Scaling High-Optical-Purity Indoxacarb Intermediates with Novel Cinchonine Derivatives
The global demand for high-efficiency agrochemicals is driving a critical shift towards single-enantiomer active ingredients, particularly for broad-spectrum insecticides like Indoxacarb. Patent CN114105972A introduces a transformative approach to this challenge by disclosing a novel class of cinchonine derivatives that serve as highly efficient organocatalysts. Historically, the industrial synthesis of the key Indoxacarb intermediate, (S)-5-chloro-2-methoxycarbonyl-2-hydroxy-1-indanone, has been plagued by poor stereocontrol, often yielding racemic mixtures or low enantiomeric excess (ee) values that necessitate costly recycling or purification steps. This patent addresses these bottlenecks by modifying the C-9 hydroxyl position of the cinchonine backbone through amidation, resulting in catalysts that dramatically enhance both yield and enantioselectivity in asymmetric alpha-hydroxylation reactions. The strategic structural evolution presented in this intellectual property not only solves a long-standing synthetic problem but also opens new avenues for reliable agrochemical intermediate suppliers seeking to optimize their manufacturing portfolios.
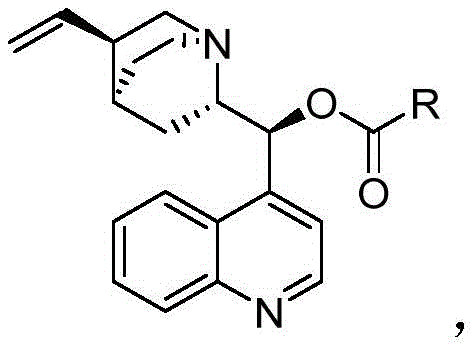
The significance of this technological leap cannot be overstated for the fine chemical sector. By achieving an S-isomer content of greater than 90 percent, compared to the mere 50 percent obtained with unmodified cinchonine, this innovation directly translates to higher quality end-products and reduced waste streams. For R&D directors and process chemists, the ability to access such high optical purity without resorting to complex transition metal complexes or harsh reaction conditions represents a paradigm shift. The simplicity of the catalyst design, coupled with its robust performance in the presence of peroxide oxidants, suggests a mature technology ready for immediate evaluation in pilot and commercial plants. This report delves deep into the mechanistic advantages, synthetic accessibility, and commercial implications of adopting this novel catalytic system for the production of high-value chiral building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing the chiral indanone intermediate of Indoxacarb have faced significant hurdles regarding efficiency and environmental impact. The legacy industrial process, famously utilized by major agrochemical corporations, relied on unmodified cinchonine alkaloids which yielded an enantiomeric excess of only around 50 percent. This low selectivity meant that half of the produced material was the inactive R-isomer, effectively doubling the raw material consumption for the active ingredient and creating a massive burden on downstream separation processes. Furthermore, alternative academic approaches involving chiral phosphine-Cu(I) complexes, Salen-Zr polymers, or tartaric acid-derived guanidines often suffered from prohibitive costs, toxicity concerns associated with heavy metals, and instability under process conditions. These conventional routes frequently required stringent exclusion of moisture and oxygen, specialized equipment, and complex ligand synthesis, rendering them economically unviable for multi-ton scale production. The cumulative effect of these limitations was a constrained supply capacity that struggled to meet the growing global demand for high-purity Indoxacarb, capping production potential at levels far below market requirements.
The Novel Approach
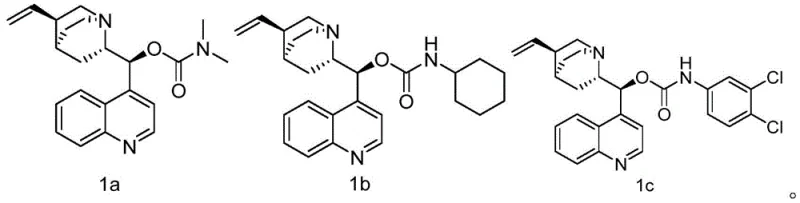
In stark contrast, the novel approach detailed in the patent data leverages a rationally designed modification of the naturally abundant cinchonine scaffold to overcome these historical barriers. By introducing bulky amide or carbamate groups at the C-9 position, the new catalysts (specifically formulas 1a, 1b, and 1c) create a highly defined chiral pocket that directs the incoming oxidant with exceptional precision. This structural refinement allows the reaction to proceed with enantioselectivity exceeding 90 percent ee, effectively doubling the efficiency of the chiral induction compared to the parent alkaloid. The versatility of this approach is evident in the variety of substituents tolerated, ranging from dimethylamino groups to cyclohexyl and dichlorophenyl moieties, allowing process chemists to fine-tune solubility and reactivity profiles. Moreover, the synthesis of these advanced catalysts is remarkably straightforward, involving a direct one-step derivatization that avoids the multi-step sequences typical of bespoke ligand construction. This simplicity ensures that the cost of goods for the catalyst remains low, facilitating its adoption in cost-sensitive agrochemical manufacturing environments.
Mechanistic Insights into Asymmetric Alpha-Hydroxylation Catalysis
The superior performance of these cinchonine derivatives stems from a sophisticated interplay of steric hindrance and hydrogen bonding interactions within the catalytic cycle. In the asymmetric alpha-hydroxylation of the 5-chloro-1-indanone-2-methyl formate substrate, the quinuclidine nitrogen of the catalyst likely acts as a general base to activate the substrate or stabilize the transition state, while the bulky C-9 substituent shields one face of the enolate intermediate. This facial discrimination is crucial for ensuring that the peroxide oxidant attacks exclusively from the Si-face (or Re-face, depending on the specific absolute configuration desired), thereby locking in the S-configuration with high fidelity. The rigidity introduced by the urea or carbamate linkage at the C-9 position prevents free rotation that might otherwise blur the chiral information, a common failure mode in flexible organocatalysts. Additionally, the aromatic quinoline ring may participate in pi-stacking interactions with the substrate, further anchoring it in the optimal orientation for reaction. Understanding these mechanistic nuances allows manufacturers to predict how slight variations in solvent polarity or temperature might influence the outcome, providing a robust framework for process optimization.
From an impurity control perspective, this organocatalytic route offers distinct advantages over transition metal-catalyzed alternatives. The absence of copper, zirconium, or other heavy metals eliminates the risk of metal leaching into the final product, a critical quality attribute for agrochemical registration and environmental compliance. Metal residues often require expensive scavenging resins or additional crystallization steps to reduce to ppm levels, adding both time and cost to the manufacturing timeline. By utilizing a metal-free organic catalyst, the impurity profile is significantly simplified, consisting primarily of organic byproducts that are easier to separate via standard aqueous workups or crystallization. The patent data indicates that the reaction can be driven to high conversion with minimal side reactions, suggesting a clean reaction pathway that maximizes atom economy. This cleanliness is further supported by the use of relatively benign oxidants like tert-butyl peroxide, which decomposes into volatile byproducts, leaving behind a high-purity solid product after simple concentration and filtration.
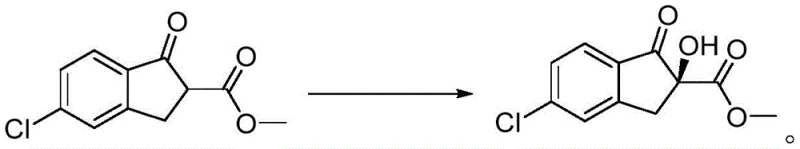
How to Synthesize (S)-5-Chloro-2-methoxycarbonyl-2-hydroxy-1-indanone Efficiently
The implementation of this catalytic technology in a production setting follows a logical and scalable protocol that minimizes operational complexity. The process begins with the preparation of the catalyst itself, which can be stockpiled in advance due to its stability, followed by the main hydroxylation reaction which operates under mild thermal conditions. The key to success lies in maintaining strict temperature control during the oxidant addition phase to prevent exothermic runaway and preserve enantioselectivity. Detailed standardized operating procedures for the synthesis of the catalyst and its subsequent application in the hydroxylation reaction are outlined below, providing a clear roadmap for technical teams to replicate the high yields and ee values reported in the patent literature. This step-by-step guidance ensures that the transition from laboratory bench to pilot plant is seamless, reducing the risk of scale-up failures.
- Dissolve cinchonine in anhydrous tetrahydrofuran under nitrogen protection and cool the mixture to approximately 5°C.
- Add sodium hydride (NaH) in batches as the base, stirring for 30 minutes to ensure complete deprotonation of the hydroxyl group.
- Dropwise add the derivatizing agent (dimethylcarbamoyl chloride or substituted isocyanate) and stir at room temperature for 5 to 12 hours to form the final catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel cinchonine derivative technology presents a compelling value proposition centered on cost reduction and supply security. The primary economic driver is the drastic reduction in catalyst loading; whereas older methods might require stoichiometric or near-stoichiometric amounts of chiral auxiliaries, this system operates effectively at loadings as low as 0.02 to 0.03 molar equivalents. This high turnover number means that a small quantity of catalyst can produce a vast amount of product, significantly lowering the unit cost of the active intermediate. Furthermore, the raw materials for the catalyst—cinchonine and simple isocyanates or carbamoyl chlorides—are commodity chemicals with stable global supply chains, insulating manufacturers from the volatility often associated with specialized chiral ligands. The elimination of expensive transition metals also removes the need for costly metal scavengers and the associated waste disposal fees, contributing to a leaner and more sustainable cost structure.
- Cost Reduction in Manufacturing: The economic benefits extend beyond just the price of the catalyst. By achieving high enantioselectivity directly in the reaction, the need for costly resolution steps or recycling of the unwanted isomer is largely eliminated. This streamlining of the process flow reduces the number of unit operations, saving on energy consumption, solvent usage, and labor hours. The simplified workup procedure, which involves basic aqueous washes and crystallization, avoids the need for complex chromatography or distillation columns that are difficult to scale. Consequently, the overall cost of goods sold (COGS) for the Indoxacarb intermediate is substantially reduced, enhancing the competitiveness of the final agrochemical product in the global market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and simplicity of the synthetic route. The reaction conditions are mild, utilizing common solvents like toluene and operating at near-ambient or slightly cooled temperatures, which reduces the strain on utility systems and equipment. The catalyst itself is stable and easy to handle, posing fewer logistical challenges than air-sensitive metal complexes that require inert atmosphere shipping and storage. This ease of handling ensures consistent quality across different batches and manufacturing sites, reducing the risk of supply disruptions due to technical failures. Moreover, the high yield of the process ensures that less raw material is wasted, maximizing the output from existing reactor volumes and improving overall asset utilization.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this metal-free organocatalytic process aligns perfectly with modern green chemistry principles. The absence of heavy metals simplifies regulatory compliance and wastewater treatment, as there is no risk of toxic metal contamination in the effluent. The use of organic peroxides requires careful handling, but the low catalyst loading and controlled addition rates mitigate the risks of thermal runaway. The process generates minimal hazardous waste, and the solvents used are readily recoverable and recyclable. This environmental friendliness not only reduces disposal costs but also enhances the corporate sustainability profile, a factor increasingly important to downstream customers and regulatory bodies. The scalability is proven by the successful demonstration of the reaction on meaningful scales in the patent examples, indicating readiness for multi-ton commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a reliable basis for decision-making. Understanding these specifics helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing infrastructure.
Q: How does the new cinchonine derivative improve enantioselectivity compared to standard cinchonine?
A: By amidating the hydroxyl group at the C-9 position, the new derivatives create a more rigid and sterically demanding chiral environment, boosting the S-isomer content from 50% to over 90%.
Q: What are the typical reaction conditions for the asymmetric alpha-hydroxylation using these catalysts?
A: The reaction typically proceeds in toluene at low temperatures (-5°C to 0°C) using tert-butyl peroxide as the oxidant, requiring only 0.02 to 0.03 molar equivalents of the catalyst.
Q: Is this catalytic system suitable for large-scale industrial production?
A: Yes, the catalyst synthesis is a simple one-step reaction with high yields, and the catalytic loading is extremely low, making it highly cost-effective and scalable for ton-level manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indoxacarb Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the catalytic technologies described in patent CN114105972A for the agrochemical industry. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into robust industrial processes. Our team of expert process chemists is well-versed in asymmetric organocatalysis and can rapidly optimize these cinchonine derivative protocols to meet your specific throughput and purity requirements. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC and NMR capabilities to guarantee that every batch of Indoxacarb intermediate meets the highest global standards for optical purity and chemical quality.
We invite you to collaborate with us to leverage this cutting-edge technology for your supply chain. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this novel catalytic route for your specific volume needs. We encourage you to reach out today to discuss specific COA data and route feasibility assessments tailored to your project timelines. Let us help you secure a competitive advantage through superior chemistry and reliable supply.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
