Advanced Catalytic Route for High-Purity Thiophene Derivatives and Commercial Scale-Up
Advanced Catalytic Route for High-Purity Thiophene Derivatives and Commercial Scale-Up
The global demand for high-performance conductive polymers has necessitated a rigorous re-evaluation of monomer synthesis pathways, particularly for critical intermediates like 3,4-ethylenedioxythiophene (EDOT). Patent CN101910181A introduces a transformative manufacturing methodology that addresses the longstanding challenges of purity and yield in thiophene derivative production. This technical disclosure outlines a sophisticated two-step catalytic process that begins with the nucleophilic substitution of halogenated thiophenes and culminates in an acid-catalyzed ether exchange reaction. By integrating dynamic solvent removal strategies and specific aromatic sulfonic acid catalysts, this innovation enables the production of thiophene derivatives with gas chromatography purity exceeding 99.5%. For R&D directors and procurement specialists in the electronic materials sector, this represents a pivotal shift towards more reliable electronic chemical supplier capabilities, ensuring that the foundational monomers for next-generation optoelectronic devices meet the stringent quality standards required for commercial viability.
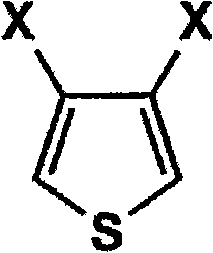
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of key thiophene derivatives such as 3,4-ethylenedioxythiophene has been plagued by inefficiencies inherent in traditional reflux methods. Conventional techniques often rely on heating 3,4-dimethoxythiophene with ethylene glycol in the presence of p-toluenesulfonic acid without effective byproduct management. This static approach frequently results in incomplete conversions and the accumulation of impurities that are notoriously difficult to separate due to similar boiling points and chemical properties. Furthermore, older methodologies for preparing the precursor 3,4-dimethoxythiophene often utilized metallic sodium in methanol with copper oxide catalysts, a process that introduces heavy metal contaminants and poses significant safety hazards regarding exothermic control. The resulting products from these legacy routes typically suffer from insufficient purity, which directly compromises the electrical conductivity and transparency of the final conductive polymers, rendering them unsuitable for high-end applications in solid electrolytic capacitors or advanced display technologies.
The Novel Approach
The patented process fundamentally reengineers the reaction thermodynamics by introducing a dynamic solvent removal system during the initial substitution phase. Instead of maintaining a constant solvent volume, the method involves reacting a halogenated thiophene with an alkali metal alkoxide in an alcoholic solvent while simultaneously distilling the solvent out of the reaction system. This clever manipulation increases the concentration of the alkali metal alkoxide from an initial range of 15% to 55% by weight, drastically accelerating the reaction kinetics and driving the equilibrium towards the desired dialkoxythiophene product. In the subsequent ether exchange step, the process employs specific aromatic sulfonic acids, such as xylenesulfonic acid or cumenesulfonic acid, rather than simple mineral acids. Coupled with a continuous azeotropic distillation loop that removes the byproduct alcohol (e.g., methanol) from the reaction zone, this approach ensures near-quantitative conversion rates. The result is a robust synthetic pathway that minimizes side reactions and polymerization, delivering a high-purity thiophene derivative that serves as a superior raw material for electronic chemical manufacturing.
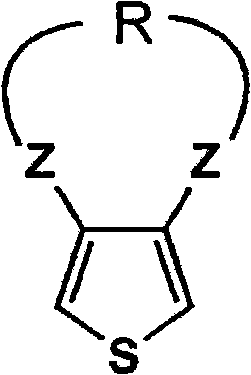
Mechanistic Insights into Acid-Catalyzed Ether Exchange and Substitution
The core of this innovation lies in the precise control of nucleophilic substitution and equilibrium shifts during the ether exchange reaction. In the first stage, the halogenated thiophene, represented by General Formula (1) where X can be fluorine, chlorine, bromine, or iodine, undergoes a nucleophilic attack by the alkoxide ion. The patent specifies that maintaining the alkali metal alkoxide concentration between 30% and 50% by weight at the completion of this step is critical for suppressing the formation of mono-substituted intermediates. By continuously removing the alcoholic solvent, the system prevents the reverse reaction and ensures that the concentration of the reactive species remains high enough to overcome steric hindrance at the 3 and 4 positions of the thiophene ring. This results in a dialkoxythiophene intermediate with halogenated thiophene residues reduced to below 0.05%, a level of purity that is exceptionally difficult to achieve with static batch processes.
In the second stage, the mechanism shifts to an acid-catalyzed transetherification where the dialkoxythiophene reacts with a diol compound, such as ethylene glycol. The choice of catalyst is paramount; the patent highlights the superiority of alkyl-substituted aromatic sulfonic acids over aliphatic sulfonic acids or inorganic acids. These specific catalysts effectively protonate the methoxy groups, facilitating their departure as methanol while preventing the acid-induced polymerization of the thiophene ring, a common degradation pathway. The continuous removal of methanol via azeotropic distillation with an aromatic solvent like toluene is the driving force that pushes the equilibrium towards the formation of the fused ring structure found in General Formula (5). This mechanistic understanding allows for the fine-tuning of reaction conditions, such as temperature ranges between 80°C and 150°C, to optimize yield while maintaining the structural integrity of the sensitive thiophene core.
How to Synthesize 3,4-Ethylenedioxythiophene Efficiently
The synthesis of 3,4-ethylenedioxythiophene (EDOT) via this patented route requires careful attention to solvent dynamics and catalyst selection to ensure optimal outcomes. The process begins with the preparation of 3,4-dimethoxythiophene through the reaction of 3,4-dibromothiophene with sodium methoxide, where the methanol solvent is distilled off to concentrate the reactants. Following the isolation of the high-purity intermediate, the material is subjected to an ether exchange reaction with ethylene glycol in the presence of an aromatic sulfonic acid catalyst and toluene. The detailed standardized synthesis steps, including specific molar ratios, temperature gradients, and distillation rates required for reproducibility, are outlined in the guide below.
- React halogenated thiophene with alkali metal alkoxide in alcoholic solvent while distilling off the solvent to increase concentration and drive the formation of dialkoxythiophene.
- Perform an ether exchange reaction between the dialkoxythiophene and a diol compound using an aromatic sulfonic acid catalyst.
- Remove byproduct alcohols via azeotropic distillation and washing, followed by vacuum distillation with polyhydric alcohols to prevent residue solidification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis technology offers substantial strategic benefits beyond mere chemical yield. The primary advantage lies in the drastic simplification of the purification train. Traditional methods often require extensive post-reaction processing to remove heavy metal catalysts like copper or to separate complex mixtures of mono- and di-substituted byproducts. By utilizing a catalyst system based on organic sulfonic acids and achieving high conversion rates through dynamic solvent removal, the need for expensive and time-consuming purification steps is significantly reduced. This streamlining of the downstream processing directly translates to cost reduction in display material manufacturing, as it lowers the consumption of solvents, energy, and labor associated with multi-stage recrystallization or chromatographic separation.
- Cost Reduction in Manufacturing: The elimination of heavy metal catalysts such as copper oxide, which were prevalent in prior art methods, removes the necessity for specialized heavy metal scavenging agents and the associated waste disposal costs. Furthermore, the high selectivity of the aromatic sulfonic acid catalysts minimizes the formation of oligomeric byproducts, thereby increasing the overall mass balance efficiency of the plant. This improved atom economy means that less raw material is wasted, and the throughput of the reactor is maximized, leading to a lower cost per kilogram of the final high-purity thiophene derivative without compromising on quality specifications.
- Enhanced Supply Chain Reliability: The robustness of this chemical process enhances supply chain continuity by reducing the risk of batch failures due to impurity spikes. The patent data indicates that the process is tolerant to variations in starting material quality, provided the halogenated thiophene is within specification, because the dynamic concentration adjustment compensates for minor fluctuations. Additionally, the use of commercially available and stable reagents like sodium methoxide and ethylene glycol ensures that the supply of raw materials is not subject to the volatility often seen with exotic or highly specialized reagents, securing a steady production flow for critical electronic chemical components.
- Scalability and Environmental Compliance: A standout feature of this technology is its proven scalability, as demonstrated by the successful operation of a 300L reaction tank described in the patent examples. The design incorporates a continuous azeotropic distillation and washing loop that efficiently manages byproduct removal without requiring excessive solvent volumes. This design is inherently safer and more environmentally friendly, as it reduces the total volume of organic waste generated per unit of product. The ability to maintain reaction times under 5 hours even at larger scales ensures that commercial scale-up of complex thiophene derivatives can be achieved rapidly, meeting the aggressive timelines of the consumer electronics and energy storage markets.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and performance of this thiophene derivative synthesis method. These insights are derived directly from the experimental data and process descriptions found within the patent documentation, providing clarity on purity benchmarks, catalyst selection, and operational parameters. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into existing manufacturing frameworks or for sourcing partners assessing the quality of the final monomer.
Q: What purity levels can be achieved with this thiophene derivative synthesis method?
A: The patented process consistently achieves gas chromatography purity levels exceeding 99.5%, with some embodiments reaching up to 99.8%, which is critical for high-performance conductive polymer applications.
Q: Why is the removal of alcohol solvent critical in the first reaction step?
A: Distilling off the alcoholic solvent during the reaction increases the concentration of the alkali metal alkoxide to a range of 15% to 55% by weight, significantly accelerating the reaction rate and minimizing halogenated thiophene residues to below 0.05%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates scalability using a 300L reaction tank with a continuous azeotropic distillation and washing loop, maintaining reaction times under 5 hours even at commercial scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4-Ethylenedioxythiophene Supplier
The technical advancements detailed in patent CN101910181A underscore the complexity and precision required to produce monomers for high-performance conductive polymers. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production necessary to bring such sophisticated chemistries to the global market. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of 3,4-ethylenedioxythiophene meets the exacting standards demanded by the semiconductor and display industries. We understand that consistency is key in electronic materials, and our process controls are designed to deliver that reliability batch after batch.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this high-efficiency process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will strengthen your supply chain and enhance the performance of your final electronic products.
