Revolutionizing Nucleoside Production: A Deep Dive into Mild Gold-Catalyzed Glycosylation for Commercial Scale-Up
Introduction to Next-Generation Nucleoside Manufacturing
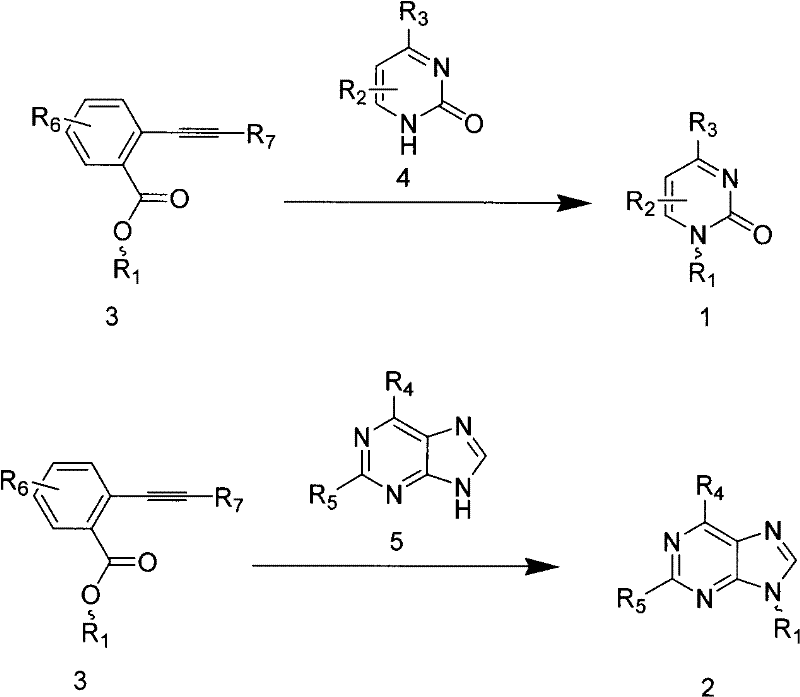
The pharmaceutical industry's relentless pursuit of potent antiviral and antitumor agents has placed nucleoside analogs at the forefront of medicinal chemistry. However, the chemical synthesis of these critical building blocks has long been hindered by harsh reaction conditions and poor selectivity. Patent CN102127135A introduces a transformative preparation method for pyrimidine and purine nucleoside compounds that addresses these historical bottlenecks. By leveraging a novel glycosylation strategy involving ortho-alkynylbenzoate sugar donors and alkynophilic Lewis acid catalysis, this technology enables the efficient construction of the glycosidic bond under remarkably mild conditions. For R&D directors and process chemists, this represents a paradigm shift from the traditional Vorbrüggen methodology, offering a pathway to higher purity intermediates with reduced environmental impact. The ability to synthesize complex nucleosides at room temperature without the need for stoichiometric strong Lewis acids is a significant technical breakthrough that promises to streamline the supply chain for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nucleosides has relied heavily on the Vorbrüggen method, which utilizes peracetylated sugars and strong Lewis acid promoters such as tin tetrachloride (SnCl4) or trimethylsilyl trifluoromethanesulfonate (TMSOTf). While effective for simple substrates, this conventional approach suffers from severe limitations when applied to complex or sensitive molecules. The requirement for stoichiometric amounts of corrosive Lewis acids necessitates rigorous quenching and purification steps, often leading to product degradation and lower overall yields. Furthermore, the reaction conditions typically involve refluxing in chlorinated solvents, which poses significant safety and environmental hazards on a commercial scale. Another critical drawback is the lack of regioselectivity, particularly in purine synthesis, where mixtures of N7 and N9 isomers are frequently obtained, complicating downstream processing and increasing manufacturing costs due to the need for extensive chromatographic separation.
The Novel Approach
In stark contrast, the methodology disclosed in CN102127135A utilizes ortho-alkynylbenzoate protected sugars as glycosyl donors, activated by catalytic amounts of alkynophilic gold complexes. This innovative approach allows the glycosylation reaction to proceed efficiently at room temperature or slightly elevated temperatures (0-80°C), drastically reducing thermal stress on the substrates. The use of a catalytic system rather than stoichiometric promoters not only minimizes waste generation but also simplifies the work-up procedure, as there is no need to neutralize large quantities of strong acids. Crucially, this method demonstrates exceptional regioselectivity, particularly for purine nucleosides, favoring the formation of the biologically relevant N9-glycoside while suppressing the N7-isomer. This precision in bond formation translates directly to higher crude purity, reducing the burden on purification teams and enhancing the overall economic viability of the process for reliable nucleoside intermediate suppliers.
Mechanistic Insights into Gold-Catalyzed Alkynophilic Activation
The core innovation of this technology lies in the unique activation mechanism of the glycosyl donor. Unlike traditional oxocarbenium ion pathways driven by hard Lewis acids, this reaction proceeds through the activation of the alkyne moiety on the benzoate leaving group by a soft, alkynophilic gold catalyst. The gold center coordinates with the triple bond of the ortho-alkynylbenzoate, increasing the electrophilicity of the carbonyl carbon and facilitating the departure of the leaving group to generate the reactive oxocarbenium ion species in situ. This soft activation mode is compatible with a wide range of functional groups that might otherwise be sensitive to harsh acidic conditions. The silylated nucleobase, prepared in situ using reagents such as BSTFA, HMDS, or BSA, then attacks the anomeric center to form the glycosidic bond. This mechanism ensures a smooth reaction profile with minimal side reactions, providing a robust platform for the synthesis of diverse nucleoside analogs.
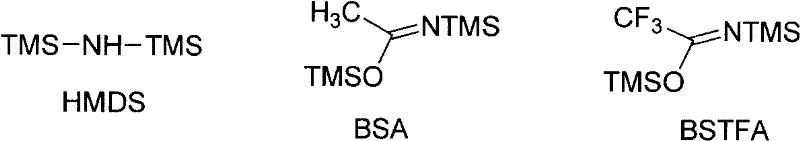
Furthermore, the choice of catalyst and additives plays a pivotal role in controlling the stereochemical outcome and reaction rate. The patent specifies the use of gold complexes such as AuCl, AuCl3, or phosphine-ligated gold salts like Ph3PAuNTf2, often in conjunction with molecular sieves to maintain anhydrous conditions. For purine synthesis, the inclusion of drying agents like 3Å or 4Å molecular sieves is particularly beneficial in driving the equilibrium towards product formation and preventing hydrolysis of the sensitive intermediates. The mild nature of this catalytic cycle allows for the preservation of delicate protecting groups on the sugar moiety, such as benzoyl or acetyl groups, ensuring that the final product retains the necessary functionality for subsequent deprotection steps. This level of control over the reaction environment is essential for producing high-purity OLED materials or pharmaceutical intermediates where trace impurities can be detrimental.
How to Synthesize Protected Nucleoside Compounds Efficiently
The practical implementation of this synthesis route involves a two-stage process that is amenable to standard laboratory and pilot plant equipment. First, the glycosyl donor is prepared by esterifying a fully protected sugar with ortho-alkynylbenzoic acid using standard coupling agents like DCC or EDC. This step is high-yielding and produces a stable donor that can be stored or used immediately. The second stage involves the coupling of this donor with the heterocyclic base. The detailed standardized synthesis steps, including specific molar ratios, solvent choices, and purification protocols, are outlined in the guide below. This streamlined workflow eliminates the need for specialized high-pressure equipment or cryogenic conditions, making it accessible for contract development and manufacturing organizations looking to expand their nucleoside capabilities.
- Prepare the glycosyl donor by esterifying fully protected sugar hydroxyls with ortho-alkynylbenzoic acid using DCC/DMAP or EDC coupling agents.
- Activate the nucleobase (pyrimidine or purine) using silylating agents like BSTFA or HMDS in dry acetonitrile or dichloromethane.
- Add a catalytic amount of alkynophilic Lewis acid (e.g., Ph3PAuNTf2) at room temperature to initiate glycosylation, followed by purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this gold-catalyzed technology offers tangible benefits that extend beyond mere chemical elegance. The shift from stoichiometric harsh reagents to catalytic mild systems fundamentally alters the cost structure and risk profile of nucleoside manufacturing. By operating at room temperature, the process significantly reduces energy consumption associated with heating and cooling, contributing to lower utility costs and a smaller carbon footprint. Moreover, the high regioselectivity observed in purine synthesis eliminates the costly and time-consuming separation of N7/N9 isomers, which is a common bottleneck in traditional processes. This efficiency gain translates directly into shorter production cycles and improved throughput, allowing suppliers to respond more rapidly to market demands for antiviral and anticancer ingredients.
- Cost Reduction in Manufacturing: The elimination of stoichiometric strong Lewis acids like SnCl4 removes the need for extensive aqueous work-ups and neutralization steps, which are major sources of chemical waste and effluent treatment costs. Additionally, the catalytic nature of the gold promoter means that expensive metal usage is minimized, and the high yields reported (often exceeding 85-90%) reduce the cost of goods sold by maximizing raw material utilization. The simplified purification process further lowers operational expenses by reducing solvent consumption and column chromatography time.
- Enhanced Supply Chain Reliability: The mild reaction conditions enhance the robustness of the supply chain by reducing the risk of batch failures due to thermal runaway or reagent instability. The compatibility of this method with a wide variety of protecting groups and sugar scaffolds ensures that manufacturers can flexibly produce different nucleoside analogs using a unified platform. This versatility mitigates supply risks associated with specialized reagents and allows for quicker switching between products to meet fluctuating customer requirements for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling up exothermic reactions involving strong Lewis acids presents significant engineering challenges, but this gold-catalyzed method operates safely at ambient temperatures, facilitating easier scale-up from kilogram to tonne scales. The reduction in hazardous waste generation aligns with increasingly stringent environmental regulations, ensuring long-term compliance and sustainability. This green chemistry approach not only protects the environment but also enhances the corporate reputation of manufacturers as responsible partners in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel nucleoside synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this process into their existing manufacturing workflows.
Q: How does this gold-catalyzed method improve regioselectivity for purine nucleosides?
A: Unlike traditional methods that often yield mixtures of N7 and N9 isomers, this protocol utilizes specific alkynophilic activation that selectively targets the N9 position of the purine ring, eliminating the need for difficult chromatographic separation of isomers.
Q: What are the advantages of using ortho-alkynylbenzoate donors over traditional acetate donors?
A: Ortho-alkynylbenzoate donors allow for catalytic activation using mild gold complexes at room temperature, whereas traditional acetate donors typically require stoichiometric amounts of harsh Lewis acids like SnCl4 and elevated temperatures, which can degrade sensitive substrates.
Q: Is this method scalable for industrial production of antiviral intermediates?
A: Yes, the reaction operates under mild conditions (room temperature to 30°C) and uses catalytic amounts of promoter, which significantly simplifies work-up procedures and reduces waste generation, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nucleoside Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of advanced synthetic methodologies in delivering high-quality pharmaceutical ingredients. Our team of expert chemists has extensively evaluated the gold-catalyzed glycosylation technique described in CN102127135A and possesses the technical expertise to implement this route effectively. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of nucleoside intermediate meets the exacting standards required by global regulatory bodies.
We invite potential partners to engage with our technical procurement team to discuss how this innovative technology can optimize your supply chain. By leveraging our capabilities, you can access a Customized Cost-Saving Analysis tailored to your specific product portfolio. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing us to demonstrate how our commitment to cutting-edge chemistry can drive value and efficiency in your nucleoside sourcing strategy.
