Advanced Manufacturing of 3,4-Ethylenedioxythiophene: A Cost-Effective Route for Global Supply Chains
The global demand for high-performance conductive polymers has surged, driven by applications in organic light-emitting diodes (OLEDs), solar cells, and antistatic coatings. At the heart of this revolution lies 3,4-ethylenedioxythiophene (EDOT), the critical monomer for poly(3,4-ethylenedioxythiophene) or PEDOT. Patent CN101062927B discloses a transformative preparation method that shifts the paradigm from expensive halogenated precursors to a cost-effective thiodiglycol-based route. This technical insight report analyzes the proprietary five-step synthesis—comprising esterification, condensation, Williamson etherification, hydrolysis, and decarboxylation—which delivers a total recovery rate exceeding 32% with exceptional purity. For R&D directors and procurement strategists, understanding this pathway is vital for securing a reliable electronic chemical supplier capable of meeting the rigorous standards of the optoelectronics industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of EDOT has been hindered by reliance on the Ullmann reaction, which utilizes 3,4-dibromothiophene as the primary starting material. This conventional approach presents severe economic and operational bottlenecks for commercial scale-up of complex electronic chemicals. The raw material, 3,4-dibromothiophene, is not only prohibitively expensive due to complex upstream bromination processes but also introduces significant supply chain volatility. Furthermore, the Ullmann coupling requires harsh reaction conditions, often involving high temperatures and stoichiometric amounts of copper, which leads to substantial waste generation and difficult downstream purification. The resulting low product yield and high production costs have traditionally confined this method to laboratory-scale synthesis, rendering it unsuitable for the mass manufacturing demands of the modern display and energy sectors.
The Novel Approach
In stark contrast, the novel methodology outlined in the patent leverages thiodiglycol, a readily available and inexpensive bulk chemical, as the foundational feedstock. This strategic shift fundamentally alters the cost structure of EDOT manufacturing by eliminating the dependency on scarce halogenated thiophenes. The process employs a logical sequence of classical organic transformations that are well-understood and easily controllable in a reactor setting. By breaking down the synthesis into five distinct stages—esterification, condensation, etherification, hydrolysis, and decarboxylation—the method allows for precise optimization at each juncture. This modularity not only enhances the overall yield but also facilitates the recycling of solvents like methanol and toluene, aligning with green chemistry principles and reducing the environmental footprint associated with specialty chemical production.
Mechanistic Insights into Thiodiglycol-Based Cyclization and Decarboxylation
The core of this synthesis lies in the construction of the thiophene ring and the subsequent fusion of the ethylenedioxy bridge. The process initiates with the acid-catalyzed esterification of thiodiglycol with methanol, yielding dimethyl thiodiglycolate with nearly 95% efficiency. This intermediate then undergoes a Dieckmann-type condensation with diethyl oxalate in the presence of sodium methylate. This step is critical as it forms the thiophene backbone, generating the 2,5-dicarbomethoxy-3,4-dihydroxythiophene intermediate. The reaction must be maintained under strictly anhydrous conditions at low temperatures (0-40°C) to prevent side reactions and ensure the structural integrity of the sensitive enediol moiety. Following this, the Williamson etherification closes the dioxane ring using 1,2-dihaloethane and potassium carbonate, facilitated by quaternary ammonium phase-transfer catalysts which enhance the nucleophilic substitution rate in organic solvents.
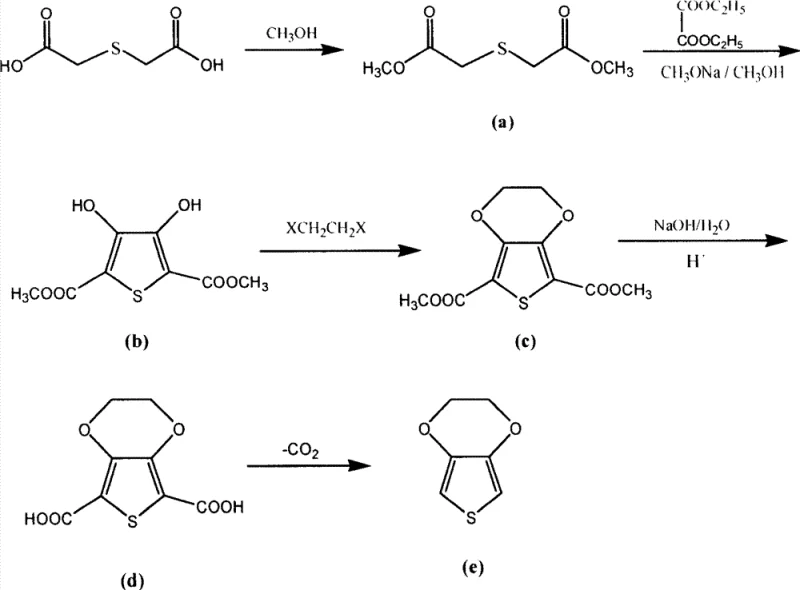
The final transformation involves the hydrolysis of the ester groups to carboxylic acids followed by thermal decarboxylation. The hydrolysis step utilizes aqueous sodium hydroxide to cleave the methyl esters, a robust reaction that proceeds with over 90% yield. The concluding decarboxylation is the most technically demanding step, requiring a copper-based catalyst (such as copper carbonate or hydroxide) in a high-boiling solvent like quinoline or DMSO. Under nitrogen protection at temperatures between 80°C and 200°C, the dicarboxylic acid loses carbon dioxide to aromatize and form the final EDOT product. The use of copper catalysts here is pivotal; they lower the activation energy for decarboxylation, allowing the reaction to proceed at manageable temperatures while minimizing thermal degradation of the sensitive thiophene ring, ultimately delivering a product with purity levels suitable for electronic applications.
How to Synthesize 3,4-Ethylenedioxythiophene Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters defined in the patent to maximize yield and purity. The process flow involves precise control of stoichiometry, particularly in the condensation and etherification steps where reagent ratios significantly impact the formation of by-products. Operators must ensure rigorous temperature management during the exothermic condensation phase and utilize high-efficiency phase-transfer catalysts to drive the etherification to completion. While the general workflow is established below, specific operational details regarding solvent volumes, catalyst loading percentages, and distillation cuts are critical for reproducibility.
- Esterify thiodiglycol with methanol using an acid catalyst to form dimethyl thiodiglycolate.
- Perform condensation with diethyl oxalate and sodium methylate to generate the dihydroxy thiophene intermediate.
- Execute Williamson etherification using 1,2-dihaloethane and a phase-transfer catalyst to close the dioxane ring.
- Hydrolyze the ester groups using aqueous sodium hydroxide to obtain the dicarboxylic acid derivative.
- Conduct thermal decarboxylation using a copper-based catalyst in a high-boiling solvent to yield pure EDOT.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this thiodiglycol-based route offers profound strategic advantages beyond simple unit cost metrics. The primary benefit stems from the decoupling of EDOT production from the volatile market of halogenated heterocycles. By utilizing thiodiglycol, a commodity chemical produced in vast quantities for other industries, manufacturers can secure a stable, long-term supply of raw materials that is immune to the shortages often plaguing specialized brominated intermediates. This stability translates directly into enhanced supply chain reliability, ensuring consistent delivery schedules for downstream polymer producers who cannot afford interruptions in their coating or film fabrication lines.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the drastic reduction in raw material expenses. Replacing expensive 3,4-dibromothiophene with low-cost thiodiglycol and diethyl oxalate creates a substantially lower cost basis for the final product. Additionally, the process design emphasizes solvent recovery; methanol and toluene can be distilled and reused multiple times, significantly lowering the variable costs associated with consumable inputs. The elimination of complex heavy metal removal steps, often required in palladium or nickel-catalyzed couplings, further simplifies the downstream processing infrastructure, reducing both capital expenditure on purification equipment and operational expenditure on waste treatment.
- Enhanced Supply Chain Reliability: The reliance on bulk chemicals ensures that production is not held hostage by the capacity constraints of niche fine chemical suppliers. Thiodiglycol and standard inorganic bases like potassium carbonate and sodium hydroxide are available from multiple global sources, mitigating the risk of single-source dependency. This diversification of the supply base allows for more flexible procurement strategies and better negotiation leverage. Furthermore, the robustness of the reaction conditions means that production can be scaled across different geographic locations without requiring highly specialized or rare reactor configurations, facilitating a more resilient and distributed manufacturing network.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this route is inherently cleaner. The avoidance of stoichiometric heavy metal waste and the ability to recycle solvents align with increasingly stringent global environmental regulations. The process operates at atmospheric or moderate pressures, removing the need for high-pressure autoclaves which pose safety risks and require expensive maintenance. The simplicity of the workup procedures—primarily filtration and distillation—means that the process can be scaled from pilot plants to multi-ton commercial reactors with minimal engineering hurdles, ensuring that capacity can be ramped up quickly to meet surging market demand for conductive polymers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this EDOT synthesis route. These insights are derived directly from the experimental data and process descriptions within the patent documentation, providing clarity on yield expectations, purity profiles, and operational requirements for potential manufacturing partners.
Q: Why is the thiodiglycol route preferred over the traditional Ullmann reaction for EDOT production?
A: The traditional Ullmann reaction relies on 3,4-dibromothiophene, which is prohibitively expensive and requires harsh reaction conditions leading to low yields. The thiodiglycol route utilizes inexpensive, bulk-available starting materials and operates under milder conditions, significantly improving economic feasibility for industrial scale-up.
Q: What represents the critical yield bottleneck in this five-step synthesis?
A: While individual steps like esterification and hydrolysis achieve yields near 95%, the Williamson etherification and final decarboxylation steps typically range between 65% and 70%. Optimizing phase-transfer catalysts in the etherification step and copper catalyst loading in decarboxylation is essential for maximizing overall throughput.
Q: How does this process ensure the high purity required for electronic applications?
A: The process incorporates high-vacuum fractional distillation in the final purification stage, collecting fractions at specific boiling points (75-77°C at 1.33 kPa). This effectively removes high-boiling solvent residues and by-products, ensuring the colorless, transparent liquid quality necessary for high-performance conductive polymer films.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4-Ethylenedioxythiophene Supplier
The technical viability of the thiodiglycol route underscores the immense potential for scaling EDOT production to meet the needs of the next generation of organic electronics. As a premier CDMO partner, NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with the advanced distillation columns and corrosion-resistant reactors necessary to handle the acidic and basic conditions of this five-step synthesis safely and efficiently. We maintain stringent purity specifications through our rigorous QC labs, ensuring that every batch of EDOT meets the exacting standards required for high-conductivity PEDOT applications in displays and photovoltaics.
We invite global partners to collaborate with us to optimize this supply chain further. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this route can improve your margin structure. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the rapidly evolving electronic materials market.
