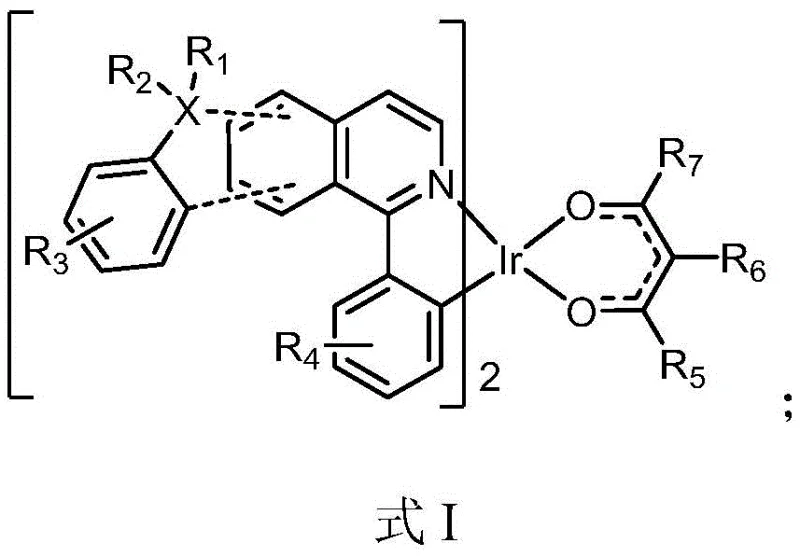
Advanced Iridium Complex Synthesis for High-Efficiency Red Electroluminescent Materials and Commercial OLED Production
The rapid evolution of the organic light-emitting diode (OLED) industry has placed immense pressure on material suppliers to deliver compounds that not only emit pure colors but also operate with maximum energy efficiency and longevity. Patent CN112010908B introduces a groundbreaking class of iridium complexes specifically designed for red light electroluminescent materials, addressing the critical bottlenecks of high synthesis costs and complex purification processes that have historically plagued phosphorescent material production. This technology leverages a unique structural framework where a transition metal iridium is coordinated with specific heterocyclic ligands, creating a robust molecular architecture capable of harvesting both singlet and triplet excitons for theoretically 100% internal quantum efficiency. For R&D directors and procurement specialists alike, this patent represents a pivotal shift towards more manufacturable high-performance emissive dopants that can be integrated into next-generation display panels without compromising on yield or environmental safety standards.
By utilizing a generalized formula where substituents can be independently selected from a wide range of functional groups including deuterium, alkyl, and aryl moieties, the invention offers unparalleled tunability for optimizing the emission spectrum and charge transport properties. The core innovation lies not just in the molecular structure but in the accessible synthetic pathway that avoids the use of exotic catalysts or extreme reaction conditions, making it an ideal candidate for reliable OLED material supplier networks aiming to secure long-term supply chains for electronic chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of phosphorescent iridium complexes for OLED applications has been fraught with significant technical and economic challenges that hinder widespread commercial adoption. Traditional methods often rely on harsh reaction conditions that require precise temperature control and the use of expensive, moisture-sensitive reagents which drastically increase the operational expenditure for fine chemical manufacturers. Furthermore, the purification of these complexes is notoriously difficult due to the formation of closely related impurities and isomers that are hard to separate, leading to lower overall yields and higher waste generation. The prior art frequently suffers from low atom economy and generates substantial amounts of hazardous byproducts, creating a heavy burden on environmental compliance teams and increasing the cost of waste disposal. Additionally, many conventional routes involve multi-step sequences with low convergence, meaning that any failure in an early step results in the loss of valuable iridium metal, which is one of the most expensive precious metals used in catalysis and materials science.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a streamlined two-step process that significantly simplifies the production workflow while maintaining high product integrity. The novel approach employs a mixed solvent system of ethylene glycol ethyl ether and water for the initial cyclometalation step, which facilitates better solubility of the organic ligands while allowing for the efficient precipitation of the intermediate chloro-bridged dimer. This strategic choice of solvent not only reduces the reliance on purely organic volatile solvents but also enables a straightforward isolation procedure via suction filtration, eliminating the need for complex extraction protocols. The subsequent ligand exchange reaction is conducted under mild thermal conditions at 120°C, which is easily achievable in standard industrial reactors without requiring specialized high-pressure equipment. This robustness translates directly into cost reduction in electronic chemical manufacturing by minimizing energy consumption and reducing the risk of batch failures due to thermal runaway or sensitivity issues.
Mechanistic Insights into Cyclometalation and Ligand Exchange
The synthesis mechanism begins with the activation of the C-H bond on the phenyl ring of the main ligand (Compound A) by the iridium center, a process known as cyclometalation, which forms a stable five-membered metallacycle. This step is crucial as it establishes the primary coordination sphere around the metal, locking the ligand in a rigid conformation that prevents non-radiative decay pathways and enhances phosphorescence quantum yield. The use of iridium trichloride trihydrate as the metal source allows for the formation of a chloro-bridged dimer intermediate (Compound B), which serves as a versatile precursor for further functionalization. The presence of chloride bridges in this dimer is labile enough to be displaced by stronger chelating agents but stable enough to allow for isolation and purification before the final step, providing a critical quality control checkpoint in the manufacturing process.
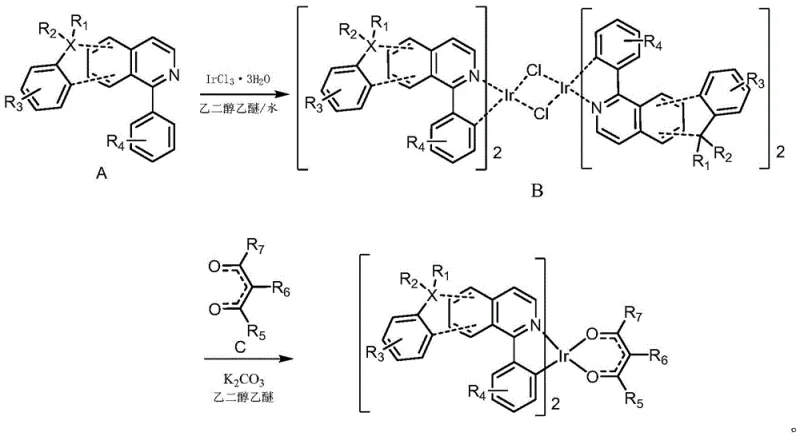
In the second stage, the addition of the auxiliary ligand (Compound C), typically a beta-diketonate derivative, drives the cleavage of the chloro-bridges through a ligand exchange mechanism facilitated by the base potassium carbonate. The base neutralizes the hydrochloric acid generated during the displacement, pushing the equilibrium towards the formation of the neutral, homoleptic iridium complex. This mechanistic pathway ensures that the final product is electrically neutral, which is essential for preventing charge trapping issues within the OLED emissive layer. The structural flexibility allowed by the variable R groups on both the main and auxiliary ligands enables fine-tuning of the HOMO-LUMO gap, allowing manufacturers to precisely target the deep red emission region required for high-color-gamut displays without altering the core synthetic strategy.
How to Synthesize Red Light Electroluminescent Iridium Complex Efficiently
The preparation of these high-performance materials follows a reproducible protocol that balances reaction kinetics with ease of handling, making it suitable for both laboratory scale-up and large-scale commercial production. The process relies on standard unit operations such as reflux, filtration, and column chromatography, which are well-understood by process engineers in the fine chemical industry. By adhering to the specific molar ratios and solvent volumes outlined in the patent examples, manufacturers can consistently achieve high purity levels exceeding 99% as verified by HPLC analysis. The following guide outlines the standardized steps derived from the patent embodiments to ensure optimal yield and quality control.
- React Compound A with Iridium Trichloride Trihydrate in a mixed solvent of ethylene glycol ethyl ether and water under reflux to form the bridging ligand B.
- Mix the bridging ligand B with Compound C and potassium carbonate in ethylene glycol ethyl ether, stirring at 120°C under nitrogen protection to obtain the final iridium complex.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond mere technical performance metrics, directly impacting the bottom line through operational efficiencies. The elimination of exotic catalysts and the use of commodity chemicals like potassium carbonate and ethylene glycol ethyl ether significantly lowers the raw material cost profile, making the final dopant material more price-competitive against legacy technologies. Moreover, the robustness of the reaction conditions reduces the dependency on highly specialized operators and allows for production in facilities with standard chemical processing capabilities, thereby expanding the potential supplier base and mitigating supply chain risks associated with single-source dependencies.
- Cost Reduction in Manufacturing: The streamlined two-step synthesis eliminates the need for expensive transition metal catalysts often required in cross-coupling reactions, resulting in substantial cost savings on reagent procurement. The ability to isolate the intermediate bridging ligand via simple filtration rather than complex distillation or chromatography reduces solvent consumption and energy usage, further driving down the cost of goods sold. Additionally, the high yield of the target product minimizes the loss of precious iridium metal, which is a major cost driver in organometallic synthesis, ensuring that every gram of raw material is converted into saleable product with maximum efficiency.
- Enhanced Supply Chain Reliability: The starting materials, including various substituted phenylquinoline derivatives and beta-diketonates, are structurally simple and can be sourced from multiple global vendors, preventing bottlenecks caused by raw material shortages. The synthetic route's tolerance to minor variations in reaction parameters means that batch-to-batch consistency is easier to maintain, reducing the rate of out-of-specification material that often disrupts downstream device fabrication lines. This reliability is critical for display manufacturers who require uninterrupted supply of emissive materials to meet tight production schedules for consumer electronics.
- Scalability and Environmental Compliance: The process operates at atmospheric pressure and moderate temperatures, making it inherently safer and easier to scale from kilogram to tonne quantities without requiring significant capital investment in high-pressure reactors. The use of water as a co-solvent in the first step aligns with green chemistry principles by reducing the volume of organic waste generated, simplifying wastewater treatment processes and lowering environmental compliance costs. The solid-state nature of the intermediates and final products facilitates safe storage and transportation, reducing the logistical complexities associated with handling hazardous liquid reagents.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and performance of this iridium complex technology, based on the detailed experimental data provided in the patent documentation. Understanding these aspects is vital for R&D teams evaluating the material for integration into their device stacks and for quality assurance personnel establishing incoming inspection criteria. The answers reflect the specific findings from the patent examples, ensuring accuracy and relevance to real-world application scenarios.
Q: What is the purity level of the synthesized iridium complex?
A: According to the patent data, the synthesized iridium complexes achieve an HPLC purity of greater than 99%, ensuring high performance in electroluminescent devices.
Q: How does this new iridium complex improve OLED device performance?
A: The complex significantly reduces the driving voltage of the photoelectric device while remarkably improving current efficiency and prolonging the service life compared to conventional materials.
Q: What are the reaction conditions for the final ligand exchange step?
A: The final step involves stirring the bridging ligand and Compound C with potassium carbonate in ethylene glycol ethyl ether for 24 hours at 120°C under a nitrogen atmosphere.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Iridium Complex Supplier
As the demand for high-efficiency red emitters continues to surge in the OLED market, partnering with a manufacturer that possesses deep expertise in organometallic synthesis is essential for securing a competitive advantage. NINGBO INNO PHARMCHEM stands at the forefront of this technology, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver materials that meet the most stringent purity specifications. Our state-of-the-art rigorous QC labs ensure that every batch of iridium complex undergoes comprehensive analysis, including HPLC, mass spectrometry, and elemental analysis, to guarantee performance consistency that matches the high standards set by the patent embodiments.
We invite you to collaborate with our technical team to explore how this novel iridium complex can enhance your display products while optimizing your manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced synthesis capabilities can support your journey towards next-generation electroluminescent devices.
