous nature of the material, confirmed by XRD analysis showing broad halos rather than sharp crystalline peaks, is highly desirable for OLED applications as it prevents the formation of grain boundaries that can act as leakage current paths. This structural integrity contributes to the observed thermal decomposition temperatures exceeding 400°C, ensuring that the material can withstand the thermal stresses of device fabrication and operation without degradation.
How to Synthesize Alkyl-Substituted Phenyl Benzoglyoxaline Iridium Complexes Efficiently
The synthesis route described in the patent offers a robust and scalable pathway for producing these high-performance materials. The process begins with the condensation of benzaldehyde derivatives with o-phenylenediamine to form the benzimidazole core, followed by N-alkylation with functionalized alkyl bromides to introduce the hole-transport moieties. The final step involves coordination with iridium precursors, either via a chloro-bridged dimer intermediate or direct reaction with iridium acetylacetonate. This modular approach allows for easy tuning of the emission color and electronic properties by varying the substituents on the benzaldehyde or the nature of the hole-transport group. Detailed standardized synthesis steps see the guide below.
- Condense benzaldehyde derivatives with o-phenylenediamine in DMF with sodium metabisulfite to form the phenyl benzoglyoxaline core.
- Alkylate the ligand precursor with hole-transport group substituted alkyl bromides (e.g., carbazole derivatives) in acetone under basic conditions.
- Coordinate the functionalized ligand with iridium trichloride to form a chloro-bridged dimer, followed by reaction with glycerol and potassium carbonate to yield the final neutral complex.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits regarding cost structure and supply reliability. The starting materials, such as benzaldehyde derivatives, o-phenylenediamine, and carbazole, are commodity chemicals available from multiple global suppliers, reducing the risk of single-source dependency. The reaction conditions are relatively mild, utilizing common solvents like DMF, acetone, and glycerol, which simplifies the infrastructure requirements for production facilities. The elimination of complex vacuum deposition equipment in favor of solution processing downstream significantly lowers the barrier to entry for manufacturing partners. This shift not only reduces the initial capital investment but also decreases the ongoing operational costs associated with maintaining high-vacuum systems.
- Cost Reduction in Manufacturing: The synthetic pathway utilizes readily available raw materials and avoids the use of exotic or prohibitively expensive catalysts in the ligand formation steps. By integrating the host and guest functions into a single molecule, the formulation process for the final OLED ink is simplified, removing the need for precise blending ratios and compatibility testing of separate host and guest materials. This consolidation of components leads to substantial cost savings in the overall bill of materials. Additionally, the high solubility of the final complex allows for higher solid content in inks, improving the throughput of coating processes and reducing solvent consumption and recovery costs.
- Enhanced Supply Chain Reliability: The robustness of the chemical synthesis, characterized by high yields in the intermediate steps and straightforward purification methods such as recrystallization and column chromatography, ensures consistent batch-to-batch quality. The use of stable intermediates like the chloro-bridged iridium dimer allows for inventory buffering, decoupling the final complexation step from upstream supply fluctuations. This flexibility enhances the resilience of the supply chain against market volatility. Furthermore, the thermal stability of the final product reduces the need for specialized cold-chain logistics, simplifying storage and transportation requirements.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction vessels and conditions that are easily translated from laboratory to pilot and commercial scales. The avoidance of harsh reagents and the ability to recover solvents like acetone and dichloromethane align with modern environmental, health, and safety (EHS) standards. The high atom economy of the condensation reactions and the efficient use of iridium, a precious metal, minimize waste generation. This environmentally conscious approach not only mitigates regulatory risks but also appeals to end-users who prioritize sustainable manufacturing practices in their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iridium complex technology. These answers are derived directly from the experimental data and structural analysis provided in the patent documentation, offering clarity on performance metrics and processing capabilities. Understanding these details is essential for evaluating the feasibility of integrating this material into existing production lines.
Q: What is the primary advantage of this iridium complex over traditional small molecule phosphors?
A: The primary advantage is its dual functionality as both emitter and host, facilitated by covalently linked hole-transport groups, which eliminates phase separation issues common in blended systems and enables efficient solution processing.
Q: How does the molecular design improve thermal stability?
A: The incorporation of rigid carbazole or triarylamine units combined with the robust phenyl benzoglyoxaline iridium core results in a thermal decomposition temperature exceeding 400°C, ensuring device longevity.
Q: Is this material suitable for large-area display manufacturing?
A: Yes, the introduction of flexible alkyl chains significantly enhances solubility in common organic solvents, making it compatible with cost-effective solution-based techniques like spin coating and inkjet printing for large-area substrates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Iridium Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chemistry described in CN101759685A for the future of the display industry. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to mass manufacturing is seamless. We are committed to delivering high-purity OLED material that meets stringent purity specifications, utilizing our rigorous QC labs to verify every batch against critical performance parameters. Our capability to handle complex organometallic synthesis under inert atmospheres guarantees the consistency and reliability required by top-tier electronics manufacturers.
We invite you to collaborate with us to optimize your supply chain for next-generation OLED materials. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and processing constraints. Contact us today to request specific COA data and route feasibility assessments for this advanced iridium coordination compound. Let us help you engineer the bottleneck out of your production process and accelerate your time to market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
