Advanced Synthetic Route for Prasugrel Intermediates: Enhancing Yield and Purity for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with economic viability, particularly for potent antiplatelet agents like Prasugrel. Patent CN101993447A introduces a transformative methodology for the artificial synthesis of Prasugrel, addressing critical bottlenecks found in earlier generations of manufacturing protocols. This innovation centers on a refined halogenation strategy using dibromohydantoin and a highly efficient coupling reaction driven by the concerted catalysis of phase transfer agents and inorganic salts. By shifting away from hazardous reagents and low-yield steps, this technology offers a compelling value proposition for manufacturers aiming to optimize their supply chains for cardiovascular therapeutics. The detailed reaction sequence demonstrates a clear evolution from laboratory-scale curiosity to a viable industrial process capable of delivering high-purity intermediates consistently. For stakeholders evaluating potential partnerships, understanding the mechanistic underpinnings of this patent is essential for appreciating its commercial potential.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of key Prasugrel intermediates has been plagued by inefficiencies that drive up costs and complicate regulatory approval processes. Prior art, such as that described in US Patent 20030134872, often relies on N-bromosuccinimide (NBS) as a halogenating agent, which is not only expensive but also generates significant succinimide byproducts that are difficult to separate. Furthermore, traditional methods frequently employ toxic chlorinated solvents like carbon tetrachloride or chloroform, posing severe environmental and occupational health risks that modern green chemistry initiatives strive to eliminate. Perhaps most critically, the coupling step to form the core thienopyridine structure in conventional routes suffers from abysmal yields, typically hovering between 32% and 35%, leading to massive material loss. The reliance on column chromatography for final purification further exacerbates these issues, as it is a batch-intensive, solvent-heavy technique that is notoriously difficult to scale for multi-ton production. These cumulative inefficiencies create a fragile supply chain vulnerable to raw material price fluctuations and stringent waste disposal regulations.
The Novel Approach
In stark contrast, the methodology outlined in CN101993447A presents a streamlined, high-efficiency alternative that directly addresses these historical pain points through intelligent reagent selection and catalytic optimization. The process initiates with the use of dibromohydantoin or dichlorohydantoin in acetic acid, a safer and more cost-effective solvent system that avoids the toxicity associated with chlorinated hydrocarbons. The core innovation lies in the coupling reaction, where the introduction of a phase transfer catalyst alongside an inorganic salt creates a synergistic effect that dramatically accelerates the nucleophilic substitution. This concerted catalysis allows the reaction to proceed at moderate temperatures of 40-55°C, achieving yields exceeding 90% in a fraction of the time required by older methods. Additionally, the purification strategy replaces complex chromatographic separation with a straightforward crystallization and recrystallization protocol using common alcohols, significantly simplifying downstream processing. This holistic redesign of the synthetic route ensures that the final product meets stringent purity specifications while maintaining a lean and scalable manufacturing footprint.
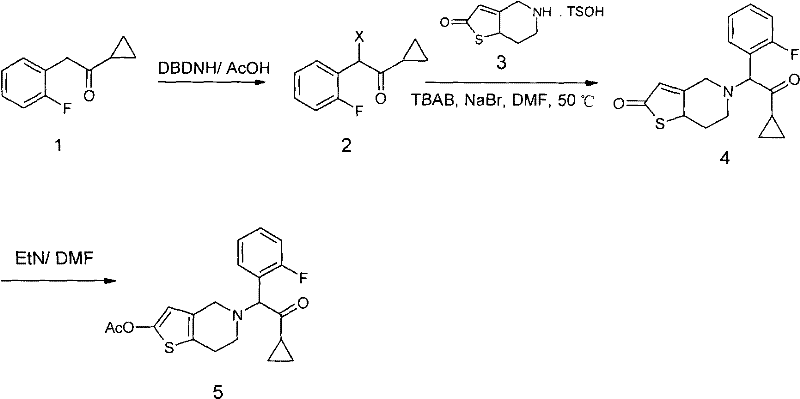
Mechanistic Insights into Concerted Phase Transfer Catalysis
The exceptional performance of this synthetic route can be attributed to the sophisticated interplay between the phase transfer catalyst and the inorganic salt additive during the nucleophilic substitution step. In this mechanism, the quaternary ammonium salt, such as tetrabutylammonium bromide (TBAB), acts as a shuttle, transporting the nucleophilic anion from the aqueous or solid phase into the organic phase where the electrophilic substrate resides. The addition of inorganic salts like sodium bromide plays a dual role; it not only provides a source of nucleophilic bromide ions that may participate in a Finkelstein-type halogen exchange to generate a more reactive intermediate in situ but also exerts a salting-out effect that enhances the reaction kinetics. This concerted action effectively lowers the activation energy of the transition state, allowing the reaction to reach completion rapidly at lower temperatures, thereby minimizing thermal degradation and side reactions. The result is a clean reaction profile with minimal formation of regioisomers or over-alkylated byproducts, which is crucial for maintaining the integrity of the sensitive thienopyridine scaffold.
Impurity control is further reinforced by the strategic choice of solvents and workup procedures designed to maximize product recovery while excluding contaminants. The use of acetic acid in the initial halogenation step ensures high selectivity for the alpha-position, preventing poly-halogenation which could lead to difficult-to-remove impurities later in the sequence. Following the coupling reaction, the workup involves simple aqueous washes and drying, avoiding the complex extraction sequences often required when using miscible solvent systems. The final crystallization from ethanol or isopropanol leverages the differential solubility of the target molecule versus its impurities, effectively purging residual starting materials and catalyst residues without the need for silica gel. This robust impurity management strategy ensures that the final active pharmaceutical ingredient precursor consistently achieves purity levels greater than 99.5%, meeting the rigorous demands of global pharmacopeias.
How to Synthesize Prasugrel Intermediate Efficiently
The synthesis of this critical cardiovascular intermediate follows a logical three-step progression that prioritizes safety, yield, and ease of operation for process chemists. The protocol begins with the controlled halogenation of the ketone precursor, followed by the pivotal catalytic coupling reaction, and concludes with acylation and crystallization. Each stage has been optimized to minimize hazard exposure and maximize throughput, making it an ideal candidate for technology transfer. For a comprehensive breakdown of the specific molar ratios, temperature profiles, and workup instructions, please refer to the standardized guide below.
- Halogenate o-fluorobenzyl cyclopropyl ketone using dibromohydantoin in acetic acid at 60-85°C to form the alpha-halo ketone intermediate.
- Perform nucleophilic substitution between the halo-ketone and thienopyridine tosylate using TBAB and Sodium Bromide in DMF at 40-55°C.
- Acylate the resulting amine with acetic anhydride and triethylamine, followed by crystallization from ethanol to achieve >99.5% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route translates directly into tangible operational efficiencies and risk mitigation strategies. The elimination of column chromatography alone represents a massive reduction in solvent consumption and processing time, which are two of the largest cost drivers in fine chemical manufacturing. By simplifying the purification train to basic crystallization units, facilities can significantly increase their batch turnover rate without requiring additional capital investment in specialized equipment. Furthermore, the switch to less hazardous reagents and solvents reduces the regulatory burden associated with waste disposal and worker safety, ensuring smoother audits and uninterrupted production schedules. These factors combine to create a more resilient supply chain capable of responding quickly to market demand fluctuations for antiplatelet therapies.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic improvement in reaction yield and the removal of expensive purification steps. By boosting the coupling yield from roughly one-third to over ninety percent, the consumption of valuable starting materials is reduced by nearly two-thirds, leading to substantial savings on raw material costs. Additionally, replacing NBS with hydantoin-based halogenating agents and utilizing common alcohols for crystallization instead of large volumes of chromatographic solvents further drives down the cost of goods sold. The overall process intensity is lowered, meaning less energy is required for heating, cooling, and solvent recovery, contributing to a leaner cost structure that enhances competitiveness in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the use of commercially abundant and stable reagents that are less susceptible to supply disruptions compared to specialized or highly regulated chemicals. The robustness of the reaction conditions, which tolerate moderate temperature variations and do not require cryogenic cooling, ensures consistent output even in facilities with varying levels of infrastructure sophistication. Shorter reaction times, specifically the reduction of the coupling step to under an hour, allow for tighter production scheduling and faster turnaround times from order to delivery. This agility is critical for maintaining continuous supply to downstream API manufacturers who operate on just-in-time inventory models.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing unit operations such as stirred tank reactors and filtration systems that are standard in multi-purpose chemical plants. The avoidance of toxic chlorinated solvents and the minimization of heavy metal or complex organic waste streams align perfectly with modern environmental, social, and governance (ESG) goals. This green chemistry profile not only simplifies permitting for new production lines but also future-proofs the manufacturing asset against tightening environmental regulations. Consequently, partners can confidently commit to long-term volume agreements knowing that the production method is sustainable and compliant with global standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation to ensure accuracy and relevance. Understanding these details is vital for technical teams evaluating the feasibility of adopting this route for their own production needs.
Q: How does the new synthesis method improve yield compared to conventional routes?
A: The novel method utilizes a concerted catalysis system involving a phase transfer catalyst and inorganic salts, which increases the coupling reaction yield from approximately 32-35% in prior art to over 90%, drastically reducing material waste.
Q: What safety improvements are offered by replacing NBS and chlorinated solvents?
A: By substituting N-bromosuccinimide (NBS) with dibromohydantoin and replacing toxic solvents like carbon tetrachloride with acetic acid, the process significantly lowers environmental toxicity and operational hazards, facilitating easier regulatory compliance.
Q: Is column chromatography required for purification in this process?
A: No, the process eliminates the need for complex column chromatography by employing a robust crystallization and recrystallization protocol using alcoholic solvents, which is far more economical and scalable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Prasugrel Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and unwavering commitment to quality. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this high-yield route are fully realized in practice. Our state-of-the-art facilities are equipped to handle the specific solvent systems and catalytic conditions required for this synthesis, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest industry standards. We understand that consistency is key in the pharmaceutical supply chain, and our process controls are designed to deliver that reliability batch after batch.
We invite you to collaborate with us to leverage this advanced technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the global market for cardiovascular medicines.
