Optimizing 1,5-Benzothiazepine Derivative Production for Global Pharmaceutical Supply Chains
The pharmaceutical industry constantly seeks robust synthetic routes for critical cardiovascular intermediates, specifically focusing on the efficient production of 1,5-benzothiazepine derivatives. Patent CN1052479C discloses a groundbreaking preparation method that addresses longstanding inefficiencies in the cyclization of propionic acid compounds to form these vital heterocyclic structures. This technology represents a significant leap forward in process chemistry, offering a pathway that dramatically enhances reaction kinetics and overall yield without compromising the stereochemical integrity required for bioactive molecules. For R&D directors and procurement specialists, understanding the nuances of this patented approach is essential for securing a reliable pharmaceutical intermediates supplier capable of meeting stringent global quality standards. The method leverages specific sulfonic acid catalysts to facilitate intramolecular ring-closing, transforming a traditionally sluggish process into a highly efficient operation suitable for modern commercial demands.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those described in Japanese patent gazette No. 18038/1978, typically relied on prolonged reflux conditions in solvents like xylene without the benefit of optimized acidic catalysis. These conventional processes often necessitated reaction times extending up to 12 hours to achieve complete conversion, which inherently limits manufacturing throughput and increases energy consumption significantly. Furthermore, the lack of precise catalytic control in these older methodologies frequently resulted in suboptimal yields, often hovering around 82%, which generates substantial waste and increases the cost per kilogram of the final active intermediate. The extended thermal exposure required by these legacy methods also poses risks to product stability, potentially leading to the formation of degradation byproducts that complicate downstream purification efforts. For supply chain heads, these inefficiencies translate directly into longer lead times and reduced flexibility in responding to market fluctuations for high-purity pharmaceutical intermediates.
The Novel Approach
The innovative methodology outlined in the patent introduces the strategic use of sulfonic acid compounds, such as p-toluenesulfonic acid or methanesulfonic acid, to catalyze the intramolecular cyclization reaction effectively. By incorporating these specific acidic promoters, the reaction time is drastically reduced from half a day to merely 30 minutes to 2 hours, depending on the specific solvent system and catalyst loading employed. This acceleration is achieved while simultaneously boosting the chemical yield to impressive levels, consistently exceeding 94% and reaching as high as 95.5% under optimal conditions. The process operates in high-boiling solvents like toluene or xylene, allowing for the continuous removal of water formed during the cyclization, which drives the equilibrium strongly towards the desired product formation. This refined approach not only maximizes resource utilization but also establishes a foundation for cost reduction in pharmaceutical intermediates manufacturing by minimizing solvent usage and energy input per batch.
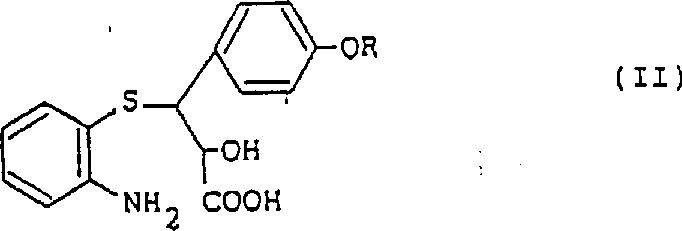
Mechanistic Insights into Sulfonic Acid-Catalyzed Cyclization
The core of this technological advancement lies in the mechanistic role played by the sulfonic acid catalyst during the intramolecular nucleophilic attack. The sulfonic acid protonates the hydroxyl group or the carbonyl oxygen of the propionic acid precursor, increasing the electrophilicity of the reaction center and facilitating the nucleophilic attack by the adjacent amino group. This activation lowers the activation energy barrier for the ring-closing step, allowing the reaction to proceed rapidly even at standard reflux temperatures without requiring extreme thermal conditions. The choice of catalyst, specifically aromatic sulfonic acids like p-toluenesulfonic acid, provides a balance of acidity and solubility that ensures homogeneous catalysis throughout the reaction medium. Understanding this mechanism is crucial for R&D teams aiming to replicate or scale this process, as it highlights the importance of maintaining anhydrous conditions to prevent catalyst deactivation and ensure consistent batch-to-batch reproducibility.
Impurity control is another critical aspect where this novel mechanism offers distinct advantages over traditional thermal cyclization methods. The rapid kinetics induced by the sulfonic acid catalyst minimize the residence time of reactive intermediates at elevated temperatures, thereby suppressing secondary decomposition pathways that often lead to colored impurities or polymeric byproducts. Additionally, the high selectivity of the acid-catalyzed pathway ensures that the stereochemistry of the chiral centers is preserved, which is vital for the biological activity of the resulting 1,5-benzothiazepine derivatives. The resulting crude product typically exhibits high purity, reducing the burden on crystallization and washing steps required to meet stringent pharmacopeial standards. For quality assurance professionals, this implies a more robust process with a narrower impurity profile, simplifying the validation of analytical methods and ensuring compliance with regulatory requirements for commercial scale-up of complex pharmaceutical intermediates.
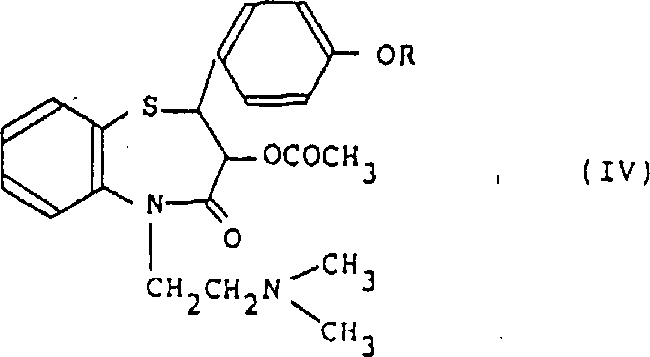
How to Synthesize 1,5-Benzothiazepine Derivatives Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry of the sulfonic acid catalyst and the efficient removal of water to drive the reaction to completion. The detailed standardized synthesis steps involve precise temperature control and specific workup procedures to isolate the crystalline product with maximum recovery. Operators must ensure that the reaction mixture is heated to reflux in a suitable solvent system while employing a Dean-Stark trap or similar apparatus to azeotropically remove the water generated during the cyclization. Following the reaction, the mixture is cooled to induce crystallization, and the solid product is collected via filtration and washed with cold ethanol to remove residual acid and solvent traces.
- Prepare the reaction mixture by combining the propionic acid precursor with a specific sulfonic acid catalyst such as p-toluenesulfonic acid in a high-boiling solvent like xylene.
- Heat the mixture to reflux temperature while continuously removing generated water to drive the equilibrium towards the cyclized product.
- Upon completion, cool the reaction mixture, filter the precipitated crystals, and wash with ethanol to isolate the high-purity benzothiazepine derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis method offers transformative benefits for procurement managers and supply chain leaders focused on efficiency and cost containment. The drastic reduction in reaction time directly correlates to increased plant capacity, allowing manufacturers to produce more batches within the same timeframe without additional capital investment in reactor volume. This enhanced throughput capability is essential for mitigating supply risks and ensuring continuity of supply for downstream drug manufacturers who rely on just-in-time delivery models for their production schedules. Furthermore, the significant improvement in chemical yield means that less raw material is required to produce the same amount of final product, leading to substantial cost savings in raw material procurement and waste disposal management. These operational efficiencies collectively contribute to a more competitive pricing structure and a more resilient supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of prolonged heating cycles and the improvement in yield from approximately 82% to over 95% fundamentally alters the cost structure of the manufacturing process. By reducing the energy consumption associated with maintaining reflux for 12 hours down to just 1 or 2 hours, utility costs are significantly lowered per kilogram of product produced. Additionally, the higher yield reduces the effective cost of goods sold by maximizing the output from each unit of expensive starting material, which is a critical factor in maintaining margin stability in the volatile fine chemical market. The simplified workup procedure also reduces labor hours and solvent consumption during purification, further driving down the overall operational expenditure required to bring this intermediate to market readiness.
- Enhanced Supply Chain Reliability: The robustness of this catalytic method ensures consistent batch quality and predictable production timelines, which are paramount for maintaining trust with global pharmaceutical clients. Shorter cycle times mean that inventory can be replenished more rapidly in response to unexpected demand spikes, reducing the need for excessive safety stock and freeing up working capital. The use of common, commercially available reagents like p-toluenesulfonic acid and toluene minimizes the risk of supply disruptions associated with specialty catalysts or exotic solvents that might face logistical bottlenecks. This reliability makes the manufacturer a preferred partner for long-term contracts where supply security is valued as highly as price competitiveness in the global sourcing strategy.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the use of standard unit operations and non-hazardous catalysts that are easy to handle in large volumes. The reduction in reaction time and the high conversion rate minimize the generation of waste streams, aligning with modern green chemistry principles and reducing the environmental footprint of the manufacturing facility. Easier waste management translates to lower compliance costs and reduced regulatory scrutiny, allowing the supply chain to operate smoothly without interruptions related to environmental permitting or waste treatment capacity constraints. This scalability ensures that the technology can support the commercial scale-up of complex pharmaceutical intermediates from pilot plant quantities to multi-ton annual production volumes seamlessly.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this specific synthesis technology for 1,5-benzothiazepine derivatives. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and relevance for industry stakeholders. Understanding these details helps potential partners evaluate the feasibility of integrating this intermediate into their own supply chains or development pipelines with confidence.
Q: What is the primary advantage of using sulfonic acid catalysts in this synthesis?
A: The use of sulfonic acids like p-toluenesulfonic acid significantly accelerates the intramolecular cyclization rate, reducing reaction time from over 12 hours to under 2 hours while improving yields to over 95%.
Q: How does this method impact impurity profiles compared to conventional techniques?
A: The optimized conditions minimize side reactions and degradation, resulting in a cleaner crude product that requires less intensive purification, thereby enhancing overall process efficiency and purity specifications.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the method utilizes common solvents like toluene or xylene and standard reflux equipment, making it highly scalable for industrial production without requiring exotic reagents or complex pressure systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Benzothiazepine Derivatives Supplier
NINGBO INNO PHARMCHEM stands ready to leverage this advanced synthetic technology to deliver high-quality intermediates that meet the rigorous demands of the global pharmaceutical industry. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from development to full-scale manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 1,5-benzothiazepine derivatives meets the exacting standards required for subsequent drug substance synthesis. We understand the critical nature of cardiovascular intermediates and are committed to providing a supply solution that balances technical excellence with commercial viability.
We invite you to engage with our technical procurement team to discuss how this optimized process can benefit your specific project requirements and cost structures. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic advantages of switching to this high-yield manufacturing route for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume needs. Partnering with us ensures access to a reliable pharmaceutical intermediates supplier dedicated to driving innovation and efficiency in your drug development journey.
