Advanced Hofmann Degradation Route for High-Purity 3-Aminopyridine Manufacturing
Advanced Hofmann Degradation Route for High-Purity 3-Aminopyridine Manufacturing
The pharmaceutical and agrochemical industries continuously demand more efficient, cost-effective, and environmentally benign synthetic routes for critical heterocyclic intermediates. Patent CN111170937A introduces a significant technological advancement in the preparation of 3-aminopyridine, a vital building block for various active pharmaceutical ingredients (APIs) and crop protection agents. This innovation leverages a refined Hofmann degradation reaction, utilizing nicotinamide as the starting material in the presence of sodium hypochlorite and sodium hydroxide. Unlike traditional methods that often suffer from harsh conditions or complex purification requirements, this novel approach offers a streamlined pathway characterized by mild reaction parameters and exceptional control over product quality. For R&D directors and process chemists, this represents a robust alternative that minimizes operational risks while maximizing output efficiency. The method ensures that the final 3-aminopyridine product achieves a purity level surpassing 99%, coupled with a total yield that can exceed 90%, making it an exceptionally attractive candidate for reliable pharmaceutical intermediate supplier networks seeking to optimize their manufacturing portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-aminopyridine has relied on several distinct pathways, each carrying significant drawbacks that hinder large-scale commercial viability. One common route involves the catalytic synthesis from halogenated pyridine compounds; however, the initial raw materials for this process are notoriously difficult to synthesize and command high market prices, thereby inflating the overall cost of goods sold. Additionally, these reactions frequently necessitate the use of precious metal catalysts, which not only adds to the expense but also introduces complex wastewater treatment challenges due to heavy metal residues. Another prevalent method involves the catalytic reduction of nitropyridine derivatives, yet this approach is plagued by harsh synthesis conditions and lower yields, rendering it economically unfeasible for high-volume production. Furthermore, routes involving cyanopyridine hydrolysis followed by degradation often result in suboptimal total yields, while methods starting from picoline require excessive synthetic steps that accumulate impurities and reduce overall process efficiency. These legacy technologies create substantial bottlenecks for procurement managers aiming to secure cost reduction in API manufacturing without compromising on supply chain stability or environmental compliance standards.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the methodology disclosed in CN111170937A utilizes nicotinamide, a readily available and cost-effective precursor, to drive a highly efficient Hofmann degradation. This innovative strategy circumvents the need for expensive metal catalysts and avoids the generation of hazardous heavy metal waste streams, aligning perfectly with modern green chemistry principles. The reaction conditions are remarkably mild, proceeding initially at low temperatures between 0°C and 20°C before a controlled heating phase, which significantly reduces energy consumption and safety risks associated with exothermic runaway reactions. By optimizing the mass ratio of nicotinamide to sodium hypochlorite and sodium hydroxide, the process achieves a conversion rate where the product content reaches greater than or equal to 98% prior to purification. This high level of conversion minimizes the burden on downstream purification units, allowing for a simpler post-treatment workflow involving rotary evaporation and recrystallization. For supply chain heads, this translates to a more predictable production schedule and reduced lead time for high-purity pharmaceutical intermediates, ensuring consistent availability for downstream drug synthesis applications.
Mechanistic Insights into Nicotinamide Hofmann Degradation
The core of this technological breakthrough lies in the precise execution of the Hofmann degradation mechanism, where the primary amide group of nicotinamide is converted into a primary amine with one fewer carbon atom. In this specific implementation, sodium hypochlorite acts as the oxidizing agent to generate the N-chloroamide intermediate in situ under strictly controlled low-temperature conditions. The subsequent addition of concentrated sodium hydroxide facilitates the rearrangement of this intermediate, leading to the formation of an isocyanate species which rapidly hydrolyzes to yield the desired 3-aminopyridine. The patent emphasizes the critical importance of temperature control during the initial addition phase, maintaining the reaction mixture between 0°C and 20°C to prevent the decomposition of the unstable N-chloroamide and to suppress side reactions that could generate chlorinated byproducts. This meticulous thermal management ensures that the reaction pathway remains selective, directing the majority of the starting material towards the desired amine product rather than degradation fragments. Such mechanistic control is essential for R&D teams focused on impurity profiling, as it inherently limits the formation of difficult-to-remove structural analogues that often plague pyridine chemistry.
Furthermore, the process incorporates rigorous analytical monitoring via High-Performance Liquid Chromatography (HPLC) to track the conversion of raw materials and the formation of the product in real-time. The protocol dictates sampling at regular intervals during the heating phase (50°C to 80°C) to confirm when the product content stabilizes at or above 98%, signaling the optimal endpoint for the reaction. This data-driven approach prevents over-reaction, which could lead to the oxidation of the sensitive amino group or the pyridine ring itself. Following the reaction, the workup procedure involves rotary evaporation to remove volatiles, followed by extraction with an organic solvent to isolate the crude product. A final recrystallization step is employed to refine the crude material into a light yellow or white solid, effectively removing residual salts and minor organic impurities. This multi-stage purification strategy guarantees that the final 3-aminopyridine meets stringent purity specifications of over 99%, satisfying the rigorous quality standards required for commercial scale-up of complex pharmaceutical intermediates.
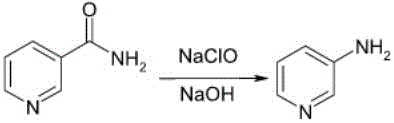
How to Synthesize 3-Aminopyridine Efficiently
The synthesis protocol outlined in the patent provides a clear, reproducible framework for manufacturing 3-aminopyridine with high efficiency and minimal environmental impact. The process begins with the preparation of the oxidant solution, where a specific quantity of sodium hypochlorite is cooled to below 20°C to establish the necessary thermal baseline for the exothermic chlorination step. Solid nicotinamide is then introduced gradually to maintain the temperature within the 0-20°C window, ensuring complete dissolution and conversion to the intermediate without thermal degradation. Once the low-temperature reaction is complete, the system is shifted to a strongly alkaline environment through the addition of 10% sodium hydroxide, triggering the rearrangement and hydrolysis phases upon heating to 50-80°C. The detailed standardized synthesis steps below outline the precise operational parameters required to replicate these results in a pilot or production setting.
- Cool sodium hypochlorite solution below 20°C and add solid nicotinamide while maintaining temperature between 0-20°C.
- Adjust pH to strongly alkaline using 10% sodium hydroxide solution and heat the mixture to 50-80°C for reaction completion.
- Perform rotary evaporation, extraction, recrystallization, and vacuum drying to isolate the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of this nicotinamide-based route offers profound strategic advantages over traditional synthesis methods. The primary benefit stems from the elimination of expensive and supply-constrained raw materials such as halogenated pyridines or nitro-compounds, which are subject to volatile market pricing and regulatory scrutiny. By shifting to nicotinamide, a commodity chemical with a stable global supply chain, manufacturers can significantly stabilize their input costs and mitigate the risk of raw material shortages. Moreover, the absence of transition metal catalysts removes the need for costly scavenging resins or complex filtration systems designed to meet strict residual metal limits in pharmaceutical products. This simplification of the downstream processing train not only reduces capital expenditure on equipment but also lowers the operational overhead associated with waste disposal and catalyst recovery. Consequently, this leads to substantial cost savings in fine chemical manufacturing, allowing companies to improve their margins while offering more competitive pricing to their own customers in the API sector.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the use of inexpensive, bulk-available reagents like sodium hypochlorite and sodium hydroxide, which drastically lowers the variable cost per kilogram of product. Unlike catalytic hydrogenation routes that require high-pressure reactors and precious metals, this ambient pressure oxidation method utilizes standard glass-lined or stainless steel reactors, reducing both CAPEX and maintenance costs. The high yield exceeding 90% means that less raw material is wasted, directly improving the atom economy and reducing the cost of goods sold. Additionally, the simplified workup procedure reduces solvent consumption and energy usage during the drying and purification stages, further contributing to overall operational efficiency and profitability.
- Enhanced Supply Chain Reliability: Reliance on nicotinamide as a feedstock enhances supply chain resilience because it is produced on a massive scale for the vitamin and feed industries, ensuring consistent availability year-round. The reagents required for the reaction, specifically sodium hypochlorite and caustic soda, are foundational chemicals produced locally in most industrial regions, minimizing logistics risks and transportation lead times. This localization of the supply base reduces dependency on single-source suppliers for exotic catalysts or specialized intermediates, thereby insulating the production schedule from global geopolitical disruptions. For supply chain heads, this reliability translates into the ability to commit to longer-term delivery contracts with API manufacturers, fostering stronger customer relationships and market share growth.
- Scalability and Environmental Compliance: The mild reaction conditions and aqueous nature of the initial reaction medium make this process inherently safer and easier to scale from laboratory benchtop to multi-ton production vessels. The absence of hazardous reagents like bromine or strong reducing agents simplifies the permitting process and reduces the facility's environmental footprint, aligning with increasingly strict global ESG (Environmental, Social, and Governance) mandates. Wastewater treatment is streamlined since the primary byproducts are inorganic salts that are easier to manage than heavy metal sludge or complex organic waste streams. This environmental compatibility ensures long-term operational continuity without the risk of shutdowns due to regulatory non-compliance, securing the asset's value for the long term.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Hofmann degradation technology for 3-aminopyridine production. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances is critical for technical teams evaluating the feasibility of technology transfer or process optimization projects. The answers reflect the balance between chemical performance and operational practicality that defines modern fine chemical synthesis.
Q: What are the primary advantages of using nicotinamide over halogenated pyridine for 3-aminopyridine synthesis?
A: Using nicotinamide avoids the high cost and difficult synthesis associated with halogenated pyridine raw materials. Furthermore, this route eliminates the need for expensive metal catalysts and simplifies wastewater treatment compared to traditional catalytic methods.
Q: What purity levels can be achieved with this Hofmann degradation method?
A: The patented process consistently achieves product purity exceeding 99%, with optimized examples reaching up to 99.5%. This high purity is attained through controlled reaction temperatures and a secondary recrystallization purification step.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction conditions are mild (0-20°C initially, then 50-80°C) and easy to control, making it highly suitable for industrial scale-up. The total yield can exceed 90%, ensuring economic viability for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Aminopyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development pipelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering 3-aminopyridine with stringent purity specifications, leveraging advanced analytical capabilities in our rigorous QC labs to guarantee batch-to-batch consistency. Our infrastructure is designed to handle complex chemistries safely, allowing us to offer a reliable supply of this key building block to partners worldwide who demand excellence in both quality and service.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your overall material costs. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your unique synthesis needs, ensuring that we can provide the most effective solution for your supply chain challenges.
