Advanced Silylation Protection Strategy for High-Purity Ceftriaxone Sodium Manufacturing
Advanced Silylation Protection Strategy for High-Purity Ceftriaxone Sodium Manufacturing
The pharmaceutical landscape for third-generation cephalosporins continues to evolve, driven by the critical need for higher purity standards and more efficient manufacturing processes. A pivotal advancement in this domain is detailed in patent CN115232151A, which introduces a novel synthetic methodology for Ceftriaxone Sodium. This antibiotic remains a cornerstone in treating severe bacterial infections, ranging from respiratory tract issues to meningitis, necessitating a supply chain that guarantees exceptional quality and consistency. The traditional synthesis pathways often struggle with selectivity issues due to the multifunctional nature of the 7-ACT intermediate, leading to complex impurity profiles. The new approach leverages a sophisticated silylation protection strategy to overcome these inherent chemical challenges, offering a robust alternative for industrial scale-up.
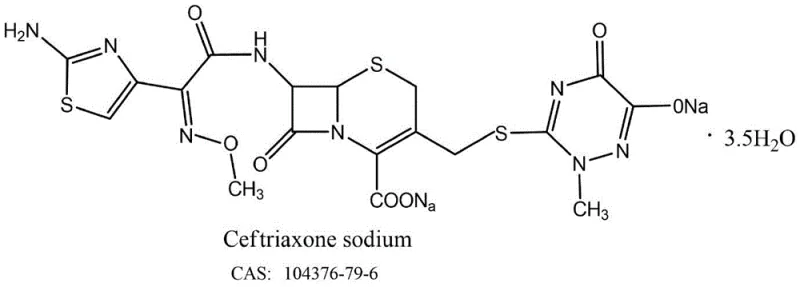
This structural complexity, characterized by the fused beta-lactam and dihydrothiazine rings alongside the methoxyimino side chain, demands precise chemical control. As a reliable ceftriaxone sodium supplier, understanding these molecular intricacies is paramount to delivering a product that meets stringent pharmacopoeial specifications. The patent highlights a shift away from brute-force synthesis towards a more elegant, protective-group-mediated strategy that aligns with modern green chemistry principles while enhancing economic viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of ceftriaxone sodium has relied on the direct condensation of 7-aminocephalosporanic acid derivatives with active esters. However, the 7-ACT intermediate possesses highly reactive amino and carboxyl groups on its nucleus. In conventional processes, if these groups are not adequately masked, they participate in unwanted side reactions during the coupling with the AE-active ester. This lack of selectivity results in poor reaction conversion rates, often hovering around 84% in comparative studies, and generates significant quantities of unknown impurities or polymers. These impurities not only compromise the safety profile of the final drug but also create substantial downstream purification burdens. Furthermore, the traditional route typically requires an intermediate isolation step where ceftriaxone acid is acidified, crystallized, and separated before being converted into the sodium salt. This multi-step isolation increases solvent consumption, energy usage, and overall process time, creating bottlenecks in cost reduction in antibiotic manufacturing.
The Novel Approach
The innovative method described in the patent fundamentally alters this workflow by introducing a pre-protection step using silylating agents. By reacting 7-ACT with reagents such as hexamethyldisilazane (HMDS) or BSA under anhydrous conditions, both the amino and carboxyl functionalities are simultaneously protected. This creates a chemically inert intermediate that allows the subsequent condensation with the AE-active ester to proceed with high regioselectivity. Crucially, this pathway eliminates the need to isolate the free acid intermediate. Instead, the reaction mixture proceeds directly to salting and crystallization after the coupling is complete. This telescoping of steps drastically simplifies the operational flow, reduces the number of unit operations, and minimizes the exposure of the sensitive beta-lactam ring to harsh acidic conditions, thereby preserving product integrity and maximizing yield.
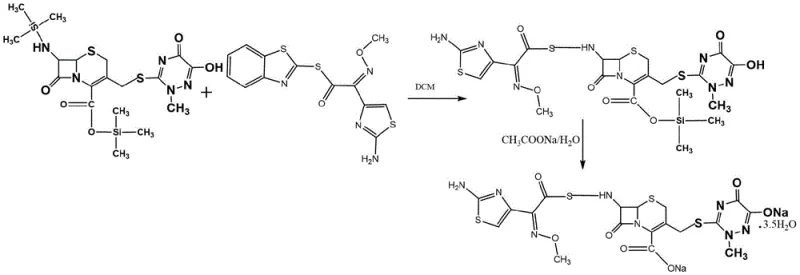
Mechanistic Insights into Silylation-Mediated Protection
The core of this technological breakthrough lies in the mechanistic efficiency of the silylation step. When 7-ACT is treated with a silylating agent in a low-boiling solvent such as dichloromethane or dimethyl carbonate, the silicon atoms form stable bonds with the nitrogen of the amino group and the oxygen of the carboxyl group. This dual protection is achieved under mild reflux conditions, specifically controlled between 38-42°C. Maintaining this narrow temperature window is critical; it provides sufficient thermal energy to drive the silylation to completion over a period of 4 to 12 hours without inducing thermal degradation of the cephalosporin core. The resulting silylated intermediate is significantly less nucleophilic at the protected sites, ensuring that when the AE-active ester is introduced, the acylation occurs exclusively at the desired 7-position amino group once the protection is strategically managed or displaced during the coupling phase.
Following the protection phase, the reaction mixture is cooled to below 10°C before the addition of the AE-active ester. This temperature drop is a vital control parameter that suppresses exothermic runaway and further enhances selectivity. The condensation proceeds for 2 to 20 hours, notably without the need for additional organic base catalysts which are common sources of impurities in other methods. The by-products generated, such as urea derivatives from the silylating agent, are easily removed via filtration or remain soluble in the aqueous workup, preventing them from contaminating the final crystal lattice. This mechanistic precision allows for the control of maximum single impurities to levels below 0.07%, a specification that far exceeds the capabilities of many legacy processes.
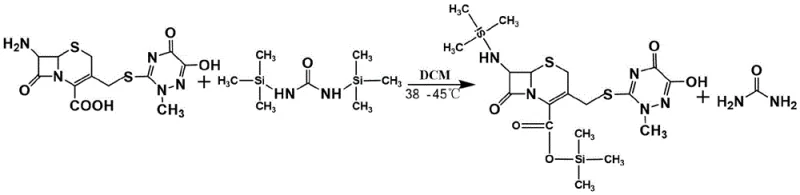
How to Synthesize Ceftriaxone Sodium Efficiently
Implementing this synthesis route requires strict adherence to anhydrous conditions and precise stoichiometric control. The process begins with the preparation of the protected 7-ACT solution, followed by the low-temperature coupling and a direct aqueous workup for salting. The elimination of the acid isolation step is the key differentiator that defines the operational simplicity of this method. For R&D teams looking to adopt this technology, the following guide outlines the critical operational parameters derived from the patent examples to ensure successful replication and scale-up.
- Protect 7-ACT using a silylating agent (e.g., HMDS, BSA) in anhydrous solvent at 38-42°C for 4-12 hours.
- Cool the protected solution below 10°C and react with AE-active ester for 2-20 hours without additional organic base catalysts.
- Quench with water, adjust pH to 6.8-7.8 using sodium acetate for direct salting, and crystallize the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this silylation-based methodology offers compelling strategic advantages beyond mere chemical elegance. The primary value driver is the substantial simplification of the manufacturing workflow. By removing the intermediate acidification and crystallization stage, manufacturers can significantly reduce the consumption of organic solvents and the energy required for drying and handling solid intermediates. This reduction in unit operations translates directly into lower utility costs and a smaller environmental footprint, aligning with global sustainability goals. Furthermore, the higher conversion rates observed in this process mean that less raw material is wasted, optimizing the cost of goods sold (COGS) without compromising on the quality of the input materials.
- Cost Reduction in Manufacturing: The elimination of the intermediate isolation step removes the need for extensive filtration, washing, and drying of the ceftriaxone acid intermediate. This streamlining reduces labor hours and equipment occupancy time, allowing for faster batch turnover. Additionally, the ability to recover by-products like urea through simple filtration further contributes to waste minimization and potential cost recovery, creating a leaner and more economically efficient production model.
- Enhanced Supply Chain Reliability: The robustness of the silylation protection method ensures consistent batch-to-batch quality, reducing the risk of failed batches due to impurity spikes. High-purity pharmaceutical intermediates with conversion rates exceeding 95% provide a more predictable output volume, enabling supply chain planners to maintain tighter inventory controls and meet delivery commitments with greater confidence. The use of common, commercially available silylating agents also mitigates the risk of raw material shortages.
- Scalability and Environmental Compliance: The reaction conditions are mild and do not require extreme temperatures or pressures, making the process inherently safer and easier to scale from pilot plants to commercial production. The reduction in solvent usage and the avoidance of harsh acidification steps simplify wastewater treatment protocols. This ease of compliance with environmental regulations ensures long-term operational continuity and reduces the liability associated with hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the technology's practical implications for industrial application.
Q: How does the silylation protection method improve impurity control compared to conventional synthesis?
A: By simultaneously protecting the reactive amino and carboxyl groups on the 7-ACT nucleus prior to condensation, the method prevents side reactions and polymerization, controlling maximum single impurities below 0.07%.
Q: What are the energy consumption benefits of this new synthetic route?
A: The process eliminates the intermediate acidification and crystallization step required to isolate ceftriaxone acid, allowing for direct salting and crystallization which significantly reduces solvent usage and processing time.
Q: Which silylating agents are preferred for this synthesis?
A: Preferred reagents include hexamethyldisilazane (HMDS), hexamethyldisilazane urea (BSU), and BSA, used in a molar ratio of 0.5 to 2 relative to 7-ACT.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ceftriaxone Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced synthetic routes like the silylation protection method is essential for maintaining competitiveness in the global antibiotic market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are seamlessly translated into industrial reality. We operate with stringent purity specifications and utilize rigorous QC labs to verify that every batch of ceftriaxone sodium meets the highest international standards, including the control of critical impurities identified in modern pharmacopoeias.
We invite pharmaceutical partners to collaborate with us to leverage these technological advancements for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to determine how our optimized manufacturing capabilities can support your long-term strategic goals in the antibiotics sector.
