Revolutionizing Cephalosporin Stability: Commercial Scale-up of Ceftriaxone Sodium Tetrahydrate
Revolutionizing Cephalosporin Stability: Commercial Scale-up of Ceftriaxone Sodium Tetrahydrate
In the highly regulated landscape of beta-lactam antibiotic manufacturing, the physical stability of the active pharmaceutical ingredient (API) is often just as critical as its chemical potency. Patent CN109134503B introduces a groundbreaking advancement in this domain by disclosing a novel crystalline form of Ceftriaxone Sodium, specifically defined as a tetrahydrate containing four moles of water per mole of the drug substance. This innovation addresses long-standing industry pain points associated with the conventional 3.5 hydrate form, particularly regarding hygroscopicity and powder flow characteristics. For R&D directors and supply chain managers alike, this patent represents a pivotal shift towards more robust manufacturing processes. By stabilizing the crystal lattice through precise hydration control, the technology promises to reduce batch-to-batch variability and enhance the shelf-life of the final injectable formulations. As a leading entity in fine chemical synthesis, understanding the nuances of this tetrahydrate structure is essential for maintaining competitiveness in the global cephalosporin market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the commercial production of Ceftriaxone Sodium has relied heavily on the 3.5 hydrate crystalline form, which, while effective therapeutically, presents significant logistical and processing challenges. The primary deficiency lies in its inherent hygroscopic nature; under standard storage conditions, the 3.5 hydrate tends to absorb atmospheric moisture, leading to caking and agglomeration. This physical instability complicates downstream processing operations such as milling, blending, and encapsulation, often requiring stringent environmental controls that drive up operational expenditures. Furthermore, the conventional crystallization processes frequently result in products with higher levels of related substances and impurities, necessitating additional purification steps that erode overall yield. The poor flowability, characterized by a high angle of repose, further exacerbates these issues, causing inconsistent dosing and potential equipment blockages in high-speed automated lines. These cumulative inefficiencies create a bottleneck for manufacturers seeking to optimize cost structures while adhering to increasingly rigorous quality standards.
The Novel Approach
The methodology outlined in patent CN109134503B offers a sophisticated solution by engineering a tetrahydrate crystal structure that fundamentally alters the physicochemical profile of the API. By incorporating a fourth molecule of water into the crystal lattice, the inventors have created a thermodynamically more stable arrangement that resists further moisture uptake from the environment. This structural modification results in a material with exceptionally low hygroscopicity, ensuring that the powder remains free-flowing even under elevated relative humidity conditions. The synthesis route is designed to be operationally simple, utilizing mild reaction temperatures and readily accessible reagents, which contrasts sharply with the harsh conditions often required for older methods. This approach not only simplifies the manufacturing workflow but also inherently suppresses the formation of degradation byproducts, leading to a final product with purity levels exceeding 99%. For procurement teams, this translates to a raw material that is easier to handle, store, and process, effectively de-risking the supply chain.
Mechanistic Insights into Crystal Hydrate Stabilization
The superior performance of this novel compound stems from the precise stoichiometric integration of water molecules within the crystal lattice. In the tetrahydrate form, the four water molecules act as structural pillars, forming an extensive network of hydrogen bonds that lock the ceftriaxone sodium molecules into a rigid, stable configuration. This tight packing prevents the intrusion of external water vapor, thereby eliminating the deliquescence issues common to the 3.5 hydrate. The structural integrity is further evidenced by thermogravimetric analysis, which shows a weight loss consistent with exactly four moles of water, confirming the stability of the hydrate up to significant temperatures. From a mechanistic standpoint, the synthesis employs a specific activation strategy where aminothiazolyloxime acetic acid is activated using sulfonyl chloride in the presence of dimethylformamide. This generates a highly reactive intermediate that couples efficiently with the 7-ACT tetramethylguanidine salt. The use of the tetramethylguanidine salt is particularly ingenious, as it enhances the nucleophilicity of the 7-amino group, facilitating a cleaner acylation reaction with minimal racemization or side reactions.
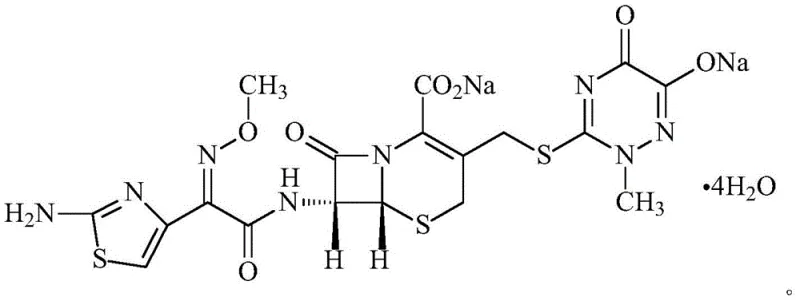
Following the coupling reaction, the crystallization process is meticulously controlled to ensure the exclusive formation of the tetrahydrate polymorph. Parameters such as pH adjustment, solvent composition (water/methanol/acetone), and cooling rates are optimized to favor the nucleation of the desired crystal habit. The resulting crystals exhibit a distinct morphology that contributes to their excellent flow properties, as quantified by a low angle of repose. This control over the solid-state form is critical for R&D teams aiming to file robust regulatory dossiers, as polymorphic consistency is a key requirement for drug approval. The mechanism essentially bypasses the kinetic traps that lead to amorphous or metastable hydrate forms, driving the system directly to the thermodynamic minimum represented by the stable tetrahydrate. This level of control ensures that every batch produced meets the stringent specifications required for parenteral administration.
How to Synthesize Ceftriaxone Sodium Efficiently
The synthesis of this high-stability Ceftriaxone Sodium Tetrahydrate is achieved through a refined four-step sequence that balances reaction kinetics with crystallization thermodynamics. The process begins with the activation of the side chain, followed by coupling with the cephalosporin nucleus, and concludes with a carefully managed salt formation and precipitation step. Each stage is optimized to maximize yield while minimizing the generation of impurities, ensuring a final product that meets the highest pharmacopeial standards. The detailed standardized synthesis steps, including specific molar ratios, temperature profiles, and stirring speeds, are provided in the guide below to assist technical teams in replicating this advanced process.
- Activate aminothiazolyloxime acetic acid using sulfonyl chloride and DMF in dichloromethane to form a reactive intermediate solution.
- Prepare 7-aminocephalosporanetrizine (7-ACT) tetramethylguanidine salt in dichloromethane under cooling conditions to ensure stability.
- Couple the activated side chain with the 7-ACT salt, adjust pH, decolorize, and crystallize the ceftriaxone acid intermediate.
- Convert the acid to the sodium salt using sodium hydroxide in a water-methanol mixture, precipitate with acetone, and dry to obtain the tetrahydrate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this tetrahydrate technology offers tangible strategic benefits that extend beyond mere chemical specification compliance. The enhanced physical stability of the material directly correlates with reduced waste and lower logistics costs. Because the compound is non-hygroscopic, it does not require the expensive, energy-intensive climate-controlled warehousing that the 3.5 hydrate often demands. This relaxation of storage constraints allows for greater flexibility in distribution networks and reduces the risk of product rejection due to moisture-related specification failures during transit. Furthermore, the improved flowability of the powder ensures smoother operation in downstream formulation plants, reducing machine downtime caused by bridging or rat-holing in hoppers. These operational efficiencies accumulate to provide a significant competitive advantage in a margin-sensitive market.
- Cost Reduction in Manufacturing: The synthesis route described in the patent eliminates the need for complex purification cycles often required to remove high levels of impurities found in conventional batches. By achieving high purity directly from the crystallization step, manufacturers can significantly reduce solvent consumption and waste disposal costs. Additionally, the high yield and simplified operational parameters mean that production throughput can be increased without proportional increases in capital expenditure. The removal of aggressive drying steps, which are often necessary to manage moisture in unstable hydrates, further contributes to energy savings and equipment longevity.
- Enhanced Supply Chain Reliability: The thermodynamic stability of the tetrahydrate form ensures a longer shelf life, allowing for larger inventory buffers without the fear of degradation. This reliability is crucial for maintaining continuous supply to global markets, especially in regions with challenging climatic conditions. The robustness of the crystal form minimizes the risk of batch failures during quality control testing, ensuring that released products consistently meet release specifications. This predictability allows supply chain planners to optimize inventory levels and reduce the safety stock required to mitigate supply disruptions.
- Scalability and Environmental Compliance: The process utilizes mild reaction conditions and avoids the use of hazardous reagents where possible, aligning with modern green chemistry principles. The simplicity of the workup procedure, involving standard filtration and washing steps, makes the process highly scalable from pilot plant to multi-ton commercial production. The reduced generation of organic waste and the ability to recycle solvents like acetone and methanol further enhance the environmental profile of the manufacturing process. This compliance with environmental regulations future-proofs the supply chain against tightening ecological legislation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Ceftriaxone Sodium Tetrahydrate technology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a reliable basis for decision-making. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this material into their existing product portfolios.
Q: What distinguishes Ceftriaxone Sodium Tetrahydrate from the conventional 3.5 hydrate?
A: The tetrahydrate form contains four moles of crystal water per mole of drug, creating a more robust crystal lattice that significantly reduces hygroscopicity and improves powder flowability compared to the traditional 3.5 hydrate.
Q: How does the new crystalline form impact long-term storage stability?
A: Accelerated stability testing demonstrates that the tetrahydrate maintains purity and moisture content over six months at elevated temperatures, whereas comparative 3.5 hydrate samples show increased degradation and moisture uptake.
Q: Is the synthesis process scalable for industrial manufacturing?
A: Yes, the patent describes a method using readily available reactants and mild reaction conditions (10-40°C), avoiding complex cryogenic steps, which facilitates easy scale-up from pilot to commercial production volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ceftriaxone Sodium Supplier
The technical potential of Ceftriaxone Sodium Tetrahydrate is immense, offering a pathway to superior product quality and operational efficiency in antibiotic manufacturing. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this advanced crystalline form to the global market. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, ensuring that every gram of material we produce meets the exacting standards of the pharmaceutical industry. We understand the critical nature of beta-lactam stability and have invested heavily in the infrastructure necessary to handle these sensitive compounds with the utmost care and precision.
We invite you to collaborate with us to leverage this technology for your next-generation cephalosporin formulations. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in crystallization engineering can drive value for your organization.
