Advanced Total Synthesis of Camptothecin Derivatives for Scalable API Production
Introduction to Patent CN103408559A
The pharmaceutical industry continuously seeks robust and scalable methodologies for producing complex alkaloids, particularly those with potent anticancer properties like camptothecin. Patent CN103408559A introduces a groundbreaking rapid synthesis method for camptothecin and its derivatives, addressing critical bottlenecks in traditional manufacturing. This intellectual property outlines a highly convergent strategy that bypasses the limitations of plant extraction and semi-synthesis, offering a pathway to high-purity pharmaceutical intermediates through purely chemical means. The core innovation lies in a sequence involving a one-pot oxidation-oxa-Diels-Alder reaction, a convergent aminolysis between a quinolinodihydropyrrole and a pyranolide intermediate, and a sophisticated one-step cascade reaction to form the essential D and E rings. By leveraging mild reaction conditions and simple operational procedures, this technology represents a significant leap forward for reliable pharmaceutical intermediates suppliers aiming to secure the supply chain for next-generation topoisomerase I inhibitors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
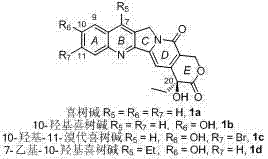
Historically, the production of camptothecin-based drugs has been heavily reliant on the extraction of natural camptothecin from Camptotheca acuminata, a plant resource that is not only geographically limited but also subject to agricultural variability and ecological protection regulations. Since the first racemic synthesis reported by Stork in 1971, numerous synthetic routes have been proposed, yet few have achieved industrial maturity due to prohibitive costs, harsh reaction conditions, and low overall yields. Conventional semi-synthetic approaches often struggle with regioselectivity issues, particularly when attempting to introduce specific substituents at the C11 position without affecting the sensitive C10 hydroxyl group. Furthermore, the poor water solubility and high toxicity of natural camptothecin necessitate complex derivatization, which is difficult to achieve efficiently when starting from a fixed natural scaffold. These factors collectively contribute to extended lead times and supply chain vulnerabilities for cost reduction in API manufacturing.
The Novel Approach
In stark contrast, the methodology described in CN103408559A employs a modular, convergent design that decouples the synthesis of the A/B/C ring system from the D/E ring construction. This approach allows for the independent optimization of each fragment before their final assembly, significantly enhancing process flexibility. The utilization of a one-pot oxidation-oxa-Diels-Alder reaction eliminates the need for isolating unstable intermediates, thereby streamlining the workflow and reducing waste generation. Moreover, the ability to introduce diverse functional groups at the C11 position during the early stages of the quinoline fragment synthesis enables the rapid generation of analog libraries for structure-activity relationship (SAR) studies. This synthetic versatility, combined with the use of mild reagents and ambient temperature protocols, positions this technology as a superior alternative for the commercial scale-up of complex alkaloids, ensuring consistent quality and availability regardless of seasonal plant harvests.
Mechanistic Insights into One-Pot Oxa-Diels-Alder and Cascade Cyclization
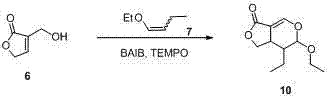
The heart of this synthetic innovation is the efficient construction of the pyran ring system via an intermolecular oxa-Diels-Alder reaction. As illustrated in the reaction scheme, the process begins with the in situ oxidation of a furan alcohol derivative using a BAIB/TEMPO catalytic system. This generates a reactive aldehyde species that immediately undergoes cycloaddition with an electron-rich enol ether. This tandem sequence is remarkable for its atom economy and operational simplicity, occurring effectively at temperatures ranging from -10°C to 30°C. The mechanistic pathway ensures high stereocontrol during the formation of the dihydropyran scaffold, which serves as the precursor for the E-ring lactone. By avoiding the isolation of the transient aldehyde, the process minimizes side reactions such as polymerization or over-oxidation, which are common pitfalls in traditional stepwise syntheses. This level of control is crucial for maintaining the integrity of the sensitive lactone moiety throughout the subsequent transformations.
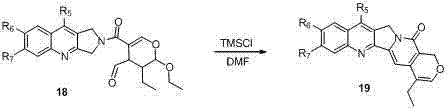
Following the assembly of the pyran fragment, the synthesis proceeds through a convergent coupling with a quinoline-dihydropyrrole amine. The final closure of the pentacyclic skeleton is achieved through a TMSCl-mediated cascade reaction. In this critical step, an aldehyde-functionalized intermediate undergoes intramolecular cyclization facilitated by trimethylchlorosilane in DMF. This reaction simultaneously constructs the D-ring pyridone and closes the E-ring lactone, establishing the rigid planar structure characteristic of camptothecin. The use of TMSCl acts as a Lewis acid promoter, activating the carbonyl groups for nucleophilic attack by the adjacent amide nitrogen and hydroxyl groups. This cascade mechanism is highly efficient, transforming a linear precursor into the complex five-ring core in a single operation. Such mechanistic elegance not only reduces the number of purification steps but also significantly improves the overall yield, making it an ideal candidate for reducing lead time for high-purity pharmaceutical intermediates.
How to Synthesize Camptothecin Derivatives Efficiently
The synthesis protocol detailed in the patent provides a clear roadmap for producing camptothecin analogs with high fidelity. The process is divided into three main phases: the preparation of the pyranolactone fragment, the synthesis of the substituted quinoline amine, and the final convergent coupling and cyclization. Each phase utilizes readily available starting materials and standard laboratory equipment, facilitating easy transfer from pilot scale to full commercial production. The detailed standardized synthesis steps see the guide below for specific molar ratios, solvent systems, and workup procedures that ensure reproducibility.
- Preparation of the pyranolactone intermediate via a one-pot oxidation and intermolecular oxa-Diels-Alder reaction using BAIB and TEMPO.
- Synthesis of the quinoline-dihydropyrrole amine fragment through reduction, mesylation, and ammonolysis of substituted quinoline aldehydes.
- Convergent coupling of the two fragments followed by TMSCl-mediated cascade cyclization to construct the critical D and E rings of the camptothecin skeleton.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers transformative benefits that extend beyond mere technical feasibility. By shifting production from a agriculture-dependent model to a fully synthetic chemical process, organizations can mitigate the risks associated with crop failures, geopolitical instability in sourcing regions, and fluctuating raw material prices. The streamlined nature of the reaction sequence, characterized by fewer unit operations and milder conditions, translates directly into lower energy consumption and reduced solvent usage. This efficiency drives substantial cost savings in manufacturing overheads while simultaneously aligning with increasingly stringent environmental regulations regarding waste disposal and carbon footprint.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts in favor of organocatalytic systems like TEMPO/BAIB significantly lowers the cost of goods sold (COGS). Furthermore, the high convergence of the route means that expensive chiral building blocks are introduced at later stages, minimizing the financial impact of any potential yield losses in early steps. The ability to perform key transformations in one pot reduces the need for intermediate isolation and purification, which are typically the most labor-intensive and costly parts of API production. This logical optimization of the synthetic tree ensures that the final product can be offered at a highly competitive price point without compromising on quality standards.
- Enhanced Supply Chain Reliability: Dependence on natural plant extracts creates inherent supply chain fragility; this synthetic alternative provides a robust, year-round production capability that is immune to seasonal variations. The starting materials, such as furfural derivatives and substituted quinolines, are commodity chemicals available from multiple global suppliers, ensuring redundancy and security of supply. By establishing a fully synthetic route, manufacturers can guarantee consistent batch-to-batch quality and impurity profiles, which is critical for regulatory compliance in the pharmaceutical sector. This reliability allows downstream partners to plan their inventory and production schedules with greater confidence, reducing the need for safety stock and freeing up working capital.
- Scalability and Environmental Compliance: The reaction conditions described, predominantly operating at or near room temperature, are inherently safer and easier to scale than processes requiring cryogenic temperatures or high-pressure reactors. The use of common organic solvents like DCM, THF, and toluene simplifies solvent recovery and recycling infrastructure. Additionally, the high atom economy of the cascade cyclization step minimizes the generation of hazardous byproducts, easing the burden on wastewater treatment facilities. This alignment with green chemistry principles not only reduces environmental liability but also enhances the corporate sustainability profile, a key metric for modern procurement decisions in the life sciences industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical aspects of adopting this route for large-scale manufacturing.
Q: Why is C11 substitution significant in camptothecin derivatives?
A: According to the patent background, modifying the C11 position while retaining the 10-hydroxyl group can significantly influence anticancer activity and potentially increase the stability of the E-ring lactone in plasma, thereby extending the drug's half-life.
Q: What are the primary advantages of this synthetic route over traditional methods?
A: The disclosed method offers a convergent and concise route that operates under mild conditions, often at room temperature or 0°C. Unlike semi-synthesis which relies on scarce plant resources, this total synthesis allows for flexible structural diversification and scalable production independent of agricultural variables.
Q: How is stereochemical control achieved in the final steps?
A: The process utilizes established asymmetric synthesis techniques, specifically employing Sharpless asymmetric dihydroxylation with chiral ligands like (DHQD)2-PYR, followed by iodine-mediated oxidation to secure the correct stereochemistry at the C20 position.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Camptothecin Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of securing a stable supply of critical oncology intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative chemistry described in CN103408559A can be seamlessly translated into industrial reality. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and potency of every batch. Our commitment to quality assurance means that we can deliver camptothecin derivatives that meet the exacting standards required for clinical and commercial API applications.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this synthetic method. We are prepared to provide specific COA data and route feasibility assessments tailored to your project requirements, ensuring a partnership built on transparency, technical excellence, and mutual growth.
