Advanced Phthalide Synthesis for High-Purity Agrochemical and Pharmaceutical Intermediates
Advanced Phthalide Synthesis for High-Purity Agrochemical and Pharmaceutical Intermediates
The global demand for high-performance agrochemicals, particularly methoxyacrylate fungicides like kresoxim-methyl and trifloxystrobin, relies heavily on the consistent supply of key intermediates such as phthalide (o-hydroxymethylbenzoic acid lactone). Patent CN101638406A introduces a groundbreaking preparation method that fundamentally shifts the production paradigm from hazardous high-pressure hydrogenation to a safer, more controllable free-radical chlorination and thermal cyclization process. This technological leap addresses critical pain points in the fine chemical industry, offering a robust pathway for manufacturing high-purity phthalide without the need for specialized high-pressure autoclaves. By leveraging standard reactor equipment and optimizing reaction kinetics through precise initiator dosing, this method ensures superior product quality while drastically simplifying the operational complexity typically associated with lactone synthesis.
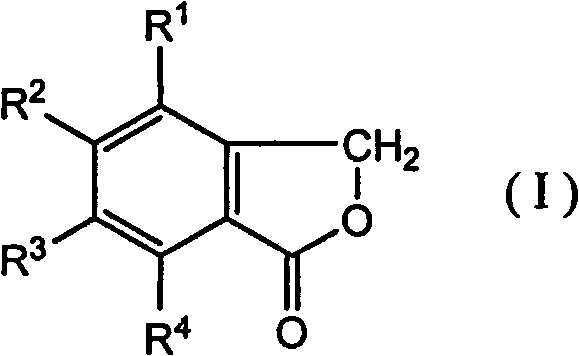
For procurement managers and supply chain directors, the implications of this patent are profound. The ability to produce phthalide using conventional infrastructure translates directly into reduced capital expenditure and enhanced supply chain resilience. Unlike traditional methods that depend on scarce noble metal catalysts and dangerous hydrogen gas handling, this novel approach utilizes readily available chlorine gas and organic initiators like AIBN. This shift not only mitigates safety risks but also stabilizes production costs by removing dependency on volatile catalyst markets. Furthermore, the process demonstrates exceptional versatility, successfully applied to both unsubstituted phthalide and halogenated derivatives like 5-bromophthalide, making it an ideal solution for a reliable agrochemical intermediate supplier aiming to diversify their portfolio with minimal retooling.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of phthalide has been dominated by catalytic hydrogenation of phthalic anhydride, a process fraught with significant engineering and economic challenges. Traditional protocols, such as those described in EP-B-89417 and US-A-2114696, necessitate the use of high-pressure autoclaves operating at hydrogen pressures exceeding 165 bar and temperatures around 150-180°C. These extreme conditions impose severe constraints on equipment selection, requiring expensive alloy-steel reactors and rigorous safety systems to prevent catastrophic failure. Moreover, the reliance on Raney nickel or supported nickel catalysts introduces substantial variability; catalyst deactivation, difficult recovery, and the generation of heavy metal waste streams complicate downstream processing and environmental compliance. The high energy consumption required to maintain high-pressure hydrogenation further erodes profit margins, making cost reduction in phthalide manufacturing increasingly difficult under conventional regimes.
The Novel Approach
In stark contrast, the method disclosed in CN101638406A circumvents these hurdles by employing a two-stage sequence involving free-radical chlorination followed by thermal cyclization under vacuum. This approach operates at near-atmospheric pressure during the chlorination phase (80-90°C) and utilizes standard vacuum distillation equipment for the cyclization step (160-170°C at -0.1 MPa). By eliminating the need for high-pressure hydrogen, the process dramatically lowers the barrier to entry for production facilities, allowing for the use of standard glass-lined or stainless steel reactors. The strategic control of chlorination conversion—stopping specifically when the monochloride intermediate reaches 40-43%—prevents the formation of undesirable dichloride byproducts, ensuring a cleaner reaction profile. This innovation represents a paradigm shift towards safer, more scalable chemistry that aligns perfectly with modern green manufacturing principles.
Mechanistic Insights into Free Radical Chlorination and Thermal Cyclization
The core of this synthesis lies in the precise manipulation of free radical kinetics during the initial chlorination of methyl o-toluate. As illustrated in the reaction scheme below, the process initiates with the homolytic cleavage of the initiator (AIBN or benzoyl peroxide) to generate radicals that abstract a hydrogen atom from the benzylic methyl group of the starting ester. This generates a benzylic radical which subsequently reacts with molecular chlorine to form the crucial intermediate, methyl o-chloromethylbenzoate (Formula III). The patent emphasizes the critical importance of monitoring this step via gas chromatography; maintaining the conversion of the monochloride between 40% and 43% is essential to maximize yield while minimizing the formation of the dichloride impurity, which is strictly controlled to be ≤1.5%. This kinetic control ensures that the subsequent cyclization step proceeds with high efficiency.
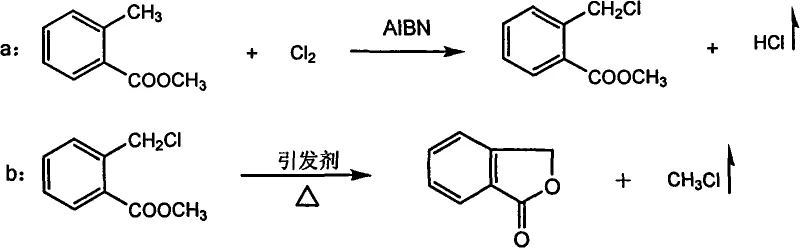
Following chlorination, the reaction mixture undergoes a thermal cyclization driven by the elimination of methyl chloride gas. This intramolecular nucleophilic substitution is facilitated by heating the mixture to 160-170°C under a high vacuum (-0.098 MPa). Interestingly, the patent reveals that adding a small amount of phthalide itself acts as an effective initiator (Initiator 2) for this cyclization, likely stabilizing the transition state or facilitating proton transfer. The continuous removal of methyl chloride gas shifts the equilibrium towards the product side according to Le Chatelier's principle, driving the reaction to completion. Unreacted methyl o-toluate is then recovered via fractional distillation and recycled, enhancing atom economy. This mechanistic elegance allows for the production of high-purity phthalide with yields reaching up to 93%, demonstrating superior selectivity compared to reduction methods.
How to Synthesize Phthalide Efficiently
Implementing this synthesis route requires careful attention to temperature gradients and vacuum levels to ensure optimal conversion and safety. The process is designed to be compatible with standard batch reactors equipped with fractionation columns, making it accessible for both pilot-scale development and full-scale commercial production. Operators must strictly adhere to the specified temperature ranges during chlorination to prevent runaway exothermic reactions and utilize real-time GC analysis to determine the exact endpoint for chlorine introduction. The following guide outlines the standardized operational parameters derived from the patent examples, providing a clear roadmap for technical teams to replicate these results.
- React methyl o-toluate with chlorine gas in the presence of AIBN initiator at 80-90°C until monochloride content reaches 40-43%.
- Heat the reaction mixture under negative pressure (-0.1 MPa) with a phthalide initiator to induce cyclization and eliminate methyl chloride.
- Distill off unreacted starting material under vacuum, then recrystallize the crude residue from methanol or ethanol to obtain pure phthalide.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this technology offers compelling advantages that directly address the priorities of procurement and supply chain leadership. By transitioning away from high-pressure hydrogenation, manufacturers can achieve significant cost savings through the elimination of expensive high-pressure vessels and the associated maintenance costs. The removal of noble metal catalysts like Raney nickel not only reduces raw material expenses but also simplifies waste disposal protocols, leading to lower environmental compliance costs. Furthermore, the ability to recycle unreacted starting materials back into the process enhances overall material efficiency, reducing the effective cost per kilogram of the final API intermediate. These factors combine to create a more resilient and cost-effective supply chain capable of withstanding market fluctuations.
- Cost Reduction in Manufacturing: The elimination of high-pressure autoclaves and expensive hydrogenation catalysts results in a substantially lower capital expenditure (CAPEX) and operational expenditure (OPEX). The process utilizes commodity chemicals like chlorine and AIBN, which are widely available and cost-stable, unlike specialized catalysts. Additionally, the recycling of unreacted methyl o-toluate minimizes raw material waste, driving down the variable cost of production significantly without compromising yield.
- Enhanced Supply Chain Reliability: Operating under standard pressure and vacuum conditions reduces the risk of unplanned shutdowns due to equipment failure or safety incidents common in high-pressure hydrogenation. The use of robust, standard reactor types ensures that production can be easily scaled or transferred between facilities without extensive requalification. This flexibility guarantees a continuous supply of critical intermediates, reducing lead times for customers relying on just-in-time delivery models for their agrochemical formulations.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, primarily consisting of recyclable solvents and manageable salt byproducts, avoiding the heavy metal contamination issues associated with nickel catalysts. The mild reaction conditions and absence of high-pressure hydrogen gas simplify safety protocols, making it easier to scale from pilot batches to multi-ton commercial production. This alignment with green chemistry principles facilitates smoother regulatory approvals and enhances the sustainability profile of the final product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this phthalide synthesis technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing clarity on yield expectations, purity standards, and equipment requirements. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific manufacturing needs.
Q: What are the primary advantages of this phthalide synthesis method over traditional hydrogenation?
A: This method eliminates the need for high-pressure autoclaves and expensive Raney nickel catalysts required in traditional hydrogenation. It operates under atmospheric pressure for chlorination and standard vacuum for cyclization, significantly reducing equipment investment and safety risks associated with high-pressure hydrogen gas.
Q: How is the purity of the final phthalide product controlled?
A: Purity is controlled through precise gas chromatography monitoring during chlorination to limit dichloride byproducts to less than 1.5%. Final purification is achieved via vacuum distillation to remove unreacted esters followed by recrystallization, consistently achieving purity levels of ≥99%.
Q: Can this process be adapted for substituted phthalides like 5-bromophthalide?
A: Yes, the process is highly versatile. The patent demonstrates successful application for synthesizing 5-bromophthalide from methyl 4-bromo-2-methylbenzoate using identical reaction conditions, proving its scalability for various halogenated derivatives used in specialty agrochemicals.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phthalide Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the global agrochemical and pharmaceutical sectors. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex chemistries like the phthalide cyclization described above are executed with precision and consistency. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of phthalide or 5-bromophthalide supports your downstream synthesis without interruption. Our facility is equipped to handle the specific vacuum distillation and recrystallization steps required by this advanced protocol, ensuring superior product quality.
We invite you to collaborate with us to optimize your supply chain for methoxyacrylate fungicide production. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can enhance your project's success and reduce your overall time-to-market.
