Advanced Camptothecin Synthesis: Scalable Technology for Global Pharmaceutical Intermediates
Advanced Camptothecin Synthesis: Scalable Technology for Global Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust manufacturing routes for critical antineoplastic agents, and the production of camptothecin (CPT) remains a pivotal challenge due to its scarcity in nature. Patent CN1228341C introduces a transformative methodology that economically produces large quantities of camptothecin, serving as the essential starting compound for irinotecan hydrochloride and various derivatives. This innovation addresses the critical bottleneck of relying on limited natural extraction from Camptotheca acuminata, which often yields insufficient quantities for global demand. By leveraging 9-methoxycamptothecin (9-MC), a compound often discarded as a by-product in natural material processing, this technology unlocks a stable and abundant supply chain for high-purity API intermediates. The process is characterized by its operational simplicity and the use of readily available reagents, positioning it as a superior alternative for a reliable API intermediate supplier seeking to optimize their portfolio.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the acquisition of camptothecin has been heavily dependent on the direct extraction from natural plants, a method plagued by severe limitations in scalability and consistency. The content of CPT in natural materials is extremely low, making the isolation process labor-intensive and economically inefficient for meeting the high demand of derivatives like CPT-11. Furthermore, total synthesis approaches have historically struggled with practical application due to complex equipment requirements, low overall yields, and prohibitive costs associated with multi-step organic synthesis. These conventional pathways often involve harsh conditions or expensive catalysts that complicate the purification process, leading to higher impurity profiles that are unacceptable for pharmaceutical grade materials. Consequently, the industry has faced persistent challenges in cost reduction in pharmaceutical intermediates manufacturing, as the supply cannot reliably match the escalating clinical needs for topoisomerase I inhibitors.
The Novel Approach
The novel approach detailed in the patent circumvents these historical constraints by utilizing 9-methoxycamptothecin as a strategic starting material, effectively turning a waste product into a high-value precursor. This method involves a streamlined sequence where 9-MC is hydrolyzed to 9-hydroxycamptothecin (9-HC), followed by derivatization and hydrogenolysis to yield the target camptothecin. By focusing on the chemical conversion of abundant analogues rather than scarce natural CPT, the process ensures a continuous and scalable feedstock supply. The reaction conditions are optimized for efficiency, utilizing common solvents and avoiding the need for exotic reagents that drive up production expenses. This strategic shift not only stabilizes the supply chain but also significantly simplifies the purification workflow, allowing manufacturers to achieve commercial scale-up of complex alkaloids with greater confidence and reduced operational risk.
Mechanistic Insights into 9-MC to Camptothecin Conversion
The core of this technology lies in a precise three-step chemical transformation that maximizes yield while minimizing side reactions. The initial step involves the hydrolysis of the methoxy group in 9-MC using 47% hydrobromic acid at elevated temperatures ranging from 160-180°C, which efficiently cleaves the ether bond to form 9-HC. Subsequently, the 9-hydroxyl group is activated through triflation, typically using trifluoromethanesulfonyl chloride or anhydrous trifluoromethanesulfonic acid in the presence of a base like triethylamine. This creates a highly reactive 9-O-trifluoromethanesulfonyl derivative (9-OTfC), which serves as an excellent substrate for the final reduction step. The mechanistic elegance of this route ensures that the stereochemistry at the C-20 position is preserved, yielding the biologically active 20(S)-camptothecin isomer essential for therapeutic efficacy.
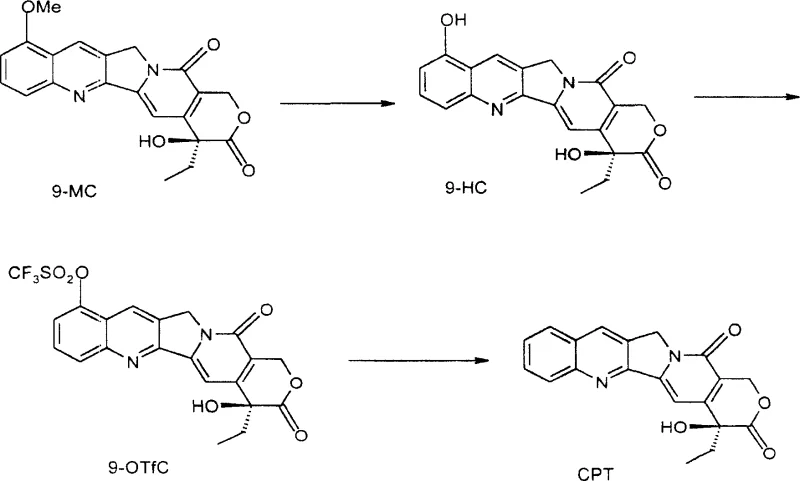
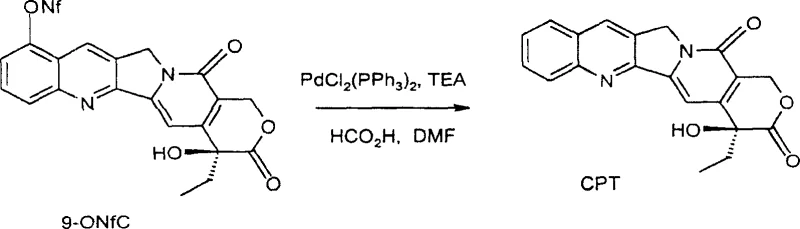
The final hydrogenolysis step is particularly noteworthy for its safety and efficiency, utilizing formic acid as a hydrogen source in the presence of a palladium catalyst rather than high-pressure hydrogen gas. This transfer hydrogenation mechanism proceeds smoothly at moderate temperatures around 60°C, facilitating the removal of the triflate group to restore the aromatic system of the camptothecin core. The use of formic acid eliminates the need for specialized high-pressure reactors, thereby reducing capital expenditure and enhancing operational safety within the manufacturing facility. Furthermore, the reaction conditions are mild enough to prevent the degradation of the sensitive lactone ring, ensuring high purity and minimizing the formation of difficult-to-remove impurities. This meticulous control over reaction parameters underscores the process's suitability for producing high-purity camptothecin required for downstream drug synthesis.
How to Synthesize Camptothecin Efficiently
Implementing this synthesis route requires careful attention to reaction conditions and reagent stoichiometry to ensure optimal conversion rates and product quality. The process begins with the suspension of 9-MC in hydrobromic acid, followed by rigorous degassing and heating under an inert atmosphere to prevent oxidation. Detailed standard operating procedures regarding temperature control, reaction times, and work-up protocols are critical for reproducibility on a large scale. For R&D teams looking to adopt this technology, understanding the nuances of the triflation step and the subsequent catalytic reduction is key to maximizing overall yield. The detailed standardized synthesis steps see the guide below for specific operational parameters and safety considerations.
- Hydrolyze 9-methoxycamptothecin using 47% hydrobromic acid at 160-180°C to obtain 9-hydroxycamptothecin.
- Convert 9-hydroxycamptothecin to 9-trifluoromethanesulfonyloxycamptothecin using triflating agents in DMF.
- Perform hydrogenolysis of the triflate derivative using a palladium catalyst and formic acid to yield camptothecin.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers substantial benefits that directly address the pain points of procurement managers and supply chain directors in the fine chemical sector. By shifting the raw material base from scarce natural CPT to abundant 9-MC, manufacturers can achieve significant cost savings in raw material acquisition and processing. The elimination of complex total synthesis steps and the use of inexpensive reagents like formic acid further drive down the cost of goods sold, making the final API intermediate more competitive in the global market. Additionally, the simplified equipment requirements mean that production can be scaled up rapidly without massive capital investment in specialized high-pressure infrastructure, enhancing supply chain reliability and responsiveness to market fluctuations.
- Cost Reduction in Manufacturing: The utilization of 9-methoxycamptothecin, often available as a by-product from natural extraction processes, drastically reduces the cost of starting materials compared to purchasing pure camptothecin. Furthermore, the replacement of high-pressure hydrogen gas with formic acid for the hydrogenolysis step eliminates the need for expensive safety measures and specialized equipment, leading to lower operational expenditures. The overall reduction in step count and the use of common solvents contribute to a leaner manufacturing process that minimizes waste disposal costs and energy consumption. These factors combined result in a more economically viable production model that allows for competitive pricing without compromising on quality standards.
- Enhanced Supply Chain Reliability: Relying on natural extraction for camptothecin creates a fragile supply chain vulnerable to agricultural variances and seasonal limitations, whereas this chemical synthesis route offers a consistent and predictable production schedule. The availability of 9-MC from various natural sources or synthetic pathways ensures that raw material shortages are unlikely to disrupt manufacturing operations. This stability is crucial for long-term supply agreements with pharmaceutical companies that require guaranteed delivery timelines for their drug development pipelines. By diversifying the source of the key intermediate, manufacturers can mitigate risks associated with single-source dependency and ensure continuous availability of critical oncology ingredients.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from laboratory scale to multi-ton commercial production without significant re-optimization. The use of formic acid as a hydrogen source generates carbon dioxide as a by-product, which is easier to manage and vent compared to the risks associated with handling large volumes of hydrogen gas. Moreover, the streamlined workflow reduces the volume of chemical waste generated per kilogram of product, aligning with increasingly stringent environmental regulations and sustainability goals. This eco-friendly profile enhances the marketability of the product to green-conscious pharmaceutical partners and facilitates smoother regulatory approvals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this camptothecin production technology. These answers are derived directly from the patent specifications and practical considerations for industrial application, providing clarity for stakeholders evaluating this route. Understanding these aspects is essential for making informed decisions about process adoption and supply chain integration. The responses highlight the balance between technical feasibility and commercial viability that defines this innovative approach.
Q: What is the primary advantage of using 9-methoxycamptothecin as a starting material?
A: Utilizing 9-methoxycamptothecin, often found as a by-product or in discarded natural materials, significantly reduces raw material costs compared to extracting camptothecin directly from limited natural sources.
Q: How does the hydrogenolysis step ensure safety and efficiency?
A: The process uses formic acid as a hydrogen source instead of high-pressure hydrogen gas, which simplifies equipment requirements and enhances operational safety while maintaining high conversion rates.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the method employs common reaction apparatus and inexpensive reagents, making it highly adaptable for commercial scale-up of complex alkaloids without requiring specialized high-pressure infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Camptothecin Supplier
The technical potential of this synthesis route is immense, offering a pathway to secure, high-quality camptothecin for the global oncology market. NINGBO INNO PHARMCHEM, as a seasoned CDMO expert, possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this technology to fruition. Our facility is equipped with stringent purity specifications and rigorous QC labs to ensure that every batch meets the exacting standards required for pharmaceutical intermediates. We understand the critical nature of supply continuity in the drug development lifecycle and are committed to delivering materials that support your clinical and commercial goals without compromise.
We invite you to discuss how this optimized process can benefit your specific supply chain requirements and cost structures. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs and quality expectations. Please contact us to request specific COA data and route feasibility assessments that demonstrate our capability to be your long-term partner in chemical manufacturing. Let us help you secure a stable supply of high-purity camptothecin for your next generation of therapeutic agents.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
