Advanced One-Step Cyclization for High-Purity 1,5-Benzothiazepine Intermediates
The pharmaceutical industry continuously seeks robust synthetic pathways for critical cardiovascular medications, particularly calcium channel blockers like Diltiazem Hydrochloride. Patent CN1160713A introduces a groundbreaking methodology for the preparation of 1,5-benzothiazepine derivatives, which serve as pivotal intermediates in this therapeutic class. This innovation focuses on a novel one-step intramolecular cyclization of specific 3-(2-amino-substituted or unsubstituted phenylthio)-2-hydroxy-3-phenylpropionamide compounds. By bypassing traditional multi-step ester hydrolysis protocols, this technique offers a direct route to the benzothiazepine core with exceptional efficiency. For R&D directors and process chemists, understanding this mechanism is vital for optimizing impurity profiles and ensuring consistent batch quality. The ability to generate these complex heterocyclic structures in a single operational unit represents a significant leap forward in process chemistry, promising enhanced throughput and reduced environmental footprint for large-scale manufacturing facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cis-3-hydroxy-2,3-dihydro-2-(4-methoxyphenyl)-1,5-benzothiazepine-4(5H)-one has relied heavily on ester-based precursors. Conventional routes typically involve the reaction of 2-aminothiophenol with methyl 3-(4-methoxyphenyl)-2,3-epoxypropionate, followed by a mandatory hydrolysis step to convert the ester into a carboxylic acid before cyclization can occur. This multi-stage approach introduces significant inefficiencies, including the need for additional reagents, extended reaction times, and complex workup procedures to remove hydrolysis byproducts. Furthermore, the handling of ester intermediates often necessitates strict moisture control and can lead to variable yields due to competing side reactions during the hydrolysis phase. From a supply chain perspective, the reliance on multiple discrete steps increases the risk of material loss and complicates the validation of the manufacturing process, ultimately driving up the cost of goods sold for the final active pharmaceutical ingredient.
The Novel Approach
In stark contrast, the methodology disclosed in CN1160713A utilizes novel propionamide derivatives as the direct cyclization substrates. This approach fundamentally alters the reaction landscape by enabling the formation of the 1,5-benzothiazepine nucleus directly from the amide without prior hydrolysis. The process is remarkably versatile, capable of proceeding either thermally or under mild acid catalysis using agents like methanesulfonic acid or p-toluenesulfonic acid. A distinct advantage arises when the amide nitrogen substituents are hydrogen atoms; in this scenario, ammonia is evolved as a gas during the ring closure, effectively driving the equilibrium toward product formation without the need for complex separation techniques. This streamlined protocol not only reduces the number of unit operations but also significantly simplifies the purification workflow, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
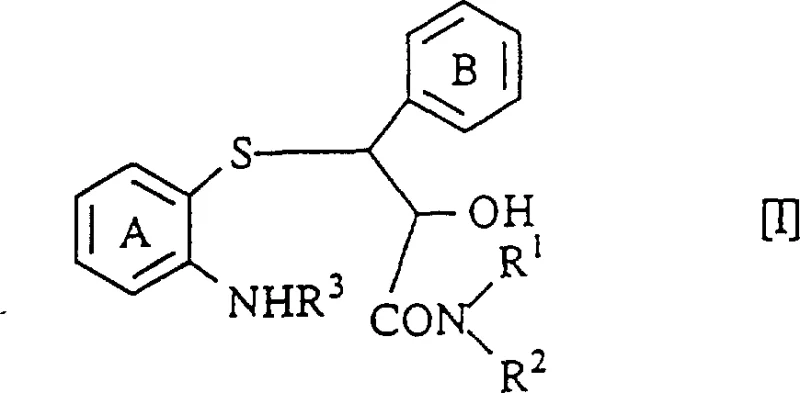
Mechanistic Insights into Acid-Catalyzed Intramolecular Cyclization
The core of this technological advancement lies in the unique reactivity of the 3-(2-aminophenylthio)-2-hydroxy-3-phenylpropionamide scaffold. Unlike esters, which are generally more susceptible to nucleophilic attack, amides are traditionally considered less reactive due to resonance stabilization. However, under the specific thermal and acidic conditions described in the patent, the amide carbonyl becomes sufficiently activated to undergo nucleophilic attack by the adjacent ortho-amino group on the aromatic ring. This intramolecular condensation results in the expulsion of water or ammonia, depending on the substitution pattern, and the formation of the seven-membered benzothiazepine ring. The presence of the 2-hydroxyl group on the propionamide chain plays a crucial role in stabilizing the transition state and ensuring the correct stereochemical outcome. Detailed analysis suggests that the reaction proceeds through a concerted mechanism that preserves the chiral integrity of the starting material, which is paramount for producing biologically active isomers.
Impurity control is another critical aspect where this mechanism excels. By avoiding the harsh basic conditions often required for ester hydrolysis, the potential for epimerization at the chiral centers is minimized. The patent data highlights that optically active starting materials yield optically active products with high enantiomeric excess, often exceeding 99% ee when analyzed by chiral HPLC. This stereochemical fidelity is achieved through careful selection of reaction parameters, including temperature ranges between 80°C and 200°C and the use of non-nucleophilic solvents like xylene or chlorobenzene. The robustness of this cyclization against racemization ensures that the final intermediate meets the stringent purity specifications required for downstream conversion into Diltiazem Hydrochloride, thereby reducing the burden on downstream purification processes and enhancing overall process reliability.
How to Synthesize 1,5-Benzothiazepine Derivatives Efficiently
Implementing this synthesis requires precise control over reaction conditions to maximize yield and purity. The process begins with the preparation of the novel propionamide precursor, which can be synthesized by reacting 2-aminothiophenol with a 2,3-epoxypropionamide derivative, potentially utilizing an iron catalyst to facilitate the ring-opening step. Once the precursor is obtained, it is subjected to cyclization in a high-boiling solvent system. The detailed standardized synthesis steps below outline the specific operational parameters derived from the patent examples, ensuring reproducibility and safety during scale-up. Operators should pay close attention to the stoichiometry of the acid catalyst and the reflux duration, as these variables directly influence the conversion rate and the profile of residual impurities.
- Prepare the novel 3-(2-aminophenylthio)-2-hydroxy-3-phenylpropionamide derivative by reacting 2-aminothiophenol with a 2,3-epoxypropionamide derivative, optionally using an iron catalyst.
- Suspend the propionamide derivative in a high-boiling solvent such as xylene, chlorobenzene, or toluene within a reaction vessel equipped for reflux.
- Add a catalytic amount of acid, such as methanesulfonic acid or p-toluenesulfonic acid, and heat the mixture to reflux (80-200°C) to induce intramolecular cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this cyclization technology offers tangible strategic benefits beyond mere technical elegance. The elimination of the ester hydrolysis step translates directly into a reduction in raw material consumption and waste generation. By consolidating what was previously a multi-step sequence into a single pot operation, manufacturers can significantly decrease the total processing time per batch. This efficiency gain allows for better utilization of reactor capacity and reduces the energy load associated with heating, cooling, and agitation across multiple stages. Furthermore, the solvents employed in this process, such as xylene and toluene, are commodity chemicals with stable global supply chains, mitigating the risk of raw material shortages that often plague specialized reagent markets. These factors collectively contribute to a more resilient and cost-effective supply chain for critical cardiovascular intermediates.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of the hydrolysis unit operation. Traditional methods require additional reagents for saponification and subsequent acidification, along with the associated waste treatment costs for salt byproducts. By utilizing the direct amide cyclization route, these material costs are entirely eliminated. Additionally, the evolution of ammonia gas in specific embodiments acts as a self-driving force for the reaction, reducing the need for expensive coupling agents or dehydrating reagents. This simplification of the bill of materials leads to substantial cost savings in the overall production of the intermediate, allowing for more competitive pricing in the global marketplace without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes significantly to supply continuity. The process tolerates a wide range of temperatures and uses common industrial solvents that are readily available from multiple suppliers worldwide. This flexibility reduces dependency on single-source vendors for exotic reagents. Moreover, the high yield and purity achievable in a single step minimize the need for reprocessing or recycling of off-spec material, ensuring a steady flow of qualified intermediate to the next stage of production. For supply chain planners, this predictability is invaluable for maintaining inventory levels and meeting delivery commitments to downstream API manufacturers, especially in times of market volatility.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of sensitive intermediates that require cryogenic conditions or inert atmospheres beyond standard nitrogen blanketing. The use of high-boiling solvents facilitates easy product isolation via crystallization upon cooling, a unit operation that scales linearly and efficiently. From an environmental perspective, the reduction in step count inherently lowers the E-factor (mass of waste per mass of product). The avoidance of aqueous hydrolysis steps reduces the volume of wastewater requiring treatment, aligning the manufacturing process with increasingly stringent environmental regulations and sustainability goals pursued by modern pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this 1,5-benzothiazepine synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a reliable foundation for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into existing manufacturing frameworks. Whether the concern is regarding stereochemical integrity, solvent recovery, or catalyst loading, the answers below reflect the proven capabilities of the described methodology.
Q: What are the advantages of using propionamide derivatives over propionate esters for this synthesis?
A: Using propionamide derivatives eliminates the need for a separate hydrolysis step required for esters. Furthermore, when the amide substituents are hydrogen atoms, ammonia is released as a gas during cyclization, driving the reaction forward and simplifying purification compared to ester-based routes.
Q: Can this process maintain optical purity for chiral intermediates?
A: Yes, the patent data confirms that when optically active propionamide derivatives are used, the resulting 1,5-benzothiazepine products retain their optical activity without racemization, achieving high enantiomeric excess suitable for calcium channel blocker production.
Q: Which solvents are preferred for scaling up this cyclization reaction?
A: High-boiling solvents are preferred to facilitate the reaction kinetics at elevated temperatures. Specifically, xylene, chlorobenzene, dichlorobenzene, and toluene are identified as optimal choices, allowing for efficient reflux conditions between 80°C and 200°C.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Benzothiazepine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development and production of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial reality is seamless. We are committed to delivering 1,5-benzothiazepine derivatives that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging advanced synthetic strategies like the one described in CN1160713A, we can offer our partners a secure and efficient source of supply that aligns with their cost and quality objectives.
We invite you to engage with our Customized Cost-Saving Analysis service to explore how this optimized synthesis route can benefit your specific project requirements. Our technical procurement team is ready to provide specific COA data and route feasibility assessments tailored to your needs. By collaborating with us, you gain access to a partner dedicated to innovation and excellence in fine chemical manufacturing. Contact us today to discuss your supply chain optimization strategies and secure a reliable source for your pharmaceutical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
