Advanced Iridium Catalyst Technology for Scalable Chiral Amine Manufacturing
Advanced Iridium Catalyst Technology for Scalable Chiral Amine Manufacturing
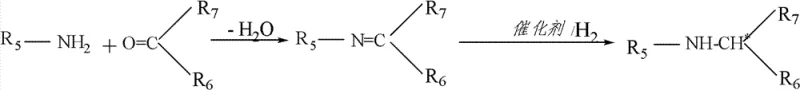
The landscape of chiral amine synthesis is undergoing a significant transformation driven by the need for more efficient and cost-effective catalytic systems, particularly for high-volume agrochemical and pharmaceutical applications. Patent CN102950025A introduces a groundbreaking asymmetric hydrogenation catalyst that addresses the longstanding challenges associated with the reduction of prochiral imines. Unlike traditional systems that struggle with the geometric isomerism (cis/trans mixtures) of open-chain imines, this novel technology utilizes a specialized ferrocene-derived ligand complexed with iridium to achieve superior stereocontrol. The core innovation lies in the ligand's unique architecture, which integrates a quaternary ammonium salt moiety directly into the chiral backbone. This structural modification not only enhances catalytic activity but also simplifies the overall process by reducing the dependency on external additives. For R&D directors and process chemists, this represents a pivotal shift towards more robust and scalable methodologies for producing critical chiral intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric hydrogenation of imines has been plagued by significant technical hurdles that limit its industrial applicability. Prior art, such as the methods disclosed in patent EP0691949B1, relies heavily on the use of external phase transfer agents like tetrabutylammonium iodide to facilitate the reaction. These conventional processes often require harsh operating conditions, including elevated temperatures around 50°C and high hydrogen pressures up to 80 atm, to achieve moderate enantioselectivity. Furthermore, the necessity of adding stoichiometric or near-stoichiometric amounts of iodide salts creates a substantial burden on downstream processing. The removal of these ionic species from the final product stream is energy-intensive and generates considerable chemical waste, thereby inflating the overall cost of goods sold (COGS). Additionally, the enantiomeric excess (e.e.) values obtained through these older methods frequently hover around 76%, which may be insufficient for stringent pharmaceutical or high-end agrochemical specifications without further costly purification steps.
The Novel Approach
The technology described in CN102950025A offers a sophisticated solution by embedding the phase-transfer functionality directly within the chiral ligand structure itself. This bifunctional design allows the catalyst to operate effectively even in the absence of external iodide salts, particularly at lower substrate-to-catalyst (S/C) ratios. The reaction can proceed under milder conditions, often at room temperature, while maintaining high conversion rates and impressive stereoselectivity. By eliminating the need for bulk external additives, the process inherently reduces the complexity of the work-up procedure. The novel catalyst system demonstrates remarkable versatility, capable of handling a wide range of imine substrates derived from various amines and ketones. This approach not only streamlines the synthetic route but also aligns with green chemistry principles by minimizing auxiliary reagent usage. The result is a cleaner reaction profile that facilitates easier isolation of the target chiral amine, making it an ideal candidate for continuous operation and large-scale manufacturing environments.
Mechanistic Insights into Ferrocene-Derived Iridium Catalysis
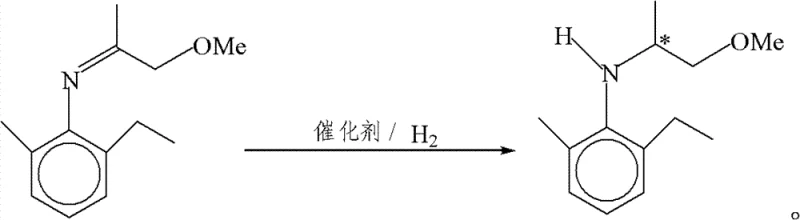
The exceptional performance of this catalyst system can be attributed to the synergistic interaction between the rigid ferrocene backbone and the flexible phosphine arms, which create a well-defined chiral pocket around the iridium center. The presence of the quaternary ammonium group on the ligand plays a dual role: it acts as an intramolecular phase transfer catalyst to enhance the solubility and reactivity of ionic species, and it potentially stabilizes the active catalytic species through electrostatic interactions. This structural feature is critical for overcoming the kinetic barriers associated with the hydrogenation of sterically hindered or electronically diverse imines. The mechanism likely involves the formation of a cationic iridium-hydride species that coordinates with the imine substrate, followed by stereoselective hydride transfer. The rigidity of the ferrocene scaffold ensures that the chiral information is effectively transmitted to the substrate, resulting in high enantiomeric differentiation between the pro-chiral faces of the imine bond. Understanding these mechanistic nuances is vital for process optimization, as it allows chemists to fine-tune reaction parameters such as solvent polarity and acid concentration to maximize both rate and selectivity.
From an impurity control perspective, the integrated design of the ligand significantly mitigates the risk of side reactions often caused by free iodide ions in solution. In conventional systems, excess iodide can lead to catalyst decomposition or the formation of unwanted by-products through nucleophilic attack on the substrate. By tethering the ammonium group to the ligand, the local concentration of the phase-transfer moiety is optimized without flooding the reaction medium with free ions. This leads to a cleaner crude product profile with fewer difficult-to-remove impurities. Furthermore, the cationic nature of the spent catalyst facilitates its separation from the neutral organic product using ion-exchange resins. This capability is a game-changer for quality control, as it ensures that residual metal levels in the final API or intermediate can be kept well below regulatory thresholds. Such precise control over the impurity profile is essential for meeting the rigorous specifications demanded by global regulatory bodies for both pharmaceutical and agrochemical products.
How to Synthesize Chiral Amines Efficiently
The implementation of this advanced catalytic system follows a straightforward protocol that is amenable to standard industrial hydrogenation equipment. The process begins with the in-situ generation of the active catalyst by mixing the ferrocene-derived ligand with a commercially available iridium precursor, such as [Ir(COD)Cl]2, in a suitable protic solvent like acetic acid. This mixture is then introduced into a pressure vessel containing the imine substrate. One of the key advantages of this method is its operational flexibility; depending on the desired throughput and substrate reactivity, the reaction can be run with or without additional iodide salts. For high-volume production where maximizing the substrate-to-catalyst ratio is critical for cost efficiency, a small amount of additive may be beneficial. However, for smaller batches or sensitive substrates, the catalyst performs robustly on its own. The reaction is typically conducted under a hydrogen atmosphere at pressures ranging from 10 to 100 atm and temperatures between ambient and 50°C.
- Prepare the catalyst precursor by mixing the ferrocene-derived chiral ligand containing a quaternary ammonium salt group with dimerized 1,5-cyclooctadiene iridium chloride in an organic acid solvent such as acetic acid.
- Introduce the prochiral imine substrate into the autoclave under nitrogen protection, optionally adding a catalytic amount of iodide salt if high substrate-to-catalyst ratios are targeted.
- Pressurize the reactor with hydrogen gas to 10-100 atm and maintain the temperature between room temperature and 50°C until hydrogen uptake ceases, indicating complete conversion.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this catalyst technology translates into tangible strategic benefits that extend beyond simple yield improvements. The primary advantage lies in the drastic simplification of the supply chain for raw materials. Since the ligand incorporates the necessary phase-transfer functionality, there is no longer a need to source, store, and handle large quantities of separate quaternary ammonium salts like tetrabutylammonium iodide. This consolidation of reagents reduces inventory complexity and minimizes the risk of supply disruptions associated with multi-component systems. Moreover, the ability to operate at lower catalyst loadings while maintaining high turnover numbers means that the consumption of precious metal iridium is optimized. This directly impacts the variable cost of production, offering a pathway to significant cost reduction in agrochemical intermediate manufacturing without compromising on quality or throughput.
- Cost Reduction in Manufacturing: The elimination of external additives and the simplification of the work-up process lead to substantial operational savings. Traditional methods require extensive washing and purification steps to remove ionic salts, which consume large volumes of water and organic solvents. By contrast, the novel catalyst system produces a cleaner reaction mixture that requires less downstream processing. This reduction in solvent usage and waste treatment costs contributes to a leaner manufacturing process. Additionally, the potential for catalyst recovery and reuse via ion-exchange techniques further enhances the economic model by extending the lifecycle of the expensive iridium component. These factors combined create a compelling business case for switching to this technology, driving down the overall cost per kilogram of the final chiral amine product.
- Enhanced Supply Chain Reliability: The robustness of the catalyst under mild conditions ensures consistent production output, which is critical for maintaining reliable delivery schedules to downstream customers. The tolerance of the system to varying substrate qualities and its ability to function effectively at room temperature reduce the risk of batch failures due to thermal runaway or equipment limitations. This reliability is paramount for just-in-time manufacturing models prevalent in the pharmaceutical industry. Furthermore, the ligand synthesis relies on readily available starting materials, ensuring that the supply of the catalyst itself is secure and scalable. This stability in the upstream supply chain mitigates the risk of production bottlenecks, allowing partners to plan their long-term capacity with greater confidence and security.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with demonstrated efficacy at substrate-to-catalyst ratios exceeding 100,000:1 in experimental settings. This high efficiency means that large commercial batches can be produced using relatively small amounts of catalyst, reducing the physical footprint of the reaction and the associated safety hazards of handling large quantities of reactive metals. From an environmental standpoint, the reduction in chemical waste and solvent consumption aligns with increasingly strict global regulations on industrial emissions and effluent discharge. The ability to recover the catalyst also supports circular economy initiatives, enhancing the sustainability profile of the manufacturing site. These attributes make the technology not only commercially viable but also future-proof against tightening environmental compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These insights are derived directly from the experimental data and process descriptions found in the underlying patent literature. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this catalyst into their existing production lines.
Q: Is the addition of external quaternary ammonium salts required for this catalyst system?
A: No, the novel ligand design incorporates a quaternary ammonium salt group directly into the structure, which acts as an internal phase transfer agent. This eliminates the need for large amounts of external additives like tetrabutylammonium iodide, significantly simplifying downstream processing and reducing impurity profiles.
Q: What are the typical reaction conditions for achieving high enantioselectivity?
A: The process operates efficiently under mild conditions, typically using acetic acid as a solvent at temperatures ranging from room temperature to 50°C. Hydrogen pressures between 10 and 100 atm are sufficient to achieve full conversion with enantiomeric excess (e.e.) values exceeding 77%.
Q: Can this catalyst be recovered and reused for industrial scale-up?
A: Yes, due to the cationic nature of the ligand's quaternary ammonium group, the catalyst can be effectively separated from the reaction mixture using medium or polymeric anion resins. This feature supports sustainable manufacturing practices and enhances the economic viability of large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Amine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge catalytic technologies to maintain a competitive edge in the global market. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the ferrocene-derived iridium catalyst are seamlessly translated from the laboratory to the plant. We are committed to delivering high-purity chiral amine intermediates that meet stringent purity specifications, leveraging our rigorous QC labs to verify every batch. Our infrastructure is designed to handle complex asymmetric syntheses with the utmost precision, guaranteeing supply continuity for our partners in the agrochemical and pharmaceutical sectors.
We invite you to collaborate with us to explore how this advanced catalyst technology can optimize your specific manufacturing processes. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your current production volumes and quality requirements. Please contact us to request specific COA data and route feasibility assessments for your target molecules. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain that is not only reliable and compliant but also driven by a relentless pursuit of technological excellence and cost efficiency.
